Abstract
-
Background
- The use of biomarkers to predict patient outcomes may be crucial for patients admitted to the intensive care unit (ICU) following surgery because biomarkers guide clinicians in tailoring treatment plans accordingly. Therefore, we aimed to identify potential biomarkers to predict the prognosis of patients with Fournier’s gangrene (FG) admitted to the ICU after surgery.
-
Methods
- We enrolled patients with FG admitted to our hospital between January 2013 and December 2022. We retrospectively analyzed patient characteristics, factors related to management, scores known to be associated with the prognosis of FG, and laboratory data.
-
Results
- The study population included 28 survivors and 13 nonsurvivors. The initial serum lactate level taken in the emergency department; white blood cell, neutrophil, and platelet counts; delta neutrophil index and international normalized ratio; albumin, glucose, HCO3, and postoperative lactate levels; and the laboratory risk indicator for necrotizing fasciitis differed between survivors and nonsurvivors. Postoperative lactate and initial albumin levels were independent predictors of mortality in patients with FG. The postoperative lactate level was the best indicator of mortality (area under the curve, 0.877; 95% confidence interval, 0.711–1.000). The optimal cutoff postoperative lactate level for predicting mortality was 3.0 mmol/L (sensitivity, 80.0%; specificity, 95.0%).
-
Conclusions
- Postoperative lactate and initial albumin levels could be potential predictors of mortality in patients with FG admitted to the ICU after surgery, and the optimal cutoff postoperative lactate and initial albumin levels to predict mortality were 3.0 mmol/L and 3.05 g/dl, respectively.
-
Keywords: albumin; biomarkers; Fournier gangrene; intensive care unit; neutrophil
INTRODUCTION
Fournier’s gangrene (FG) is a rare but serious bacterial infection that affects the genital and perineal regions of the body [1]. It is also known as necrotizing fasciitis of the perineum and can be life-threatening if left untreated. This condition is more common in men than in women [2], and usually occurs in people with weakened immune systems or other underlying health conditions [3]. It is often caused by bacteria such as Escherichia coli, Klebsiella, and Pseudomonas aeruginosa [4].
The symptoms of FG typically begin with pain, swelling, redness in the genital or perineal areas, and fever. As the infection progresses, the affected tissue may become necrotic, leading to skin and muscle death and the formation of pus and gas in the affected area. If left untreated or delayed, FG can lead to various complications such as sepsis, shock, organ failure, and even death [5]. Patients with FG and critical illnesses typically require prompt and aggressive treatment, including antibiotics, surgical debridement, and intensive care management.
Although many studies have evaluated biomarkers to predict disease prognosis [3,6-8], relatively fewer studies have evaluated patients admitted to the intensive care unit (ICU) after surgery. Although patients admitted to the ICU receive medically advanced management, the mortality rate associated FG is still high. The use of biomarkers to predict patient outcomes may be particularly crucial for patients admitted to the ICU following surgery. Therefore, we conducted this study to identify possible biomarkers that can predict the prognosis of patients with FG admitted to the ICU after surgery.
MATERIALS AND METHODS
This retrospective study was approved by the Institutional Review Board of Wonju Severance Christian Hospital (No. CR323018). Since the data were analyzed retrospectively and the identities of the patients were hidden, the requirement for informed consent was waived.
Patient Selection
A total of 84 patients (age ≥18 years) with FG were admitted via the emergency department in Wonju Severance Christian Hospital between January 2013 and December 2022. Medical records and initial computed tomography scans of these patients were reviewed retrospectively. Thirty-three patients who did not undergo surgery and/or were not admitted to the ICU were excluded. Patients with isolated scrotal and perianal abscesses without necrotizing fasciitis of the perineal or inguinal areas were also excluded. Thus, after excluding a total of 43 patients, the remaining 41 patients with FG were included in this study (Figure 1).
Management of FG in Wonju Severance Christian Hospital
Surgical intervention and medical resuscitation were performed to manage the patients with FG. Medical management included initial fluid resuscitation, since patients may present with septic shock. Vasopressors may be added when patients with low blood pressure are unresponsive to fluid resuscitation. The initiation of empirical broad-spectrum antibiotic treatment while awaiting the culture sensitivity results is also important. Targeted antibiotic therapy to cover the organisms associated with FG should be initiated after obtaining the antibiotic-sensitivity results. Radical wide excision of necrotic tissue was performed as an essential element of surgical intervention for patients with FG. Irrigation and drainage were also performed. Patients with suspected significant sphincteric involvement underwent stoma formation to prevent fecal wound contamination. The stoma site was determined intraoperatively.
Definitions and Data Collection
The medical charts of the enrolled patients were retrospectively reviewed. Patient characteristics, including clinical data (age, sex, body mass index, underlying diseases, mean blood pressure, pulse rate, body temperature, septic shock at the initial presentation, surgical treatment, stoma formation, cystostomy formaction), laboratory findings (initial lactate level taken in the emergency department, postoperative lactate level and lactate clearance; white blood cell [WBC], neutrophil, lymphocyte, and platelet counts; delta neutrophil index [DNI] and international normalized ratio [INR]; and C-reactive protein [CRP], procalcitonin, creatinine, and albumin levels), hospital length of stay (LOS), ICU LOS, and overall mortality. The Fournier’s Gangrene Severity Index (FGSI) was calculated using the formula described by Laor et al. [9]. Data for nine parameters, including body temperature, pulse rate, respiratory rate, hematocrit, leukocyte counts, and serum sodium, potassium, creatinine, and bicarbonate levels, were collected for calculation. The Uludag Fournier’s Gangrene severity index (UFGSI) proposed by Yilmazlar et al. [10] was also calculated. Age and disease dissemination scores were added to the FGSI to calculate UFGSI. Scoring was performed during initial presentation at the emergency department. The total WBC count and the hemoglobin, sodium, glucose, serum creatinine, and CRP levels were used to calculate the laboratory risk indicator for necrotizing fasciitis (LRINEC) [11]. The neutrophil-to-lymphocyte ratio was calculated.
Statistical Analysis
Continuous variables are presented as mean (standard deviation) or median (interquartile range), and a comparative analysis was performed using Student t-test or the Mann-Whitney test. Categorical variables were analyzed using the chi-square test with Yates’ correction for continuity and Fisher’s exact test. Multivariate analysis was performed using logistic regression to identify independent risk factors. A receiver operating characteristic (ROC) curve was constructed and the Youden Index method was used to determine the optimal cutoff values for predicting mortality. Statistical analyses were performed using R statistical software (ver. 4.1.0; R Foundation for Statistical Computing, Vienna, Austria). Statistical significance was set at P<0.05.
RESULTS
Patient Characteristics in the Survivor and Nonsurvivor Groups
There was a significant difference in the mean age observed between the survivor group and the nonsurvivor group (59.9±13.5 vs. 63.7±14.2, P=0.421). The proportion of male patients did not differ between the two groups (25 [89.3%] vs. 10 [76.9%], P=0.361). The nonsurvivor group showed significantly more patients with liver diseases (6 [10.3%] vs. 9 [33.3%], P=0.015). A significant difference was observed in the initial lactate level (2.1±1.6 vs. 7.0±4.8 mmol/L, P=0.004), postoperative lactate level (1.8±0.9 vs. 5.8±4.3 mmol/L, P=0.016), initial WBC count (×109/L) (16.1±9.4 vs. 9.0±6.0 ×109/L, P=0.006), initial neutrophil count (14.5±9.0 vs. 7.9±5.5 ×109/L, P=0.006), initial platelet count (208.9±128.7 vs. 112.2±111.3 ×109/L, P=0.020), DNI (7.9%±10.1% vs. 27.6%±24.4%, P=0.014), albumin level (3.2±0.8 vs. 2.4±0.5 g/dl, P<0.001), INR (1.2±0.2 vs. 1.7±0.7, P=0.043), glucose level (209.9±141.0 vs. 128.3±48.0 mg/dl, P=0.009), and HCO3 level (22.6±4.4 vs. 17.1±6.9 mmol/L, P=0.018) between the survivor and nonsurvivor groups. The nonsurvivor group demonstrated a significantly higher LRINEC score (7.5±2.8 vs. 5.7±1.5, P=0.010) compared to the survivor group. Table 1 provides a comprehensive description of all the details.
Identification of Independent Risk Factors for Predicting Mortality in Patients with FG
A logistic regression analysis was conducted to assess independent risk factors, which encompassed variables such as age, sex, body mass index, initial lactate level, postoperative lactate level, WBC count, neutrophil count, platelet count, DNI, albumin level, INR, glucose level, HCO3 level, and LRINEC. The following risk factors were independently correlated with mortality: postoperative lactate level (odds ratio [OR], 2.87; 95% confidence interval [CI], 1.18–7.02; P=0.021), and initial albumin level (OR, 0.07; 95% CI, 0.01–0.49; P=0.007) (Table 2).
Microbial Pathogens Identified in Cases of FG
In this study, E. coli was the predominant organism identified among the enrolled patients (n=15, 21.1%). Additionally, other organisms such as Klebsiella species (18.3%), Streptococci (15.5%), and Enterococci (14.1%) were also detected (Table 3).
Optimal Cutoff Values of the Postoperative Lactate and Initial Albumin Levels
ROC curves were generated for postoperative lactate and initial albumin levels to predict mortality in patients diagnosed with FG. The area under the curve (AUC) for postoperative lactate was 0.877 (95% CI, 0.711–1.000), while for initial albumin, it was 0.827 (95% CI, 0.738–0.997). The optimal cutoff value for postoperative lactate was determined as 3.0 mmol/L, with a sensitivity of 80.0% and specificity of 95.0%. Similarly, the optimal cutoff value for initial albumin was 3.05 g/dl, with a sensitivity of 92.3% and specificity of 57.1%. Detailed results can be found in Table 4 and Figure 2.
DISCUSSION
Our results showed that the postoperative lactate and preoperative albumin levels were major predictors in ICU patients with FG. The optimal cutoff values for serum postoperative lactate and initial albumin levels were 3.0 mmol/L (sensitivity, 80.0%; specificity, 95.0%) and 3.05 g/dl (sensitivity, 92.3%; specificity, 57.1%), respectively.
The serum lactate level is a widely utilized biomarker in critically ill patients that indicates the degree of tissue hypoxia and anaerobic metabolism [12]. Under conditions such as sepsis, shock, and respiratory failure, inadequate oxygen supply to tissues triggers anaerobic metabolism, resulting in lactate accumulation in the bloodstream [13]. Significant differences were observed in the initial lactate levels between survivors and nonsurvivors during the univariate analysis, but not in the multivariate analysis. In contrast, the postoperative lactate levels can be influenced by resuscitation after initial evaluation and surgical excision. Therefore, the postoperative lactate level may be a more accurate biomarker to predict mortality than the initial lactate level because the postoperative lactate level reflects the patient’s condition after initial treatment.
Although the AUC of the serum albumin level was lower than that of the postoperative lactate level, the serum albumin level emerged as an independent predictor of mortality in patients diagnosed with FG. Hypoalbuminemia can occur in critically ill patients and is influenced by various factors, including physiological responses triggered by infection [14]. Serum albumin levels can be affected by changes in albumin synthesis and catabolism as well as increased endothelial permeability leading to albumin loss into the extravascular space [14]. The latter mechanism seems to play a substantial role in hypoalbuminemia observed in patients with metabolic stress, leading to the redistribution of albumin from the intravascular to the interstitial compartment. Additionally, serum albumin levels can reflect a patient's nutritional status and immune function [15]. Therefore, hypoalbuminemia may contribute to disease progression and, conversely, a worsened disease course may further reduce serum albumin levels. A previous study indicated that hypoalbuminemia could serve as a predictive factor for mortality in patients diagnosed with FG [16].
In this study, despite the significant difference observed in LRINEC scores between survivors and nonsurvivors during the univariate analysis and its inclusion in the regression model, LRINEC was not identified as an independent predictor of mortality. Wong et al. [11], who constructed the LRINEC, demonstrated that it is considered a robust scoring system that can effectively identify even clinically early cases of necrotizing fasciitis. The LRINEC includes six laboratory markers that are important for critically ill patients. However, the patients enrolled to construct this scoring system may be not systemically ill patients with severe necrotizing fasciitis. Insufficient information was available to understand the patient’s condition. Because our study was based on critically ill patients admitted to the ICU, the LRINEC may not be accurate for predicting mortality. Previous studies have identified other scores for FG, such as the FGSI and the UFGSI, as possible predictive markers of poor outcomes [4,17-20]. However, in this study, FGSI and UFGSI scores did not differ between survivors and nonsurvivors. The observed differences in results between our study and previous studies are likely influenced by the combination of low FG prevalence, small sample sizes in both our study and previous studies, and the specific patient selection criteria focusing on ICU-admitted patients in our research. Data from 80 patients were used to construct the UFGSI, but mortality occurred in only 17 patients. In addition, only 21 of these 80 patients were admitted to the ICU [18]. Therefore, the UFGSI may show limited ability to predict outcomes in critically ill patients.
The DNI is a laboratory parameter that examines leukocyte differentials by utilizing two independent channels, namely the myeloperoxidase channel and the lobularity/nuclear density channel [21]. The DNI is derived by quantifying the variance between leukocyte differentials measured in the myeloperoxidase and lobularity/nuclear density channels, providing an indication of the proportion of immature granulocytes present in the bloodstream [21]. Numerous studies have demonstrated that the DNI can serve as a valuable biomarker in predicting disease severity and prognosis in patients with diverse infectious conditions or sepsis [3,22,23]. However, the DNI was not an independent predictor in this study, although it was significantly higher in nonsurvivors in the univariate analysis. In this study, because the target patient group was analyzed only for patients admitted to an ICU, we assumed that the difference in DNI between patients was not significant.
Several limitations need to be considered in this study. Firstly, there was a possibility of selection bias due to its retrospective nature. Secondly, the sample size was relatively small since only patients from a single center were included, which may have affected the representativeness of the enrolled patients. Thirdly, potential variations in the timing and approaches to patient management might have been present due to the involvement of different physicians treating the enrolled patients throughout the study period. Lastly, as the analysis encompassed patient data collected over an extended duration, certain confounding factors, such as advancements in ICU care over time, may not have been adequately addressed.
In conclusion, the postoperative lactate and initial albumin levels could be potential predictors of mortality in patients with FG who were admitted to the ICU after surgery, and the optimal cutoff postoperative lactate and initial albumin levels to predict mortality were 3.0 mmol/L and 3.05 g/dl, respectively. To validate our findings, it is essential to conduct large-scale multicenter prospective studies.
KEY MESSAGES
▪ The use of biomarkers to predict patient outcomes may be particularly crucial for patients admitted to the intensive care unit (ICU) following surgery.
▪ Postoperative lactate and initial albumin levels could be potential predictors of mortality in patients with Fournier’s gangrene admitted to the ICU after surgery.
NOTES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
FUNDING
None.
-
AUTHOR CONTRIBUTIONS
Conceptualization: ISS. Data curation: ISS. Formal analysis: KK. Methodology: ISS, SA, SCG. Project administration: ISS. Visualization: SA, SCG. Writing – original draft: ISS. Writing – review & editing: ISS, KK.
Acknowledgments
None.
Figure 1.Flowchart of the study.

Figure 2.Receiver operating characteristics curves for the lactate level and the albumin level between the survivors and nonsurvivors.
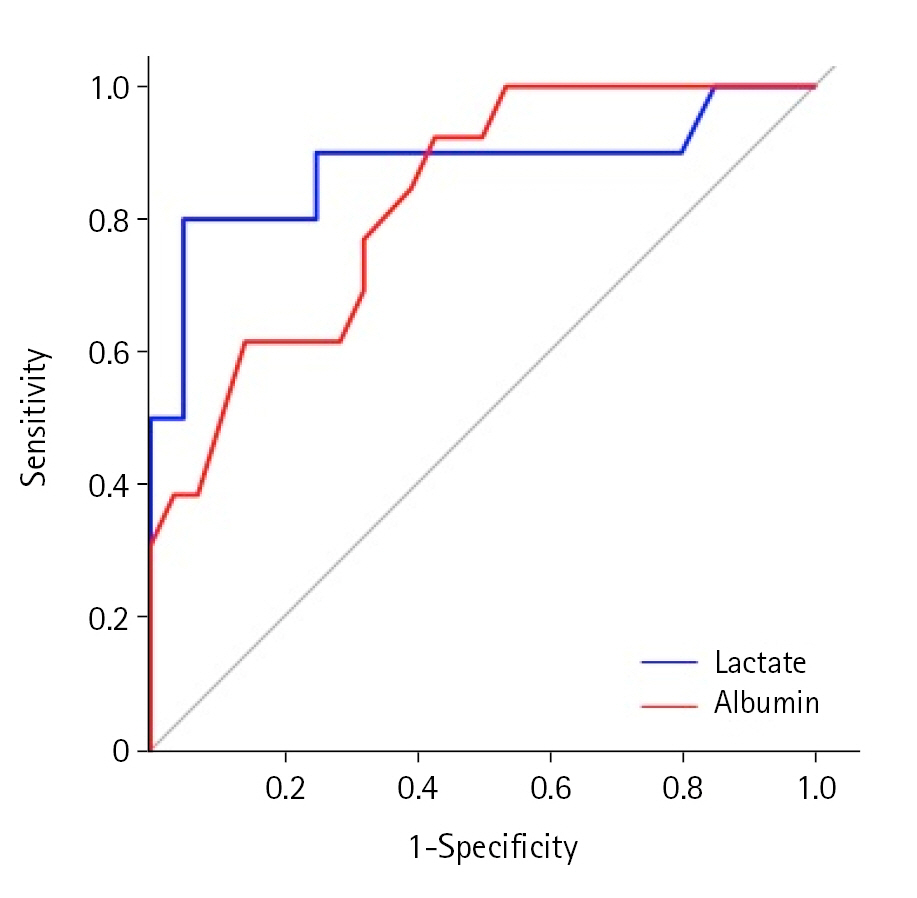
Table 1.Patient characteristics in the survivor and nonsurvivor groups
|
Variable |
Survivor (n=28) |
Nonsurvivor (n=13) |
P-value |
|
Age (yr) |
60±14 |
64±14 |
0.421 |
|
Male |
25 (89.3) |
10 (76.9) |
0.361a)
|
|
Body mass index (kg/m2) |
25.1±5.9 |
21.7±3.5 |
0.028 |
|
Underlying disease |
|
|
|
|
Hypertension |
15 (53.6) |
3 (23.1) |
0.136 |
|
Diabetes mellitus |
12 (42.9) |
7 (53.8) |
0.749 |
|
Liver disease |
5 (17.9) |
6 (46.2) |
0.073a)
|
|
Renal disease |
4 (14.3) |
1 (7.7) |
0.998a)
|
|
Pulmonary disease |
0 |
1 (7.7) |
0.317a)
|
|
Blood pressure (mm Hg) |
82.5±18.9 |
77.1±16.9 |
0.366 |
|
Pulse rate |
101.6±23.0 |
111.8±26.1 |
0.239 |
|
Body temperature (℃) |
37.5±0.9 |
37.2±1.2 |
0.379 |
|
Septic shock at the initial presentation |
5 (17.9) |
3 (23.1) |
0.692 |
|
Stoma formation |
15 (53.6) |
7 (53.8) |
0.999 |
|
Cystostomy |
2 (7.1) |
0 |
0.999a)
|
|
Laboratory finding |
|
|
|
|
Initial lactate (mmol/L) |
2.1±1.6 |
7.0±4.8 |
0.004 |
|
Postoperative lactate (mmol/L) |
1.8±0.9 |
5.8±4.3 |
0.016 |
|
Lactate clearance |
0.5±1.6 |
2.0±3.2 |
0.168 |
|
Initial WBC count (×109/L) |
16.1±9.4 |
9.0±6.0 |
0.006 |
|
Initial neutrophil count (×109/L) |
14.5±9.0 |
7.9±5.5 |
0.006 |
|
Initial lymphocyte count (×109/L) |
0.7±0.4 |
0.5±0.2 |
0.067 |
|
Initial hemoglobin (g/dl) |
11.7±2.2 |
10.9±2.6 |
0.367 |
|
Initial platelet count (×109/L) |
208.9±128.7 |
112.2±111.3 |
0.020 |
|
Initial DNI (%) |
7.9±10.1 |
27.6±24.4 |
0.014 |
|
Initial CRP level (mg/dl) |
21.7±9.6 |
20.0±16.4 |
0.731 |
|
Initial PCT level (ng/ml) |
5.4±7.0 |
13.8±12.8 |
0.119 |
|
Initial albumin level (g/dl) |
3.2±0.8 |
2.4±0.5 |
<0.001 |
|
Initial INR |
1.2±0.2 |
1.7±0.7 |
0.043 |
|
Initial creatinine level (mg/dl) |
2.1±2.1 |
2.6±2.0 |
0.439 |
|
Initial glucose level (mg/dl) |
209.9±141.0 |
128.3±48.0 |
0.009 |
|
Initial HCO3 level (mmol/L) |
22.6±4.4 |
17.1±6.9 |
0.018 |
|
FGSI |
7.0±3.6 |
8.6±5.0 |
0.301 |
|
UFGSI |
9.7±3.7 |
11.9±6.2 |
0.250 |
|
LRINEC |
5.7±1.5 |
7.5±2.8 |
0.010 |
|
Neutrophil-lymphocyte ratio |
24.5±19.0 |
16.5±14.0 |
0.142 |
|
Hospital length of stay (day) |
55.9±52.0 |
37.2±43.2 |
0.240 |
|
ICU length of stay (day) |
5.0±4.2 |
22.5±40.7 |
0.149 |
Table 2.Multivariate analysis using a logistic regression model to predict mortality
|
Variable |
Univariate analysis
|
Multivariate analysis
|
|
HR (95% CI) |
P-value |
HR (95% CI) |
P-value |
|
Male |
2.50 (0.43–14.54) |
0.308 |
|
|
|
Initial lactate (mmol/L) |
1.71 (1.16–2.54) |
0.007 |
|
|
|
Initial WBC count (×109/L) |
1.00 (1.00–1.00) |
0.027 |
|
|
|
Initial neutrophil count (×109/L) |
1.00 (1.00–1.00) |
0.028 |
|
|
|
Initial platelet count (×109/L) |
0.99 (0.99–1.00) |
0.034 |
|
|
|
Initial DNI (%) |
1.07 (1.02–1.12) |
0.007 |
|
|
|
Initial INR |
31.02 (1.54–622.99) |
0.025 |
|
|
|
Initial glucose level (mg/dl) |
0.99 (0.98–1.00) |
0.083 |
|
|
|
Initial HCO3 level (mmol/L) |
0.83 (0.71–0.96) |
0.012 |
|
|
|
Age (yr) |
1.02 (0.97–1.08) |
0.401 |
0.99 (0.92–1.07) |
0.856 |
|
Postoperative lactate (mmol/L) |
2.87 (1.18–7.02) |
0.021 |
2.87 (1.18–7.02) |
0.021 |
|
Initial albumin level (g/dl) |
0.09 (0.02–0.48) |
0.005 |
0.07 (0.01–0.49) |
0.007 |
|
LRINEC |
1.41 (1.01–2.00) |
0.044 |
1.47 (0.97–2.22) |
0.066 |
Table 3.Causative bacterial organisms for Fournier’s gangrene
|
Bacterial organism |
Total (n=71) |
|
Escherichia coli
|
15 (21.1) |
|
Klebsiella species |
13 (18.3) |
|
Streptococci |
11 (15.5) |
|
Enterococci |
10 (14.1) |
|
Staphylococci |
10 (14.1) |
|
Acinetobacter species |
3 (4.2) |
|
Pseudomonas
|
2 (2.8) |
|
Candida species |
2 (2.8) |
|
Gram positive rods |
1 (1.4) |
|
Others |
4 (5.6) |
Table 4.Characteristics of postoperative lactate and initial albumin levels
|
Variable |
Optimal cutoff value |
Sensitivity (%) |
Specificity (%) |
AUC (95% CI) |
|
Postoperative lactate (mmol/L) |
3.00 |
80.0 |
95.0 |
0.877 (0.711–1.000) |
|
Initial albumin (g/dl) |
3.05 |
92.3 |
57.1 |
0.827 (0.738–0.997) |
References
- 1. Carroll PR, Cattolica EV, Turzan CW, McAninch JW. Necrotizing soft-tissue infections of the perineum and genitalia. Etiology and early reconstruction. West J Med 1986;144:174-8.ArticlePubMedPMC
- 2. Orhan E, Şenen D. Using negative pressure therapy for improving skin graft taking on genital area defects following Fournier gangrene. Turk J Urol 2017;43:366-70.ArticlePubMedPMC
- 3. Shin IS, Gong SC, An S, Kim K. Delta neutrophil index as a prognostic factor for mortality in patients with Fournier's gangrene. Int J Urol 2022;29:1287-93.ArticlePubMedPDF
- 4. Garg G, Singh V, Sinha RJ, Sharma A, Pandey S, Aggarwal A. Outcomes of patients with Fournier's Gangrene: 12-year experience from a tertiary care referral center. Turk J Urol 2019;45:S111-6.ArticlePubMedPMC
- 5. Gadler T, Huey S, Hunt K. Recognizing Fournier's gangrene in the emergency department. Adv Emerg Nurs J 2019;41:33-8.ArticlePubMed
- 6. Kim KM, Seong SH, Won DY, Ryu H, Kim IY. The prognostic factors and severity index in Fournier’s gangrene. J Korean Soc Coloproctol 2010;26:29-33.Article
- 7. Eray IC, Dalci K, Gumus S, Yalav O, Saritas AG, Boz A, et al. The role of C-reactive protein ratio in predicting mortality in patients with Fournier gangrene. Ann Coloproctol 2023;39:223-30.ArticlePubMedPDF
- 8. Friederichs J, Hutter M, Hierholzer C, Novotny A, Friess H, Bühren V, et al. Procalcitonin ratio as a predictor of successful surgical treatment of severe necrotizing soft tissue infections. Am J Surg 2013;206:368-73.ArticlePubMed
- 9. Laor E, Palmer LS, Tolia BM, Reid RE, Winter HI. Outcome prediction in patients with Fournier's gangrene. J Urol 1995;154:89-92.ArticlePubMed
- 10. Yilmazlar T, Ozturk E, Ozguc H, Ercan I, Vuruskan H, Oktay B. Fournier's gangrene: an analysis of 80 patients and a novel scoring system. Tech Coloproctol 2010;14:217-23.ArticlePubMedPDF
- 11. Wong CH, Khin LW, Heng KS, Tan KC, Low CO. The LRINEC (Laboratory Risk Indicator for Necrotizing Fasciitis) score: a tool for distinguishing necrotizing fasciitis from other soft tissue infections. Crit Care Med 2004;32:1535-41.ArticlePubMed
- 12. Kushimoto S, Akaishi S, Sato T, Nomura R, Fujita M, Kudo D, et al. Lactate, a useful marker for disease mortality and severity but an unreliable marker of tissue hypoxia/hypoperfusion in critically ill patients. Acute Med Surg 2016;3:293-7.ArticlePubMedPMCPDF
- 13. Li X, Yang Y, Zhang B, Lin X, Fu X, An Y, et al. Lactate metabolism in human health and disease. Signal Transduct Target Ther 2022;7:305. ArticlePubMedPMCPDF
- 14. Wiedermann CJ. Hypoalbuminemia as surrogate and culprit of infections. Int J Mol Sci 2021;22:4496. ArticlePubMedPMC
- 15. Moon JJ, Kim Y, Kim DK, Joo KW, Kim YS, Han SS. Association of hypoalbuminemia with short-term and long-term mortality in patients undergoing continuous renal replacement therapy. Kidney Res Clin Pract 2020;39:47-53.ArticlePubMedPMC
- 16. Tuncel A, Aydin O, Tekdogan U, Nalcacioglu V, Capar Y, Atan A. Fournier's gangrene: three years of experience with 20 patients and validity of the Fournier's Gangrene Severity Index Score. Eur Urol 2006;50:838-43.ArticlePubMed
- 17. Sabzi Sarvestani A, Zamiri M, Sabouri M. Prognostic factors for Fournier's gangrene: a 10-year experience in Southeastern Iran. Bull Emerg Trauma 2013;1:116-22.PubMedPMC
- 18. Üreyen O, Acar A, Gökçelli U, Atahan MK, İlhan E. Usefulness of FGSI and UFGSI scoring systems for predicting mortality in patients with Fournier's gangrene: a multicenter study. Ulus Travma Acil Cerrahi Derg 2017;23:389-94.PubMed
- 19. Hong KS, Yi HJ, Lee RA, Kim KH, Chung SS. Prognostic factors and treatment outcomes for patients with Fournier's gangrene: a retrospective study. Int Wound J 2017;14:1352-8.ArticlePubMedPMCPDF
- 20. Noegroho BS, Siregar S, Mustafa A, Rivaldi MA. Validation of FGSI scores in predicting Fournier gangrene in tertiary hospital. Res Rep Urol 2021;13:341-6.ArticlePubMedPMCPDF
- 21. Nahm CH, Choi JW, Lee J. Delta neutrophil index in automated immature granulocyte counts for assessing disease severity of patients with sepsis. Ann Clin Lab Sci 2008;38:241-6.PubMed
- 22. Kim JW, Park JH, Kim DJ, Choi WH, Cheong JC, Kim JY. The delta neutrophil index is a prognostic factor for postoperative mortality in patients with sepsis caused by peritonitis. PLoS One 2017;12:e0182325.ArticlePubMedPMC
- 23. Kong T, Park YS, Lee HS, Kim S, Lee JW, You JS, et al. The delta neutrophil index predicts the development of in-hospital hypotension in initially stable patients with pyogenic liver abscess. Sci Rep 2019;9:12105. ArticlePubMedPMCPDF
Citations
Citations to this article as recorded by

- Risk Factors for Mortality Among Patients With Fournier Gangrene: A Systematic Review
Pavan Shet, Ashmit Daiyan Mustafa, Karan Varshney, Lavina Rao, Sameen Sawdagar, Florence McLennan, Siraaj Ansari, Darshan Shet, Niveshan Sivathamboo, Sian Campbell
Surgical Infections.2024;[Epub] CrossRef
 , Seong Chan Gong
, Seong Chan Gong , Sanghyun An
, Sanghyun An , Kwangmin Kim
, Kwangmin Kim




 KSCCM
KSCCM

 PubReader
PubReader ePub Link
ePub Link Cite
Cite



