Abstract
-
Background
- Hemostatic dysfunction during extracorporeal membrane oxygenation (ECMO) due to blood-circuit interaction and the consequences of shear stress imposed by flow rates lead to rapid coagulation cascade and thrombus formation in the ECMO system and blood vessels. We aimed to identify the incidence and risk factors for cannula-associated arterial thrombosis (CaAT) post-decannulation.
-
Methods
- A retrospective study of patients undergoing arterial cannula removal following ECMO was performed. We evaluated the incidence of CaAT and compared the characteristics, ECMO machine parameters, cannula sizes, number of blood products transfused during ECMO, and daily hemostasis parameters in patients with and without CaAT. Multivariate analysis identified the risk factors for CaAT.
-
Results
- Forty-seven patients requiring venoarterial ECMO (VA-ECMO) or hybrid methods were recruited for thrombosis screening. The median Sequential Organ Failure Assessment score was 11 (interquartile range, 8–13). CaAT occurred in 29 patients (61.7%), with thrombosis in the superficial femoral artery accounting for 51.7% of cases. The rate of limb ischemia complications in the CaAT group was 17.2%. Multivariate analysis determined that the ECMO flow rate–body surface area (BSA) ratio (100 ml/min/m2) was an independent factor for CaAT, with an odds ratio of 0.79 (95% confidence interval, 0.66–0.95; P=0.014).
-
Conclusions
- We found that the incidence of CaAT was 61.7% following successful decannulation from VA-ECMO or hybrid modes, and the ECMO flow rate–BSA ratio was an independent risk factor for CaAT. We suggest screening for arterial thrombosis following VA-ECMO, and further research is needed to determine the risks and benefits of such screening.
-
Keywords: blood flow; cannula-associated thrombosis; extracorporeal membrane oxygenation
INTRODUCTION
Venoarterial extracorporeal membrane oxygenation (VA-ECMO) is being used increasingly to support patients with cardiogenic shock refractory to conventional therapies [1]. The use of ECMO can result in hemostatic dysfunction through direct exposure of the patient's blood to non-biomaterials inherent in the ECMO system. This exposure can activate endogenous and exogenous coagulation cascades, platelets, leukocytes, and complements. Additionally, the effects of shear stress may promote the formation of thrombi, particularly in low blood flow positions [2,3]. Moreover, increasing stage of extracorporeal circulation increases the risk of thrombosis in critically ill patients with multiple disease-related thrombotic risk factors [2].
Limb ischemia complications with multifactorial etiologies can develop at any stage of ECMO support, including during cannulation, during support, and after decannulation, resulting in increased morbidity and mortality [4-6]. Several studies have reported a high prevalence of limb ischemia complications during ECMO support. The primary risk factors identified for such complications are female gender, younger age, and preexisting peripheral vascular disease. Additionally, using large cannulas during ECMO can lead to flow obstruction [5-8]. However, limb ischemia due to thrombosis can be high even after decannulation. The formation of arterial thrombosis in VA-ECMO is potentially more frequent in the low-flow space segment because of the low blood flow between the arterial cannula and the distal perfusion cannula [4,9,10] (Figure 1). If left unnoticed and inappropriately managed, arterial lumen occlusion could result in acute limb ischemia [4].
The risk factors for venous thrombosis following cannula removal have been well documented [11-14]. Conversely, there has been limited research into cannula-associated arterial thrombosis (CaAT) after decannulation. Reported incidence rates of CaAT in patients undergoing VA-ECMO following decannulation have varied widely, ranging from 5% to 41% across medical centers. Furthermore, research exploring the prevalence of CaAT in Asian countries is scarce [15-18]. To the best of our knowledge, there is a lack of studies that have identified risk factors associated with development of CaAT after ECMO post-decannulation [15]. Therefore, we conducted this study to determine the incidence of CaAT and to identify the risk factors associated with it in patients requiring VA-ECMO.
MATERIALS AND METHODS
Study Design and Population
This was a retrospective observational study. Consecutive patients requiring ECMO due to cardiogenic shock at the intensive care unit (ICU) were enrolled from January 2019 to February 2020. Exclusion criteria were age younger than 18 years, duration of ECMO support less than 24 hours, pulmonary embolism receiving fibrinolytic therapy, not screened for thrombosis after cannula removal, death during ECMO, and missing data.
Data Collection
We collected patient characteristics, including age; sex; body mass index; body surface area (BSA); diagnoses; comorbidities; pre-ECMO severity scores, including Sequential Organ Failure Assessment (SOFA), and Acute Physiology and Chronic Health Evaluation (APACHE) II scores; and daily hemostatic parameters, including platelet count, prothrombin time (PT), activated partial thromboplastin time ratio (aPTTr), fibrinogen, anti-Xa, antithrombin-III activity, ECMO machine parameters, and anticoagulation dose. In VA-ECMO, BSA affects the ECMO flow rate and the choice of arterial cannula size [6]. Therefore, we recorded the cannula–BSA ratio (Fr/m2), which is the arterial cannula size divided by BSA, and the ECMO flow rate–BSA ratio (100 ml/min/m2), which is the ECMO flow rate divided by BSA.
The primary outcome was limb ischemia complications, and the secondary outcomes were local limb complications (including infection at removal cannula, pseudoaneurysm, and limb amputation), ICU length of stay, hospital length of stay, and hospital mortality.
Definitions
Cannula-associated arterial thrombosis
CaAT was diagnosed when one of the following criteria was met: (1) Arterial thrombus during decannulation was recorded in the documentation of surgical vascular closure in the operating room or (2) intraluminal arterial thrombus together with abnormal peak systolic velocity distal to the occluded segments of limb decannulation was detected by bedside Doppler ultrasound within 24 hours of percutaneous vascular closure [19-21]. Bedside Doppler ultrasound was performed by sonographers, and thrombosis was confirmed by at least two sonographers.
Limb ischemia complications
A diagnosis of limb ischemia following cannula removal occurred with one of the following criteria: (1) a decrease of regional oxygen saturation (rSO2) measured by near-infrared spectroscopy (NIRS) less than 50% in the decannulated limb for a duration exceeding four minutes or (2) a difference greater than 15% between rSO2 values of the decannulated limb and the contralateral limb by NIRS [22].
ECMO Data
The ECMO system in our ICU comprised a Rotaflow console, a permanent life support membrane, and cannulas from the Maquet, Getinge Group. The cannula size ranged from 15 to 17 Fr for arterial cannulas, from 5 to 7 Fr Super Arrow-Flex sheath for distal perfusion cannulas, and from 21 to 25 Fr for venous cannulas. The ECMO circuit was implemented in the ICU, and cannulas were placed at the bedside by trained intensivists using the percutaneous Seldinger technique under ultrasound guidance [23].
Anticoagulation Strategies and ECMO Weaning
After a loading dose, unfractionated heparin (UFH) was titrated to target an aPTTr of 1.5–2.5 or an anti-Xa of 0.3–0.7 UI/ml during ECMO support. To wean a patient from ECMO, the ECMO blood flow was gradually decreased by 0.5 L/min while the hemodynamic status of the patient, including blood pressure, rhythm, and bedside cardiac output measurement, was evaluated. Low-flow rates may trigger coagulation in the ECMO system; therefore, once the ECMO flow rate was reduced to 1.0 L/min, a bolus of UFH or an increase in intravenous heparin dose was needed. Once the decision was made to withdraw the cannula, the ECMO blood flow was increased to >2.5 L/min, and heparin was stopped during preparation for decannulation. The arterial cannula was removed either by a percutaneous vascular closure technique using Proglide devices (Abbott) at the bedside or by surgical vascular closure in the operating room. After decannulation, the dosing of UFH or enoxaparin was at the discretion of the attending physician based on the risk of bleeding and thrombosis. Our center uses the NIRS machine (Nonin SenSmart Model X-100) with sensors placed on the lateral anterior side of the shins to monitor the perfusion status of both legs. This monitoring apparatus is implemented immediately after ECMO support until the patient is discharged from the ICU.
Statistical Analysis
Statistical analysis was performed using R Statistical Software (ver. 3.6.2; R Foundation for Statistical Computing). Categorical variables are presented as numerical data and percentages and were analyzed using Fisher’s exact test. Continuous variables were first tested for normality using the Shapiro-Wilk test. Variables with and without a normal distribution are reported as the mean±standard deviation and median (interquartile range [IQR]), respectively. Comparisons of continuous variables between two groups with and without CaAT were conducted using Student t-test and the Mann-Whitney U-test when appropriate. Univariate and multivariate logistic regression was used to estimate the odds ratio (OR) with a 95% confidence interval (CI) to investigate risk factors for CaAT. For all statistical tests, a two-sided P-value <0.05 was considered statistically significant.
Medical Ethics
The study was approved by the Ethics Committee in Biomedical Research of the University of Medicine and Pharmacy on February 26, 2022 (No. 122/HDĐ-ĐHYD). Informed consent was waived due to the retrospective nature of the study.
RESULTS
Baseline Characteristics of the Patients
Among the 85 patients who were supported by VA-ECMO and hybrid modes from January 2019 to February 2020, 38 were eliminated based on exclusion criteria (Figure 2), and 47 patients (39 receiving VA-ECMO and 8 receiving hybrid ECMO) were included in the final analysis. The median age was 33 years (26–46 years). Fewer patients were male (16 patients, 34%) than female. Acute myocarditis accounted for 68.1% of the main indications for ECMO. The median SOFA and APACHE II scores were 11 (IQR, 8–13) and 18 (IQR, 12.5–25), respectively. Three patients had a history of diabetes, and one patient had peripheral vascular disease. The median cannula–BSA ratio was 10.1 Fr/m2, and the difference between the two groups was not significant. The ECMO system characteristics and outcomes between the CaAT and non-CaAT groups are presented in Tables 1 and 2.
CaAT and Outcomes
Forty-seven patients with arterial cannulas were successfully weaned from ECMO. CaAT occurred in 29 patients (61.7%); among them, superficial femoral artery thrombosis was reported in 15 (51.7%), and common femoral artery thrombosis was reported in 5 (20.6%). The distribution of patients with CaAT is shown in Figure 3. Additionally, 21 of 47 patients (44.7%) underwent surgical vascular closure in the operating room, and 55.3% of patients underwent percutaneous vascular closure at the bedside. No significant difference was observed in the incidence of CaAT between the two groups, with rates of 51.7% and 48.3%, respectively. After cannula removal, 41 patients (87.2%) were administered UFH or enoxaparin to prevent or treat thromboembolism.
In our study, limb ischemia occurred in 5 of 29 patients with CaAT (17.2%), and none occurred in the other group. Arterial thrombosis was detected in all patients with limb ischemia. None of them had peripheral arterial disease. Four patients were effectively treated with anticoagulant, and one patient with progressive limb ischemia required surgical thrombectomy. All five patients survived and were discharged. We observed other vascular complications, such as infection at the removal site or pseudoaneurysm, as in Table 2. The hospital mortality rates were not different between the two groups.
Risk Factors for CaAT
There were no significant differences in age or sex between the two groups, and arterial cannula and distal perfusion cannula sizes were not significantly different. The number of blood products transfused during ECMO was higher in the CaAT group, but the difference was not significant. At our center, cannula removal is performed using two methods: (1) surgical vascular closure and (2) percutaneous vascular closure. There is no significant difference in the thrombosis rate between the two methods. The duration of ECMO support was similar in the groups with and without CaAT (6 days [IQR, 4–8] vs. 6 days [IQR, 4–7], P=0.991, respectively). The ICU length of stay was 9.0 days (IQR, 7.0–12.0) vs. 9.50 days (IQR, 8.0–21.2) (P=0.582). Risk factors among the characteristics of patients and ECMO systems are presented in Table 1.
The hemostasis parameters are presented in Table 3. We found that the minimum and maximum fibrinogen concentrations were significantly lower in the group with CaAT (P<0.05). In addition, the number of days with platelets <150 g/L as the highest count was also significantly different between the two groups (P<0.05).
Univariate logistic regression showed that the maximum fibrinogen concentration, number of days with fibrinogen >4 g/dl, and ECMO flow rate–BSA ratio were associated with CaAT. A multivariable logistic regression model for CaAT prediction revealed that only the mean ECMO flow rate–BSA ratio was an independent risk factor for CaAT, with an OR of 0.79 (95% CI, 0.66–0.95; P=0.014) (Table 4).
DISCUSSION
VA-ECMO use for refractory cardiogenic shock has markedly increased over the last decade, but hemostatic dysfunction in ECMO-supported patients remains a challenge for clinicians [9,23]. In particular, thrombotic complications after ECMO decannulation are underdiagnosed [24]. The cannula-associated deep vein thrombosis (CaDVT) rate following ECMO cannula removal ranges from 18% to 85%, and risk factors for CaDVT have been reported [11-14]. However, the incidence of CaAT after VA-ECMO decannulation has not been clearly defined. Recent studies have shown that the incidence of CaAT ranges from 5% to 41.0% [15,16,18]. The different incidence rates of CaAT among studies depend on two main causes: (1) the difference in diagnostic techniques among studies, including Doppler vascular ultrasound, vascular computed tomography (CT) scan, clinically demonstrated hypoperfusion, and surgical protocol records at the time of ECMO cannula removal and (2) the different rate of thrombosis screening post-ECMO weaning among centers. Some centers investigate CaAT based on suggestive clinical signs of limb hypoperfusion after cannula removal. Thrombosis is identified less frequently by Doppler vascular ultrasound or clinical signs of hypoperfusion than by surgical/autopsied visualization. The underestimation of arterial thromboembolic events can occur in more than 25% of patients [9,24]. In recent reports, Doppler vascular ultrasound or CT scanning of blood vessels was used to screen for CaAT when symptoms and signs of limb ischemia appeared, leading to a relatively low CaAT rate of 5%–12.5% [18,25]. In contrast, Cartwright et al. [16] reported that routine arterial thrombosis screening by Doppler vascular ultrasound could detect CaAT in up to 41% of patients after cannula removal. The present study found a higher rate of CaAT (61.7%) than in other studies [15-18]. Surgical vascular closure was performed in the operating room for up to 44.6% of patients undergoing cannula removal. Vascular surgeons routinely used a Fogarty catheter to screen for thrombosis during the operation, which may explain the higher rate of thrombosis detection. All patients whose cannulas were removed by percutaneous vascular closure with Proglide devices (Abbott Vascular) were routinely screened by vascular Doppler ultrasound for thrombosis, which also explains the high rate of CaAT in this study (Figure 4). In addition, a distal perfusion cannula, inserted in a common site of thrombus formation, was routinely inserted at our ECMO center.
The arterial cannula inserted from the common femoral artery and the distal perfusion cannula inserted from the superficial femoral artery can create a low-flow or absent-flow between them (Figure 1). Low or absent flow at this space of the femoral artery leads to an extremely high risk of thrombus formation. This may explain why some clinicians favor surgical decannulation over percutaneous decannulation to remove thrombi [9]. In our center, a distal perfusion cannula is routinely inserted in patients supported by VA-ECMO. Therefore, the superficial and common femoral arteries were the most common sites of CaAT, with thrombosis rates of 51.7% and 20.6%, respectively. Although the incidence of CaAT was high, only 17.2% of this group had limb ischemia complications. The routine use of a Fogarty catheter at surgical vascular closure, daily Doppler vascular ultrasound, and appropriate heparin dosing after decannulation can explain the low incidence of limb ischemia. Furthermore, based on the principle of "less is more," our center tends to choose arterial cannulas with a small size of 15–17 Fr (92.5%) to minimize vascular wall damage and thrombus formation [5,6]. Additionally, BSA is an essential factor to consider, as it can influence the selection of arterial cannula size and flow rate in VA-ECMO supports [6,26]. A higher cannula–BSA ratio (>11 Fr/m2) is associated with an increased incidence of lower limb ischemia [6]. In our study, the median cannula–BSA ratio was 10.1. This ratio was less than 11 Fr/m2 and was similar in the two groups, suggesting that BSA may not affect the discriminative ability of ECMO flow rate for arterial thrombosis. We suggest screening for arterial thrombosis following VA-ECMO support for early detection of complications, optimal utilization of anticoagulation after decannulation, and early thrombectomy when necessary. However, further research is needed to determine the risks and benefits of such screening.
Patients undergoing VA-ECMO are susceptible to multiple complications that can arise at different stages, with post-decannulation complications being underdiagnosed [9,24]. Bidar et al. [15] observed a post-decannulation CaAT rate of 14% in patients receiving VA-ECMO who were examined using Doppler vascular ultrasonography, despite the absence of risk factors. To the best of our knowledge, the current literature reports no risk factor for CaAT [15,16,18]. We analyzed risk factors for CaAT to minimize the possibility of thrombosis formation. Univariate logistic regression analysis showed that the maximum fibrinogen concentration, number of days of fibrinogen >4 g/dl, and ECMO flow rate–BSA ratio were associated with CaAT. In multivariate logistic regression analysis, the ECMO flow rate–BSA ratio was an independent risk factor for CaAT. For every 100 ml/min/m2 increase in ECMO flow rate–BSA ratio, the odds of developing CaAT decrease by 21%. The ECMO flow rate is the velocity at which the ECMO system pumps blood into the circulation of the patient through the arterial cannula. The higher is the ECMO flow rate, the higher is the blood flow through the low-flow space between the arterial cannula and the distal perfusion cannula, decreasing the possibility of blood clots. Moreover, the lower is the ECMO flow rate, the more slowly is the blood pumped through the arterial cannula, resulting in reduced shear pressure and longer residence time around the artery cannula, leading to increased platelet adhesion and thrombus formation [2,27]. These changes may explain the increased cannula arterial thrombus formation in the low-flow state and warrant adequate anticoagulation during the phases of low ECMO flow rates and weaning trials.
Several limitations should be noted in our investigation. First, it was a single-center retrospective study with a limited sample size. Second, some anti-Xa data were missing, so the complete assessment of anticoagulation during ECMO may not have been accurate. Third, arterial cannulas were removed either by the percutaneous vascular closure technique using ProGlide devices (Abbott) at the bedside or by surgical vascular closure in the operating room. We did not have clear selection criteria for these procedures. The preferred method was at the discretion of the attending physician; therefore, the rate of arterial thrombosis could be underestimated in the percutaneous vascular closure group because of the lower sensitivity of vascular ultrasound in evaluating the femoral to iliac artery segment. Finally, the presence of these incidental small linear thrombi, detectable through surgical exploration or vascular ultrasound, may not have a substantial influence on patient outcomes or warrant specific interventions. Although limb ischemia can be devastating and cause disability, the criteria of thrombus that result in complications remains uncertain, warranting further investigation.
Our study showed that the incidence of CaAT after ECMO decannulation was 61.7%. Moreover, the ECMO flow rate–BSA ratio was an independent risk factor for CaAT. We suggest that screening for arterial thrombosis following VA-ECMO support is necessary, and that further research is needed to determine the risks and benefits of such screening.
KEY MESSAGES
▪ The incidence of cannula-related arterial thrombosis following extracorporeal membrane oxygenation (ECMO) decannulation remains high.
▪ Screening for arterial thrombosis after venoarterial ECMO support is deemed necessary, and further research is needed to determine the risks and benefits of such screening.
▪ The ECMO flow rate–body surface area ratio is an independent risk factor for cannula-associated arterial thrombosis.
NOTES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
FUNDING
None.
-
AUTHOR CONTRIBUTIONS
Conceptualization: NHKT, XTP, DQH, TMN, TAM, QQMD, TTNP. Data curation: NHKT, LTT, TMN, TAM, QQMD. Formal analysis: XTP, DQH, TMN, BXN, TTNP. Methodology: NHKT. DQH, QQMD. Project administration: DQH, TAM, TTNP. Visualization: LTT, QQMD, BXN, TTNP. Writing–original draft: NHKT, XTP, LTT, TMN, TAM, BXN. Writing–review & editing: NHKT, XTP, TAM, TTNP.
Acknowledgments
None.
Figure 1.Position of arterial thrombus formation in the low-flow space. Created with BioRender.com.

Figure 2.Flowchart of patient inclusion. ECMO: extracorporeal membrane oxygenation; PE: pulmonary embolism; VA: arteriovenous; CaAT: cannula-associated arterial thrombosis.
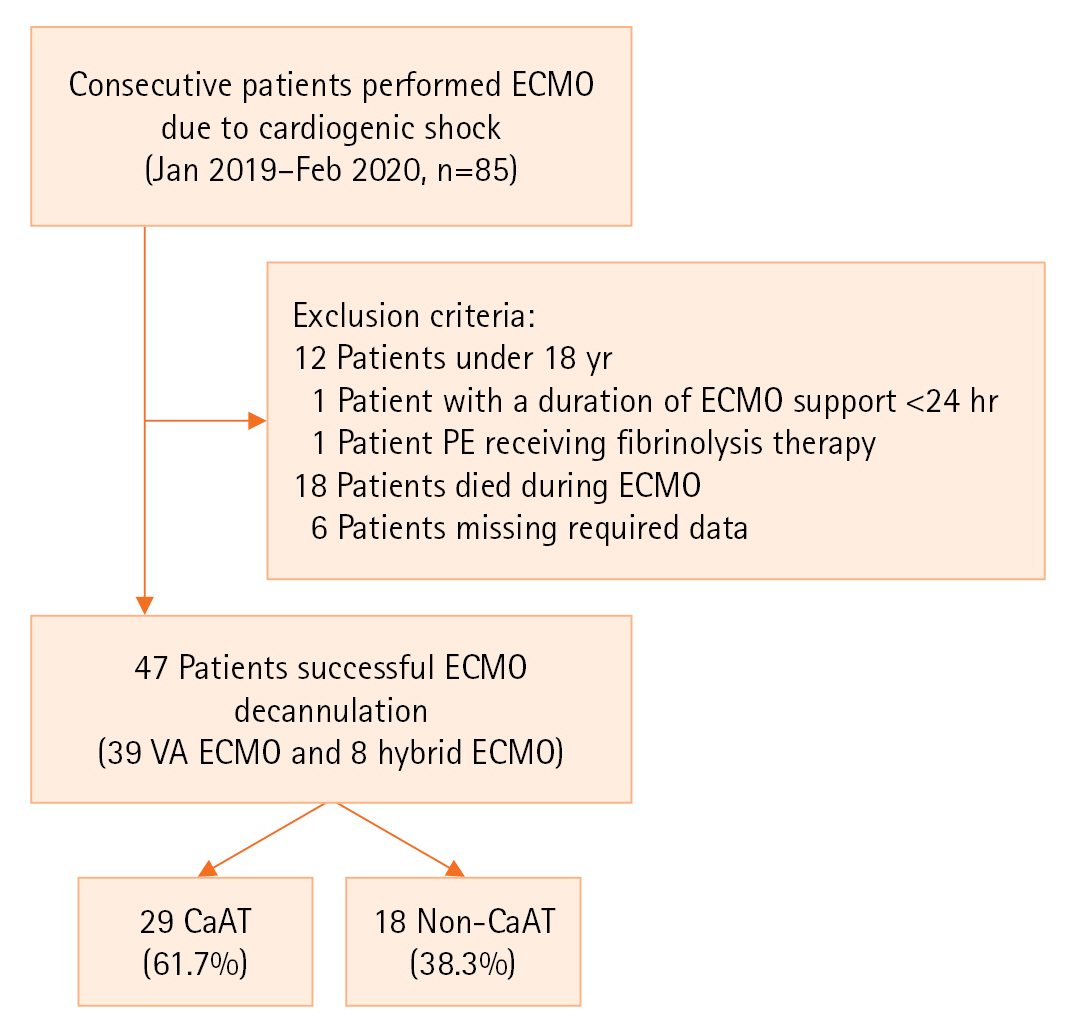
Figure 3.The distribution of cannula-associated arterial thrombosis.
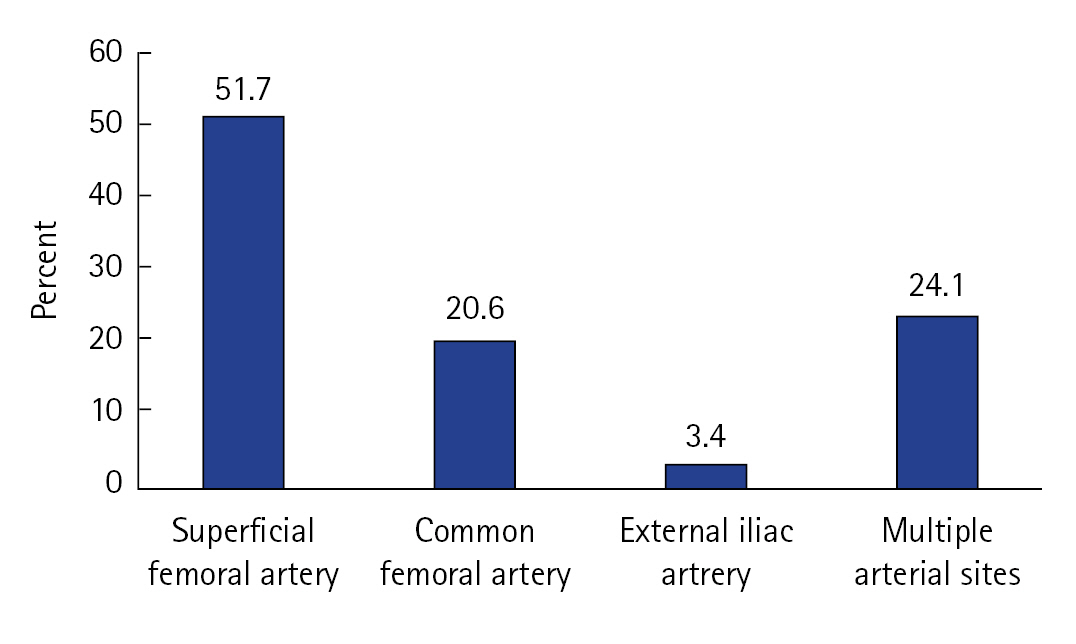
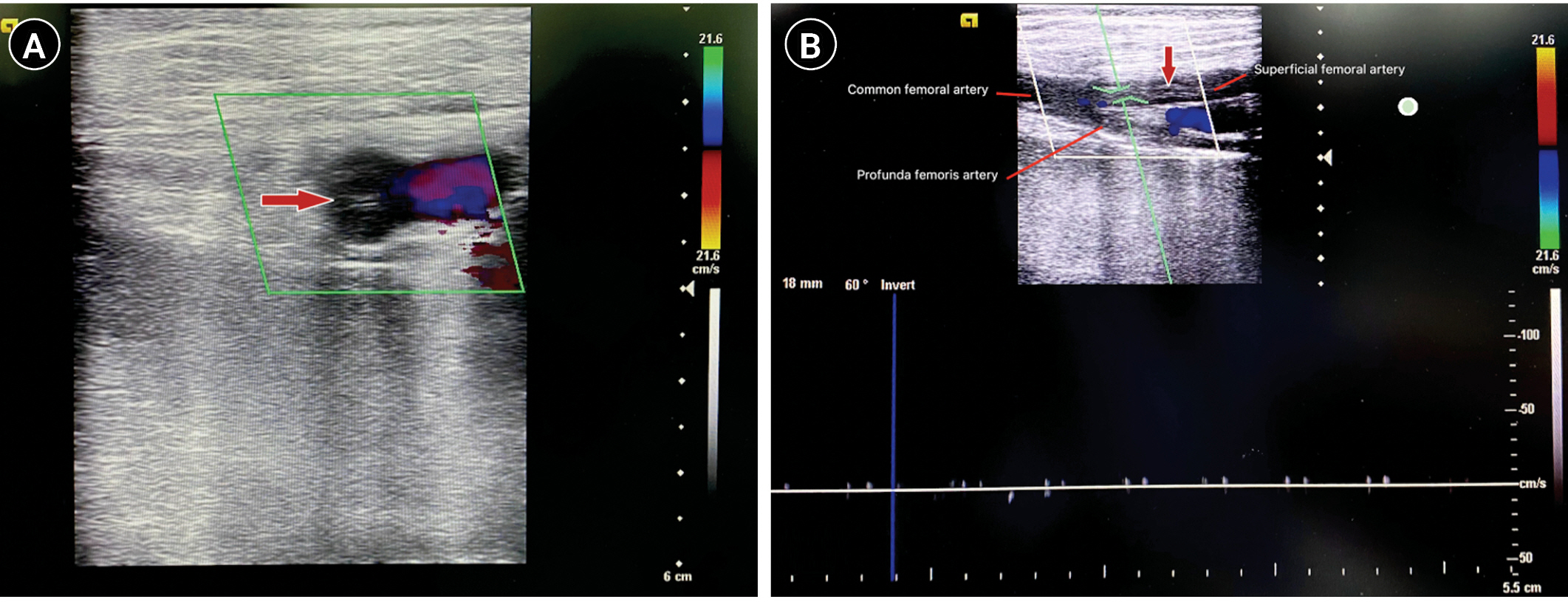
Figure 4.Detection of thrombus on bedside Doppler ultrasound on a 42-year-old male patient after supported extracorporeal membrane oxygenation due to acute myocarditis. (A) The transverse view depicts the color Doppler spectrum in the femoral vein. (B) The longitudinal view revealed nearly complete occlusion of the common femoral artery and superficial femoral artery and the absence of arterial flow by pulsed Doppler, indicating cannula-associated arterial thrombus (red arrow).
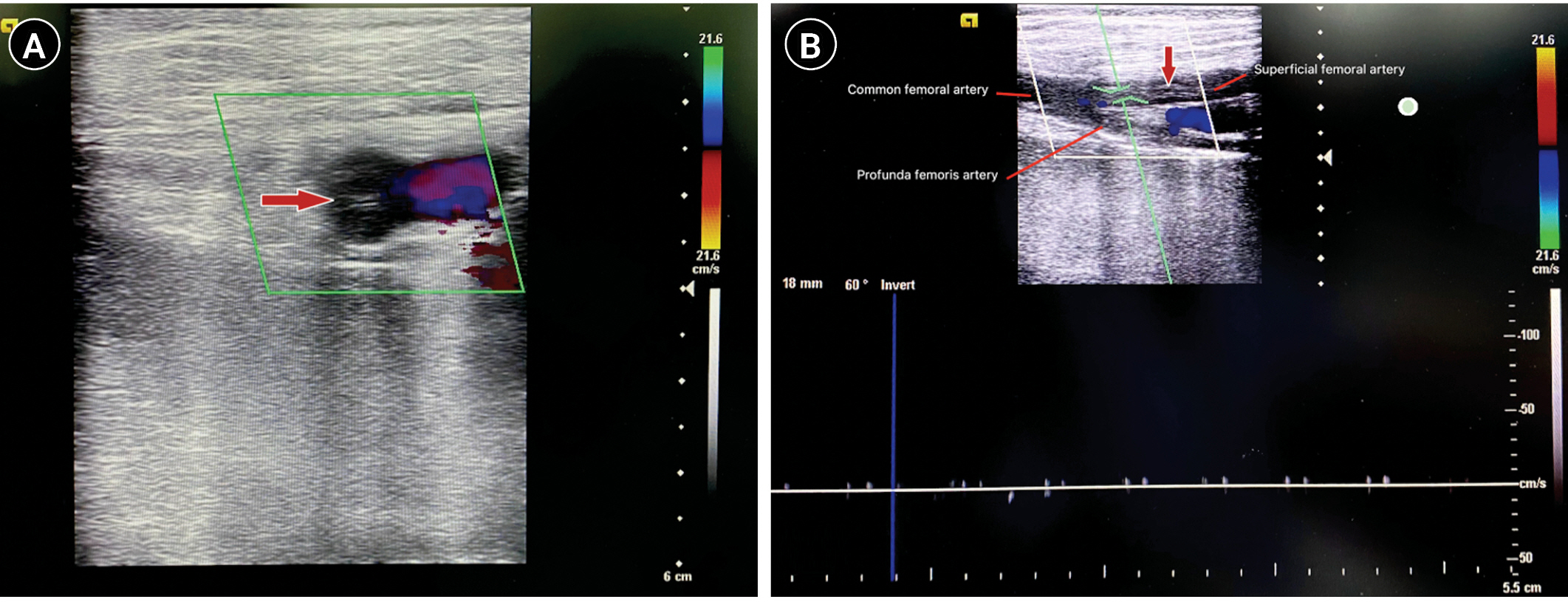
Table 1.Characteristics of the study population
|
Characteristics |
All (n=47) |
Cannula-associated arterial thrombosis
|
P-value |
|
Yes (n=29) |
No (n=18) |
|
Age (yr) |
33 (26–46) |
33 (26–39) |
38 (28–52) |
0.185 |
|
Male |
16 (34) |
7 (24.1) |
9 (50) |
0.068 |
|
BMI (kg/m2) |
21.3 (19.5–22.7) |
20.8 (19.5–22.2) |
22.0 (20.8–23.4) |
0.100 |
|
BSA (m2) |
1.54 (1.46–1.66) |
1.49 (1.41–1.66) |
1.63 (1.51–1.68) |
0.098 |
|
Comorbidity |
|
|
|
|
|
Hypertension |
5 (10.6) |
3 (10.3) |
2 (11.1) |
0.644 |
|
Diabetes |
3 (6.4) |
2 (6.9) |
1 (5.6) |
0.680 |
|
Peripheral vascular disease |
1 (2.1) |
0 |
1 (5.6) |
0.383 |
|
≥2 Comorbidities |
3 (6.4) |
2 (6.9) |
1 (5.6) |
0.680 |
|
Severity scores on ICU admission |
|
|
|
|
|
SOFA score |
11 (8–13) |
10.0 (7.0–13.0) |
11.0 (9.0–13.8) |
0.397 |
|
APACHE II score |
18 (12.5–25) |
18.0 (11.0–21.0) |
17.5 (13.8–25.2) |
0.661 |
|
Reason for ECMO |
|
|
|
|
|
Myocarditis |
32 (68.1) |
21 (72.4) |
11 (61.1) |
0.419 |
|
Myocardial infarction |
3 (6.4) |
2 (6.9) |
1 (5.6) |
0.680 |
|
Other |
12 (25.5) |
6 (20.7) |
6 (33.3) |
0.265 |
|
Blood products transfusion during ECMO |
|
|
|
|
|
Red blood cells (unit) |
2.0 (2.0–6.0) |
3.0 (2.0–6.0) |
4.5 (4.0–5.75) |
0.285 |
|
Fresh frozen plasma (unit) |
1.0 (0–5.5) |
2.0 (0–6.0) |
2.5 (0–4.0) |
0.660 |
|
Cryoprecipitate (unit) |
0 (0–0) |
0 (0–0) |
0 (0–0) |
0.621 |
|
Platelets (unit) |
0 (0–0) |
1 (0–3.25) |
2 (0.25–4.5) |
0.565 |
|
Cannula–BSA ratio |
10.1 (9.47–10.70) |
10.1 (9.72–10.78) |
10.1 (9.19–10.53) |
0.418 |
|
Arterial cannula diameter |
|
|
|
0.429 |
|
15 Fr |
27 (57.4) |
19 (65.5) |
8 (44.4) |
|
|
17 Fr |
16 (34.1) |
8 (27.6) |
8 (44.4) |
|
|
19 Fr |
4 (8.5) |
2 (6.9) |
2 (11.1) |
|
|
Distal perfusion cannula diameter |
|
|
|
0.203 |
|
5 Fr |
12 (26.1) |
9 (31.1) |
3 (17.6) |
|
|
6 Fr |
31 (67.4) |
17 (58.6) |
15 (83.3) |
|
|
7 Fr |
3 (6.5) |
3 (10.3) |
0 |
|
|
Cannulation technique |
|
|
|
0.275 |
|
Surgery |
3 (6.4) |
3 (10.3) |
0 |
|
|
Seldinger |
44 (93.6) |
26 (89.7) |
18 (100) |
|
|
Infection before the start of ECMO |
11 (51.1) |
6 (51.7) |
5 (50.0) |
0.456 |
|
Pulmonary |
7 (63.6) |
5 (83.3) |
2 (40) |
|
|
Other |
4 (36.3) |
1 (16.6) |
3 (60) |
|
|
Infection during ECMO |
22 (36.2) |
11 (34.5) |
12 (38.9) |
0.270 |
|
Pulmonary |
18 (81.1) |
11 (100) |
7 (58.3) |
|
|
Other |
5 (22.7) |
0 |
5 (41.7) |
|
|
Hybrid |
8 (17.0) |
5 (17.2) |
3 (16.7) |
0.642 |
|
IABP |
7 (14.9) |
5 (17.2) |
2 (11.1) |
0.449 |
|
Pacing |
18 (38.3) |
14 (48.3) |
4 (22.2) |
0.070 |
|
ECPR |
7 (14.9) |
4 (13.8) |
3 (16.7) |
0.550 |
|
Rotations of ECMO (rotations/min) |
2,800 (2,701–2,895) |
2,800 (2,720–2,865) |
2,793 (2,632–2,999) |
0.895 |
|
Flow rate of ECMO–BSA ratio (100 ml/min/m2) |
17.8 (14.9–21.5) |
16.8 (14.9–18.6) |
21.2 (16.0–24.7) |
0.044 |
|
Mean dose UFH (100 IU/hr) |
6.09 (4.01–7.15) |
6.26 (4.03–7.73) |
5.96 (3.91–6.90) |
0.662 |
|
Decannulation technique |
|
|
|
0.217 |
|
Surgical vascular closure |
21 (44.7) |
15 (51.7) |
6 (33.3) |
|
|
Percutaneous vascular closure |
26 (55.3) |
14 (48.2) |
12 (66.7) |
|
Table 2.Outcomes of patients in two groups
|
Variable |
All (n=47) |
Cannula-associated arterial thrombosis
|
P-value |
|
Yes (n=29) |
No (n=18) |
|
Limb ischemia complication |
5 (10.6) |
5 (17.2) |
0 |
0.141 |
|
Limb ischemia required surgical thrombectomy |
1 (20) |
1 (20) |
0 |
1.000 |
|
Anticoagulation therapy |
4 (80) |
4 (80) |
0 |
1.000 |
|
Infection at removal site |
3 (6.4) |
2 (6.9) |
1 (5.6) |
1.000 |
|
Pseudoaneurysm |
0 |
0 |
0 |
1.000 |
|
Limb amputations |
0 |
0 |
0 |
1.000 |
|
ECMO support (day) |
6 (4–7.5) |
6 (4–8) |
6 (4–7) |
0.991 |
|
ICU length of stay (day) |
9.0 (8–13) |
9.0 (7.0–12.0) |
9.5 (8.0–21.2) |
0.582 |
|
Hospital length of stay (day) |
16.0 (12.0–24.5) |
16.0 (12.0–22.0) |
15.5 (12.0–29.5) |
0.709 |
|
Hospital mortality |
5 (10.6) |
1 (3.4) |
4 (22.2) |
0.063 |
Table 3.Results of the blood coagulation parameters
|
Parameter |
All (n=47) |
Cannula-associated arterial thrombosis
|
P-value |
|
Yes (n=29) |
No (n=18) |
|
aPTT ratio |
|
|
|
|
|
Median aPTTr |
1.49 (1.36–1.73) |
1.59 (1.35–1.80) |
1.44 (1.38–1.68) |
0.393 |
|
Day with aPTTr <1.2 (%) |
0.17 (0–0.25) |
0.20 (0–0.25) |
0.13 (0–0.31) |
0.614 |
|
Day with aPTTr ≥1.2 and <1.5 (%) |
0.28 (0.13–0.50) |
0.25 (0.12–0.33) |
0.33 (0.25–0.50) |
0.142 |
|
Day with aPTTr ≥1.5 and <2 (%) |
0.33 (0.14–0.42) |
0.33 (0.12–0.38) |
0.29 (0.20–0.46) |
0.667 |
|
Day with aPTTr ≥2 (%) |
0.2 (0–0.33) |
0.25 (0–0.38) |
0.17 (0.03–0.20) |
0.194 |
|
Anti-Xa activity |
|
|
|
|
|
Day with anti-Xa activity <0.2 UI/ml (%) |
0 (0–0.13) |
0 (0–0.12) |
0.10 (0–0.16) |
0.224 |
|
Day with anti-Xa activity ≥0.2 và <0.5 UI/ml (%) |
0.10 (0–0.29) |
0.10 (0–0.25) |
0.13 (0–0.36) |
0.654 |
|
Day with anti-Xa activity ≥0.5 UI/ml (%) |
0 (0–0) |
0 (0–0) |
0 (0–0) |
0.403 |
|
Fibrinogen (g/L) |
|
|
|
|
|
Lowest fibrinogen level |
2.47 (1.74–3.1) |
2.02 (1.44–2.60) |
2.96 (2.44–3.35) |
0.006 |
|
Highest fibrinogen level |
4.52 (3.88–6.01) |
4.01 (3.60–4.99) |
5.64 (4.52–6.47) |
0.011 |
|
Day with fibrinogen <2 g/L (%) |
0 (0–0.13) |
0 (0–0.25) |
0 (0–0.1) |
0.018 |
|
Day with fibrinogen ≥2 and <4 g/L (%) |
0.63 (0.37–0.86) |
0.75 (0.50–0.88) |
0.50 (0.27–0.67) |
0.051 |
|
Day with fibrinogen ≥4 g/L (%) |
0.17 (0–0.58) |
0.11 (0–0.25) |
0.50 (0.33–0.67) |
0.005 |
|
Platelets (G/L) |
|
|
|
|
|
Lowest platelet count |
82 (56.5–110) |
85 (60–132) |
80 (52–93) |
0.158 |
|
Highest platelet count |
189 (159.5–258) |
226 (172–258) |
164 (131–231) |
0.048 |
|
Day with platelets <150 (%) |
0.8 (0.58–0.88) |
0.75 (0.33–0.87) |
0.83 (0.71–1.00) |
0.039 |
|
Day with platelets ≥150 và <400 (%) |
0.2 (0.12–0.42) |
0.25 (0.12–0.67) |
0.17 (0.10–0.29) |
0.036 |
|
Day with platelets ≥400 (%) |
0 (0–0) |
0 (0–0) |
0 (0–0) |
0.707 |
Table 4.Multivariable analysis of factors associated with CaAT
|
Variable |
aOR (95% CI) |
P-value |
|
Cannula–BSA ratio (Fr/m2) |
1.31 (0.62–2.79) |
0.476 |
|
Flow rate of ECMO–BSA ratio (100 ml/min/m2) |
0.79 (0.66–0.95) |
0.014 |
|
Highest fibrinogen level (every g/L) |
0.75 (0.41–1.37) |
0.351 |
|
Days with fibrinogen ≥4 g/L (%) |
0.24 (0.01–6.21) |
0.393 |
References
- 1. Pineton de Chambrun M, Bréchot N, Combes A. Venoarterial extracorporeal membrane oxygenation in cardiogenic shock: indications, mode of operation, and current evidence. Curr Opin Crit Care 2019;25:397-402.ArticlePubMed
- 2. Doyle AJ, Hunt BJ. Current understanding of how extracorporeal membrane oxygenators activate haemostasis and other blood components. Front Med (Lausanne) 2018;5:352. ArticlePubMedPMC
- 3. Esper SA, Levy JH, Waters JH, Welsby IJ. Extracorporeal membrane oxygenation in the adult: a review of anticoagulation monitoring and transfusion. Anesth Analg 2014;118:731-43.PubMed
- 4. Bonicolini E, Martucci G, Simons J, Raffa GM, Spina C, Lo Coco V, et al. Limb ischemia in peripheral veno-arterial extracorporeal membrane oxygenation: a narrative review of incidence, prevention, monitoring, and treatment. Crit Care 2019;23:266. ArticlePubMedPMCPDF
- 5. Bisdas T, Beutel G, Warnecke G, Hoeper MM, Kuehn C, Haverich A, et al. Vascular complications in patients undergoing femoral cannulation for extracorporeal membrane oxygenation support. Ann Thorac Surg 2011;92:626-31.ArticlePubMed
- 6. Kim J, Cho YH, Sung K, Park TK, Lee GY, Lee JM, et al. Impact of cannula size on clinical outcomes in peripheral venoarterial extracorporeal membrane oxygenation. ASAIO J 2019;65:573-9.ArticlePubMed
- 7. Tanaka D, Hirose H, Cavarocchi N, Entwistle JW. The impact of vascular complications on survival of patients on venoarterial extracorporeal membrane oxygenation. Ann Thorac Surg 2016;101:1729-34.ArticlePubMed
- 8. Yen CC, Kao CH, Tsai CS, Tsai SH. Identifying the risk factor and prevention of limb ischemia in extracorporeal membrane oxygenation with femoral artery cannulation. Heart Surg Forum 2018;21:E018-22.ArticlePubMedPDF
- 9. Lorusso R, Shekar K, MacLaren G, Schmidt M, Pellegrino V, Meyns B, et al. ELSO interim guidelines for venoarterial extracorporeal membrane oxygenation in adult cardiac patients. ASAIO J 2021;67:827-44.ArticlePubMed
- 10. Sulimov DS, Markuske M, Desch S, Thiele H. Management of dead space thrombosis during decannulation of peripherally inserted venoarterial extracorporeal membrane oxygenation. Catheter Cardiovasc Interv 2021;98:E122-3.ArticlePubMedPDF
- 11. Cooper E, Burns J, Retter A, Salt G, Camporota L, Meadows CI, et al. Prevalence of venous thrombosis following venovenous extracorporeal membrane oxygenation in patients with severe respiratory failure. Crit Care Med 2015;43:e581-4.ArticlePubMed
- 12. Fisser C, Reichenbächer C, Müller T, Schneckenpointner R, Malfertheiner MV, Philipp A, et al. Incidence and risk factors for cannula-related venous thrombosis after venovenous extracorporeal membrane oxygenation in adult patients with acute respiratory failure. Crit Care Med 2019;47:e332-9.ArticlePubMed
- 13. Menaker J, Tabatabai A, Rector R, Dolly K, Kufera J, Lee E, et al. Incidence of cannula-associated deep vein thrombosis after veno-venous extracorporeal membrane oxygenation. ASAIO J 2017;63:588-91.ArticlePubMed
- 14. Parzy G, Daviet F, Persico N, Rambaud R, Scemama U, Adda M, et al. Prevalence and risk factors for thrombotic complications following venovenous extracorporeal membrane oxygenation: a CT scan study. Crit Care Med 2020;48:192-9.ArticlePubMed
- 15. Bidar F, Lancelot A, Lebreton G, Pineton de Chambrun M, Schmidt M, Hékimian G, et al. Venous or arterial thromboses after venoarterial extracorporeal membrane oxygenation support: frequency and risk factors. J Heart Lung Transplant 2021;40:307-15.ArticlePubMed
- 16. Cartwright B, Bruce HM, Kershaw G, Cai N, Othman J, Gattas D, et al. Hemostasis, coagulation and thrombin in venoarterial and venovenous extracorporeal membrane oxygenation: the HECTIC study. Sci Rep 2021;11:7975. ArticlePubMedPMCPDF
- 17. Kohs TC, Liu P, Raghunathan V, Amirsoltani R, Oakes M, McCarty OJ, et al. Severe thrombocytopenia in adults undergoing extracorporeal membrane oxygenation is predictive of thrombosis. Platelets 2022;33:570-6.ArticlePubMed
- 18. Lamarche Y, Chow B, Bédard A, Johal N, Kaan A, Humphries KH, et al. Thromboembolic events in patients on extracorporeal membrane oxygenation without anticoagulation. Innovations (Phila) 2010;5:424-9.ArticlePubMedPDF
- 19. Avenarius JK, Breek JC, Lampmann LE, van Berge Henegouwen DP, Hamming JF. The additional value of angiography after colour-coded duplex on decision making in patients with critical limb ischaemia: a prospective study. Eur J Vasc Endovasc Surg 2002;23:393-7.ArticlePubMed
- 20. Lai DT, Huber D, Glasson R, Grayndler V, Evans J, Hogg J, et al. Colour duplex ultrasonography versus angiography in the diagnosis of lower-extremity arterial disease. Cardiovasc Surg 1996;4:384-8.ArticlePubMed
- 21. Mennitt K, Deol M, Gao J. Emergency color Doppler sonography of the extremity artery: a pictorial essay. Clin Imaging 2017;42:240-8.ArticlePubMed
- 22. Patton-Rivera K, Beck J, Fung K, Chan C, Beck M, Takayama H, et al. Using near-infrared reflectance spectroscopy (NIRS) to assess distal-limb perfusion on venoarterial (V-A) extracorporeal membrane oxygenation (ECMO) patients with femoral cannulation. Perfusion 2018;33:618-23.ArticlePubMedPDF
- 23. Thiagarajan RR, Barbaro RP, Rycus PT, Mcmullan DM, Conrad SA, Fortenberry JD, et al. Extracorporeal Life Support Organization Registry International Report 2016. ASAIO J 2017;63:60-7.ArticlePubMed
- 24. Rastan AJ, Lachmann N, Walther T, Doll N, Gradistanac T, Gommert JF, et al. Autopsy findings in patients on postcardiotomy extracorporeal membrane oxygenation (ECMO). Int J Artif Organs 2006;29:1121-31.ArticlePubMedPDF
- 25. Sertic F, Chavez L, Diagne D, Richards T, Wald J, Acker M, et al. Predictors of in-hospital mortality and midterm outcomes of patients successfully weaned from venoarterial extracorporeal membrane oxygenation. J Thorac Cardiovasc Surg 2021;161:666-78.ArticlePubMed
- 26. Kohler K, Valchanov K, Nias G, Vuylsteke A. ECMO cannula review. Perfusion 2013;28:114-24.ArticlePubMedPDF
- 27. Fraser KH, Zhang T, Taskin ME, Griffith BP, Wu ZJ. Computational fluid dynamics analysis of thrombosis potential in left ventricular assist device drainage cannulae. ASAIO J 2010;56:157-63.ArticlePubMedPMC
Citations
Citations to this article as recorded by

- Has the role of veno-arterial extracorporeal membrane oxygenation in patients with cardiogenic shock following acute myocardial infarction been fully determined? A case report
Kha Minh Nguyen, Hai Phuong Nguyen Tran, Vi Tuong Dang, Sy Van Hoang, Josip Andelo Borovac, Dmitry Duplyakov, Chiara De Biase, Pok-Tin Tang
European Heart Journal - Case Reports.2024;[Epub] CrossRef
 , Xuan Thi Phan3
, Xuan Thi Phan3 , Linh Thanh Tran2
, Linh Thanh Tran2 , Huy Minh Pham2
, Huy Minh Pham2 , Dai Quang Huynh1,2
, Dai Quang Huynh1,2 , Tuan Manh Nguyen2
, Tuan Manh Nguyen2 , Anh Tuan Mai1,2
, Anh Tuan Mai1,2 , Quan Quoc Minh Du2
, Quan Quoc Minh Du2 , Bach Xuan Nguyen1,2
, Bach Xuan Nguyen1,2 , Thao Thi Ngoc Pham1,2
, Thao Thi Ngoc Pham1,2







 KSCCM
KSCCM


 PubReader
PubReader ePub Link
ePub Link Cite
Cite