Abstract
-
Background
- Acute kidney injury (AKI) commonly occurs in critically ill patients. Estimation of renal function and antibiotics dose adjustment in patients with AKI is a challenging issue.
-
Methods
- Urinary creatinine clearance was measured in a 6-hour urine collection from patients with acute kidney injuries. The correlations between different formulas including the modified Cockcroft-Gault, modification of diet in renal disease, chronic kidney disease-epidemiology collaboration, Jelliffe, kinetic-glomerular filtration rate (GFR), Brater, and Chiou formulas were considered. The pattern of the prescribed antimicrobial agents was also compared with the patterns in the available resources.
-
Results
- Ninety-five patients with acute kidney injuries were included in the research. The mean age of the participants was 63.11±17.58 years old. The most patients (77.89%) were in stage 1 of AKI according to the Acute Kidney Injury Network criteria, followed by stage 2 (14.73%) and stage 3 (7.36), respectively. None of the formulations had a high or very high correlation with the measured creatinine clearance. In stage 1, Chiou (r=0.26), and in stage 2 and 3, kinetic-GFR (r=0.76 and r=0.37) had the highest correlation coefficient. Antibiotic over- and under-dosing were frequently observed in the study.
-
Conclusions
- The results showed that none of the static methods can predict the measured creatinine clearance in the critically ill patients. The dynamic methods such as kinetic-GFR can be helpful for patients who do not receive diuretics and vasopressors. Further studies are needed to confirm our results.
-
Keywords: acute kidney injury; critical illness; measured glomerular filtration rate
INTRODUCTION
According to the definition and setting, acute kidney injury (AKI) can occur in 1%–25% of intensive care units (ICUs) admitted patients with a mortality risk of 15%–60% [1]. A systematic review and meta-analysis showed an AKI incidences rate of 21.6% (range, 19.3–24.1) and 33.7% (range, 26.9–41.3) in adults and children, respectively [2]. Different criteria including RIFLE (Risk, Injury, Failure, or Loss of renal function, and End-stage kidney disease) and Acute Kidney Injury Network (AKIN) have been proposed for assessment of AKI [3]. Patients in later stages can have a higher mortality risk: 37.8%, 48.8%, and 76.5% for Risk, Injury, and Failure stages, respectively [4]. Patients admitted to surgical ICU may have a higher risk of AKI, and those with cardiovascular and neurological diseases or respiratory infections are at a greater risk of AKI [1].
Pharmacokinetics changes including alterations in the volume of absorption, distribution, protein binding, cytochromes activity, and elimination in patients with AKI are important factors in treating these patients [5,6]. In addition, hypervolemia, hypoalbuminemia, and augmented renal clearance affect the efficacy and toxicity of antibiotics in patients with AKI [7]. There is no standard formulation for estimating the glomerular filtration rate (GFR) or creatinine clearance (CrCl) [8]. Historically, dosing of medications in AKI people such as patients with GFR <10 ml/min had been recommended; however, this approach was led to underdosing of antimicrobial agents and therefore no longer is recommended [8]. Current guidelines recommend initial higher loading doses (e.g., doses ≥100%–150%) and normal or near-normal maintenance doses [8].
Continuous infusion of inulin, and measurement of inulin clearance with multiple urine samples is the standard method for measuring the GFR; however, it is not practical in ICU settings [9]. Iothalamate, iohexol, Cr52-EDTA (ethylenediaminetetraacetic acid), and cystatin C methods are alternative proposed approaches being used in research studies [9,10]. The urine CrCl is another method widely used in different ICU settings, although it is not standard and has its limitations [11-13]. Different formulas for estimation of GFR or CrCl including Cockcroft-Gault (CG), and chronic kidney disease-epidemiology collaboration (CKD-EPI), and modification of diet in renal disease (MDRD) are used for medication dosing in patients with stable renal function [8]. None of the above-mentioned formulas are precisely estimate GFR in patients with AKI [8]. Other formulations including Chiou and kinetic-GFR, Bratter, and Jelliffe are also recommended for patients with dynamic renal functions [14,15]. However, available guidelines recommend using collected urine (e.g., 1–12 hours) to measure CrCl and beta lactam antimicrobial optimal dosing [8,16]. Therefore, the aim of this study was to compare the correlation between the measured CrCl and static and dynamic formulas in the ICU patients with developed AKI.
MATERIALS AND METHODS
The research protocol was approved by the Ethical Committee of Kermanshah University of Medical Sciences (No. IR.KUMS.REC.1399.142). All participants or their family members (in case of unconsciousness, mental disease, or disability) signed the consent form for the study.
Patients
This study was a prospective study conducted on the critically ill patients. The adult patients with AKI were included in this study if they fulfilled the inclusion criteria. Daily serum creatinine and urine outputs before and after the development of AKI were recorded for all the included patients. The AKIN criteria were used for the detection and staging of AKI [17]. As 24 hours urine collection is impractical in critically ill patients, it also may over- or underestimate GFR. Therefore, shorter periods (e.g., 1-, 2-, 4-, 6-, and 8-hour urine collection) have been proposed for quick estimation of CrCl and minimizing procedure error [8,16,18,19]. Six hours urine collection had a good correlation with clearance of medications like vancomycin [18]. Therefore, 6-hour urines were collected and urinary CrCl were measured according to the following formula:
Urinary CrCl (ml/min)=urine creatinine (mg/dl)×urine volume (ml)/creatinine (mg/dl)×time (min).
The patients with a history of CKD (GFR <60 ml/min), those who received trimethoprim-sulfamethoxazole and cimetidine, patients who had urine outputs <0.5 ml/kg/hr or total positive fluid balance >500 ml, and those needed renal replacement therapy were excluded from this study. In order to eliminate the possible effects of vasopressors, diuretics, and comorbidities like diabetes, hypertension, and underlying heart disease on the correlations of different methods, another analysis was performed in patients without these risk factors [20].
The GFR or CrCl were calculated according to different formulas including CG, modified CG, MDRD, CKD-EPI, Jelliffe, Brater, Chiou, and kinetic-GFR. Dynamic formulas for GFR calculation are as followed.
Brater equation:
CrCl in female=male value×0.86
Chiou equation:
Creatine volume of distribution=0.6 L/kg
CrCl means creatinine clearance; sCr 1, 2: first and second serum creatinine values, Vd: creatinine volume of distribution (0.6 L/kg); and IBW: ideal body weight.
Kinetic-GFR:
Non-renal CrCl=0.048 ml∙min–1∙kg–1
Delta plasma creatinine refers to the change in plasma creatinine. It is defined as the ending creatinine minus the starting creatinine. Δ time (hr) is the interval in hours between two consecutive creatinines. MaxΔ plasma creatinine/day refers to the maximal change (increase) in the plasma creatinine that can occur per day if renal function is completely lost.
Furthermore, dosing of prescribed antibiotics was compared with dosing in the available resources including online Lexicomp, Sanford guide and nomograms, and under- or over-dosing were determined [21]. If a patient had normal serum creatinine and measured CrCl >130 ml/min, he/she would be considered as having augmented renal clearance [22].
The main outcome of the present study was the comparison between the measured 6-hour CrCl and formulas including CG, modified CG, modified CG, MDRD, CKD-EPI, Jelliffe, Brater, Chiou, and kinetic-GFR. The secondary outcome was evaluation of antimicrobial prescription dosing pattern in the patients with AKI.
Analysis
The data was analyzed using SPSS ver.16 (SPSS Inc., Chicago, IL, USA). The categorical variables were expressed as numbers and percentages, and the chi-square test and Fisher’s exact test were used for comparing the variables between the two groups. The continuous variables were reported as mean±standard deviation. The Kolmogorov-Smirnov test was used for determining the normally distributed variables. The normally distributed continuous variables were compared by the use of the student t-test, and the non-normally distributed variables were compared using the Mann-Whitney rank-sum test. The correlation between the measured and calculated GFR was obtained by the use of Spearman's correlation coefficient test. The P-values less than 0.05 were considered to be statistically significant.
RESULTS
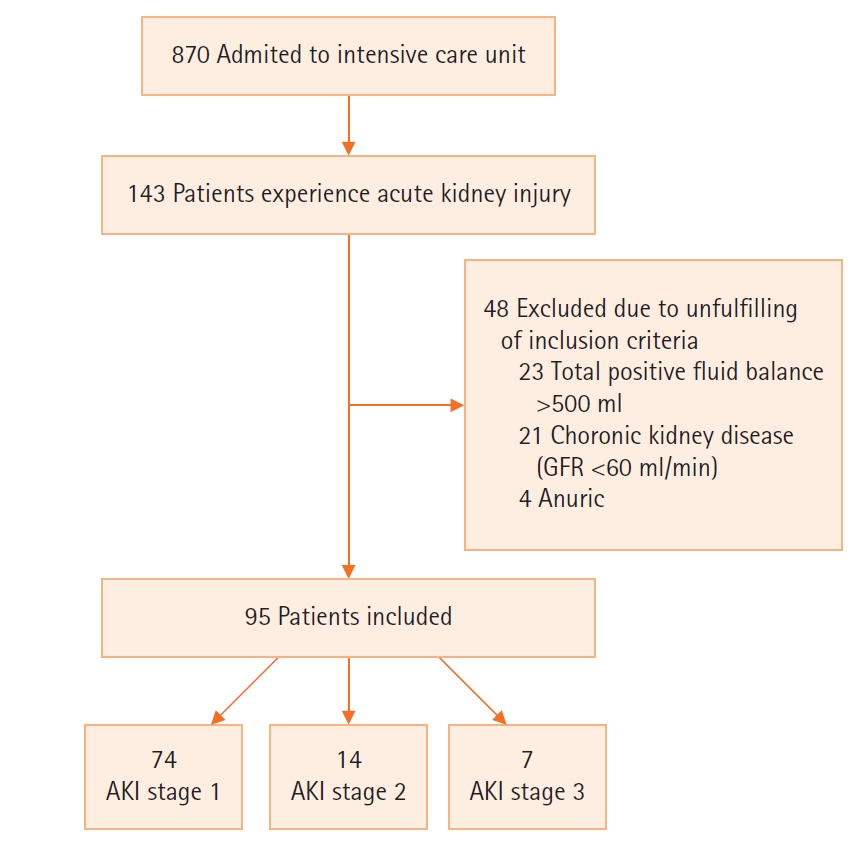
During the study period, 95 patients with AKI were recorded according to the AKIN criteria (Figure 1). Most of the patients (66.3%) were male. The mean age of the participants was 63.11±17.58 years. The mean serum creatinine at baseline and after AKI were 1.06±0.30 mg/dl and 2.04±0.84, respectively (Table 1). The mean Sequential Organ Failure Assessment (SOFA) and Acute Physiology and Chronic Health Evaluation (APACHE) II scores of all the participants were 8.09±3.52 and 20.09±7.52, respectively (Table 1). The mean measured CrCl in all the participant was 45.28±12.51 ml/min. The most common reasons for admission include the accidents of neurological problems (49.45%) and solid cancers 10.52%. Only 20% of the patients did not have any risk factor for renal failures (Table 2).
Table 3 shows the correlations between different formulas and the measured CrCl. None of the formulations had a high or very high correlation with the measured CrCl (Table 3). Most of the patients (77.89%) were in the stage 1 of AKI according to the AKIN criteria, followed by stage 2 (14.73%) and stage 3 (7.36%). In addition, three patients (all were in stage 1 on AKI) had criteria for augmented renal clearance. As shown in the Table 4, a significant moderate correlation was observed between the GFR calculated by the Brater formula and the measured CrCl (r=0.40, P<0.001). In the patients being in stage 2 of AKI, only Kinetic-GFR had a high correlation with the measured CrCl (r=0.76, P=0.004) (Table 4). Chiou and Jelliffe (r=0.26), kinetic-GFR (r=0.76), and Jelliffe and Kinet-GFR (r=37) had the highest correlation coefficients in stages 1, 2, and 3 of AKI, respectively.
When the patients who received loop diuretics (n=24) were excluded from the analysis, a significant moderate correlation was obtained for Jelliffe (r=0.46, P<0.001) and kinetic-GFR (r=0.46, P=0.001) formulations. In addition, when the patients who received vasoactive agents (n=12) were excluded from the analysis, a significant moderate correlation was observed for Jelliffe (r=0.45, P=0.001) and kinetic-GFR (r=0.45, P<0.001). Once the patients who received both diuretics and vasoactive agents were excluded (n=39), a high correlation was found for kinetic-GFR (r=0.60, P=0.001). Furthermore, all the formulations had significant correlation coefficients of more than 0.7 when only the patients with no risk factors for acute decline in CrCl.
Vancomycin, meropenem, and piperacillin-tazobactam were the most commonly prescribed antibiotics (Table 5). As shown in Table 5, over- and under-dosing of antibiotics were common in our study. Vancomycin underdosing 19.23% (10/52) or overdosing 32.69% (17/52) and meropenem overdosing 39.53% (17/43) were commonly observed as well. Vasopressors and Loop diuretics were prescribed for 18.94% (18/95) and 7.36% (7/95) of the patients, respectively. Low doses (200–300 mg/day) of hydrocortisone for septic shock were prescribed for 13.75% of the patients. In addition, venous thromboembolism prophylaxis without adjustment with enoxaparin was prescribed for 27.3% of the patients. Metoclopramide as prokinetic was needed for 25% of the patients.
DISCUSSION
The results of this study showed that none of the available formulas had a high or very high correlation with the measured CrCl in all the stages of AKI. In the critically ill patients with stable renal function CG and ideal body weight, a high correlation with inulin clearance (r=0.75, P<0.001) was found in a study by Roberts et al [23]. However, in the patients with AKI and unstable serum creatinine, measuring GFR was challenging. Evaluating the correlations between urine CrCl and CG, MDRD, and Jelliffe, Bouchard et al. [24] found significant correlations: CG (r=0.67), MDRD (r=0.89), Jelliffe (r=0.75), and modified Jelliffe (r=0.72). Furthermore, the modified CG overestimated and the modified Jelliffe underestimated GFR compared with the measured urine clearance [24].
In the present study, none of the tested formulas had a high or very high correlation with the measured urine CrCl. Jelliffe and kinetic-GFR designed for the patients with unstable renal (fluctuation in serum creatinine) functions [14,25]. Recently, Pelletier et al. [26] have used the measured GFR according to radioisotopic 99mTc-DTPA (diethylenetriaminepentaacetic acid) as a standard tool and compared it with Jelliffe, kinetic-GFR, and the measured urinary CrCl in 119 patients with AKI. They showed that there was good correlations between Jelliffe and kable to estimate a 24-hour CrClinetic-GFR equations and measured GFR (0.73 and 0.68, respectively). However, the adjusted analysis showed correlations of 0.63 and 0.47 for Jelliffe and kinetic-GFR, respectively, with reference GFR [26].
In our study, almost the same correlations were obtained for Jelliffe and kinetic-GFR. However, the correlations were lower than those observed by Al-Dorzi et al. [11]. They evaluated the correlations between different formulas and a measured 24-hour CrCl in critically ill patients with different renal functions. Although the results showed a high correlation between the measured CrCl and the formulas, a significant bias was observed in the sensitivity analysis. Therefore, they concluded that none of the commonly used formulas are able to estimate a 24-hour CrCl [11].
Carlier et al. [27] showed that the measured 1–2 hours ClCr had a better correlation with 24-hour urinary inulin clearance compared with the collected 24-hour urine ClCr. Using iothalamate and inulin clearances is not practical for all the AKI episodes. Unfortunately, urine CrCl may overestimate GFR by 10%–20% in normal renal function, which can increase as renal function decreases [28]. In a subset of the patients who did not have any risk factors for renal failure, all the formulations used had a good correlation with all the formula such as kinet-GFR and Jelliffe. It should be noted that most of the patients were in stage 1 of AKI and the guidelines recommended to consider changes in drug dosing in stage 2 of AKI [29].
All formulations, including the dynamic formulas, assume a constant rate of creatinine production and volume of distribution and may underestimate AKI severity [30]. Fluid overload may lower serum creatinine concentration and underestimate CrCl [31]. Corrected Jelliffe method has been recommended for the patients with aggressive fluid administration to eliminate the effects of positive fluid balance on serum creatinine concentration [32]. However, the patients with positive total fluid balance >500 ml were not included in the present study. Similar to our results, a recent study by Sangla et al. [33] on critically ill patients showed that the kinetic-GFR estimation model show a better performance than CG and MDRD do in the patients with unstable renal dysfunctions. In addition, they observed that all the methods including CG, MDRD, CKD-EPI, kinetic-GFR, the measured GFR overestimated GFR [33]. A bias of 21 ml/min/1.73 and an accuracy of 35% were observed for urinary GFR method [33]. Considering these limitations for statistical formulas on the kidney disease, improving the Global Outcomes guidelines about dosing in acute and CKD are recommend for dosing in patients with AKI using the measured ClCr [8]. In addition, the measured CrCl is recommended for detecting the augmented renal clearance [34].
In the present study, a higher correlation was found for kinetic-GFR in the patients with the stage of 2 of AKI. In addition, after exclusion of patients who received diuretics and vasopressors a correlation of 0.6 was found. Previous studies showed a different effect of diuretics and vasopressors on GFR [35]. Although diuretics increase urine output and renal perfusion but they didn’t associate with an increased GFR [36]. Norepinephrine and terlipressin administration were associated with increase urine output and GFR in patients with septic shock [36]. However, other factors including the rate of creatinine generation, filtration, fluid administration, and time from AKI occurrence may influence the correlations [37]. After exclusion of patients with mentioned risk factors, our results showed all formulas had a high correlation between measured CrCl and calculated values. Similar results were obtained from a study by Bouchard et al. [24]. They showed that Jelliffe and modified Jelliffe are more precise than other formulas, baseline serum creatinine in our study was lower (2.04±0.82 vs. 3.8±1.6) [24]. However, Hoste et al. [38] found CG and MDRD were not correlated with 1-hour measured CrCl.
Over- or under-dosing of antibiotics including vancomycin, piperacillin-tazobactam, and meropenem were common in the present study. Some experts recommended postponing antibiotics dose reduction in the early stages of AKI (i.e., 24–48 hours) for antibiotics with wide safety margins like beta-lactams due to low toxicity [30]. However, postponing dose adjustment for vancomycin and other nephrotoxic agents is not recommended [30]. One reason for overdosing of meropenem was the presence of multi-drug resistant organism in our ICU. The current guidelines for dosing of beta-lactams in multidrug resistant organisms recommend Cmax/MIC of 4–8 for multidrug resistant Gram-negative organisms [16]. However, in the present study, dosing of the prescribed antibiotics was compared with nomograms which can influence our conclusions [18]. Recently, Kwong et al. [39] showed that kinetic-GFR can be a better formula for antibiotics dosing and prevention of their toxicity or underdosing. Furthermore, kinetic-GFR can be a better formula for prediction of AKI and mortality [40,41]. In our study, three patients fulfilled the criteria for the augmented renal clearance, as this phenomenon was occurring in up to 30% of the critically ill patients [34]. Therefore, measuring of urinary CrCl is a helpful way to prevent underdosing of antibiotics in this population.
In our study, patients GFR≥60 ml/min were prospectively evaluated for the occurrence of acute kidney injuries. In the present study, patients GFR ≥60 ml/min were prospectively evaluated for the occurrence of acute kidney injuries. Correlation between the measured CrCl and estimated GFR in patients without risk factors for developing AKI, and those who had not received vasopressors and diuretics, were analyzed separately and dosing of antibiotics were compared with standard mammograms. However, there some limitations, which can influence our conclusions. First, we were not able to measure GFR with standard methods including inulin, iohexol or iothalamate clearance. Second, antimicrobial therapeutic monitoring for beta-lactams was not available in our center and we were not able to compare dosing according to different formulas.
The results of the present study showed that none of the available formula had a high or very high correlation with the 6-hour measured CrCl. However, kinetic-GFR and Jelliffe, although not perfect, are better formulas for estimating CrCl in patients with AKI. Further studies are needed to use the standard methods in order to clarify the role of different static and dynamic formulas for medication dose adjustment.
KEY MESSAGE
▪ In patients with acute kidney injury (AKI) dynamic methods like kinet-glomerular filtration rate (GFR) may be a better prediction for GFR.
▪ Using Cockcroft-Gault in patients with AKI overestimate GFR.
NOTES
-
CONFLICT OF INTEREST No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conceptualization: FH, HK, FS. Data curation: SK, FH, FS. Formal analysis: SK, FH, FS. Methodology: all authors. Project administration: HK, FS. Visualization: FH, HK, FS. Writing–original draft: all authors. Writing–review & editing: all authors.
Figure 1.Flowchart of included patients. GFR: glomerular filtration rate; AKI: acute kidney injury.
Table 1.Baseline characteristics of the included patients
|
Variable |
Value |
|
Age (yr) |
63.11±17.58 |
|
Male |
63/95 (66.3) |
|
Serum creatinine (mg/dl) |
1.06±0.30 |
|
SOFA score |
8.09±3.52 |
|
APACHE II score |
20.09±7.52 |
|
Charlson comorbidity index |
3.69±2.16 |
|
Body surface area |
1.83±0.14 |
|
Body mass index (kg/m2) |
25.24±3.40 |
|
Ideal body weight (kg) |
64.37±7.00 |
|
AKI stage |
|
|
Stage 1 |
74 (77.89) |
|
Stage 2 |
14 (14.73) |
|
Stage 3 |
7 (7.36) |
Table 2.Past medical history
|
Variable |
No. (%) |
|
No past medical history |
19 (20.00) |
|
HTN |
19 (20.00) |
|
Solid tumor |
12 (12.63) |
|
HTN+DM |
8 (8.42) |
|
Other |
37 (38.94) |
Table 3.Correlation of different formula with measured creatinine clearance
|
Formula |
Mean GFR |
Correlation coefficient |
P-value |
|
Modified CG |
38.97±17.16 |
0.30 |
0.006 |
|
MDRD |
40.37±17.53 |
0.24 |
0.031 |
|
CKD-EPI |
37.49±17.61 |
0.27 |
0.015 |
|
Kinetic-GFR |
33.85±17.45 |
0.29 |
0.008 |
|
Brater |
58.94±25.33 |
0.41 |
0.010 |
|
Chiou |
40.19±20.67 |
0.26 |
0.018 |
|
CG |
41.51±17.31 |
0.26 |
0.018 |
|
Jelliffe |
36.43±18.97 |
0.32 |
0.001 |
Table 4.Correlation coefficients of formulas in each stage of AKI
|
Formula |
Stage
|
|
1 |
2 |
3 |
|
Modified CG |
0.24 |
0.53 |
–0.03 |
|
MDRD |
0.17 |
0.07 |
0.03 |
|
CKD-EPI |
0.20 |
0.29 |
0.00 |
|
Kinetic-GFR |
0.17 |
0.76 |
0.37 |
|
Brater |
0.23 |
0.51 |
0.18 |
|
Chiou |
0.26 |
0.26 |
0.03 |
|
CG |
0.19 |
0.23 |
0.10 |
|
Jelliffe |
0.25 |
0.51 |
0.37 |
Table 5.Dosing of the frequently prescribed antibiotics in the patients with acute kidney injury
|
Antibiotic |
Frequency (n=95) |
Underdosed |
Optimum |
overdosed |
|
Vancomycin |
52 |
10 |
25 |
17 |
|
Meropenem |
43 |
1 |
25 |
17 |
|
Piperacillin-tazobactam |
25 |
4 |
17 |
4 |
|
Levofloxacin |
24 |
1 |
15 |
8 |
|
Imipenem/cilastatin |
8 |
1 |
4 |
3 |
|
Ampicillin/sulbactam |
6 |
1 |
3 |
3 |
|
Colistin |
5 |
1 |
2 |
2 |
References
- 1. Singbartl K, Kellum JA. AKI in the ICU: definition, epidemiology, risk stratification, and outcomes. Kidney Int 2012;81:819-25.ArticlePubMed
- 2. Susantitaphong P, Cruz DN, Cerda J, Abulfaraj M, Alqahtani F, Koulouridis I, et al. World incidence of AKI: a meta-analysis. Clin J Am Soc Nephrol 2013;8:1482-93.ArticlePubMedPMC
- 3. Palevsky PM, Liu KD, Brophy PD, Chawla LS, Parikh CR, Thakar CV, et al. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for acute kidney injury. Am J Kidney Dis 2013;61:649-72.ArticlePubMed
- 4. Mohammadi Kebar S, Hosseini Nia S, Maleki N, Sharghi A, Sheshgelani A. The incidence rate, risk factors and clinical outcome of acute kidney injury in critical patients. Iran J Public Health 2018;47:1717-24.PubMedPMC
- 5. Blanco VE, Hernandorena CV, Scibona P, Belloso W, Musso CG. Acute kidney injury pharmacokinetic changes and its impact on drug prescription. Healthcare (Basel) 2019;7:10. ArticlePubMedPMC
- 6. Zamoner W, de Freitas FM, Garms DS, de Oliveira MG, Balbi AL, Ponce D. Pharmacokinetics and pharmacodynamics of antibiotics in critically ill acute kidney injury patients. Pharmacol Res Perspect 2016;4:e00280. Article
- 7. Veiga RP, Paiva JA. Pharmacokinetics-pharmacodynamics issues relevant for the clinical use of beta-lactam antibiotics in critically ill patients. Crit Care 2018;22:233. ArticlePubMedPMC
- 8. Matzke GR, Aronoff GR, Atkinson AJ Jr, Bennett WM, Decker BS, Eckardt KU, et al. Drug dosing consideration in patients with acute and chronic kidney disease-a clinical update from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 2011;80:1122-37.ArticlePubMed
- 9. Levey AS, Inker LA. GFR as the "gold standard": estimated, measured, and true. Am J Kidney Dis 2016;67:9-12.ArticlePubMed
- 10. Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, Acute Dialysis Quality Initiative workgroup. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care 2004;8:R204-12.ArticlePubMedPMC
- 11. Al-Dorzi HM, Alsadhan AA, Almozaini AS, M Alamri A, Tamim H, Sadat M, et al. The performance of equations that estimate glomerular filtration rate against measured urinary creatinine clearance in critically ill patients. Crit Care Res Pract 2021;2021:5520653. ArticlePubMedPMC
- 12. Hahn T, Yao S, Dunford LM, Thomas J, Lohr J, Arora P, et al. A comparison of measured creatinine clearance versus calculated glomerular filtration rate for assessment of renal function before autologous and allogeneic BMT. Biol Blood Marrow Transplant 2009;15:574-9.ArticlePubMed
- 13. Tsai D, Udy AA, Stewart PC, Gourley S, Morick NM, Lipman J, et al. Prevalence of augmented renal clearance and performance of glomerular filtration estimates in Indigenous Australian patients requiring intensive care admission. Anaesth Intensive Care 2018;46:42-50.ArticlePubMed
- 14. Chen S. Retooling the creatinine clearance equation to estimate kinetic GFR when the plasma creatinine is changing acutely. J Am Soc Nephrol 2013;24:877-88.ArticlePubMed
- 15. Xiong L, Lu ZK, Quidley AM, Alford BS, Bookstaver PB. Evaluation of renal function estimation formulas specific to dynamic renal function for drug dosing in critically ill patients. South Med J 2018;111:299-306.ArticlePubMed
- 16. Guilhaumou R, Benaboud S, Bennis Y, Dahyot-Fizelier C, Dailly E, Gandia P, et al. Optimization of the treatment with beta-lactam antibiotics in critically ill patients-guidelines from the French Society of Pharmacology and Therapeutics (Société Française de Pharmacologie et Thérapeutique-SFPT) and the French Society of Anaesthesia and Intensive Care Medicine (Société Française d'Anesthésie et Réanimation-SFAR). Crit Care 2019;23:104. ArticlePubMedPMC
- 17. Mehta RL, Kellum JA, Shah SV, Molitoris BA, Ronco C, Warnock DG, et al. Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury. Crit Care 2007;11:R31. Article
- 18. Shahrami B, Najmeddin F, Mousavi S, Ahmadi A, Rouini MR, Mojtahedzadeh M. CP-184 correlations of vancomycin clearance during intermittent infusion with measured and estimated creatinine clearance in critically ill patients: 6 hour urine collection may be beneficial. Eur J Hosp Pharm 2017;24(Suppl 1):A82. Article
- 19. Carson JS, Goverman J, Fagan SP. 31 - acute renal failure in association with thermal injury. In: Herndon DN, editor. Total burn care. 5th ed. Amsterdam: Elsevier; 2018. p. 318-27.
- 20. Kane-Gill SL, Sileanu FE, Murugan R, Trietley GS, Handler SM, Kellum JA. Risk factors for acute kidney injury in older adults with critical illness: a retrospective cohort study. Am J Kidney Dis 2015;65:860-9.ArticlePubMed
- 21. Williams P, Beall G, Cotta MO, Roberts JA. Antimicrobial dosing in critical care: a pragmatic adult dosing nomogram. Int J Antimicrob Agents 2020;55:105837. ArticlePubMed
- 22. Baptista JP, Udy AA, Sousa E, Pimentel J, Wang L, Roberts JA, et al. A comparison of estimates of glomerular filtration in critically ill patients with augmented renal clearance. Crit Care 2011;15:R139. ArticlePubMedPMC
- 23. Robert S, Zarowitz BJ, Peterson EL, Dumler F. Predictability of creatinine clearance estimates in critically ill patients. Crit Care Med 1993;21:1487-95.ArticlePubMed
- 24. Bouchard J, Macedo E, Soroko S, Chertow GM, Himmelfarb J, Ikizler TA, et al. Comparison of methods for estimating glomerular filtration rate in critically ill patients with acute kidney injury. Nephrol Dial Transplant 2010;25:102-7.ArticlePubMed
- 25. Jelliffe R. Estimation of creatinine clearance in patients with unstable renal function, without a urine specimen. Am J Nephrol 2002;22:320-4.ArticlePubMed
- 26. Pelletier K, Lafrance JP, Roy L, Charest M, Bélanger MC, Cailhier JF, et al. Estimating glomerular filtration rate in patients with acute kidney injury: a prospective multicenter study of diagnostic accuracy. Nephrol Dial Transplant 2020;35:1886-1893.ArticlePubMed
- 27. Carlier M, Dumoulin A, Janssen A, Picavet S, Vanthuyne S, Van Eynde R, et al. Comparison of different equations to assess glomerular filtration in critically ill patients. Intensive Care Med 2015;41:427-35.ArticlePubMed
- 28. Delanaye P, Cavalier E, Pottel H. Serum creatinine: not so simple! Nephron 2017;136:302-8.ArticlePubMed
- 29. Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 2012;120:c179-84.ArticlePubMed
- 30. Crass RL, Rodvold KA, Mueller BA, Pai MP. Renal dosing of antibiotics: are we jumping the gun? Clin Infect Dis 2019;68:1596-602.ArticlePubMed
- 31. Macedo E, Bouchard J, Soroko SH, Chertow GM, Himmelfarb J, Ikizler TA, et al. Fluid accumulation, recognition and staging of acute kidney injury in critically-ill patients. Crit Care 2010;14:R82. Article
- 32. Sunder S, Jayaraman R, Mahapatra HS, Sathi S, Ramanan V, Kanchi P, et al. Estimation of renal function in the intensive care unit: the covert concepts brought to light. J Intensive Care 2014;2:31. ArticlePubMedPMC
- 33. Sangla F, Marti PE, Verissimo T, Pugin J, de Seigneux S, Legouis D. Measured and estimated glomerular filtration rate in the ICU: a prospective study. Crit Care Med 2020;48:e1232-41.ArticlePubMed
- 34. Tomasa-Irriguible TM, Sabater-Riera J, Pérez-Carrasco M, Ortiz-Ballujera P, Díaz-Buendía Y, Navas-Pérez A, et al. Augmented renal clearance: an unnoticed relevant event. Sci Prog 2021;104:368504211018580. ArticlePubMed
- 35. Schortgen F, Schetz M. Does this critically ill patient with oliguria need more fluids, a vasopressor, or neither? Intensive Care Med 2017;43:907-10.ArticlePubMed
- 36. Albanèse J, Leone M, Delmas A, Martin C. Terlipressin or norepinephrine in hyperdynamic septic shock: a prospective, randomized study. Crit Care Med 2005;33:1897-902.ArticlePubMed
- 37. Chen S. Kinetic glomerular filtration rate in routine clinical practice-applications and possibilities. Adv Chronic Kidney Dis 2018;25:105-14.ArticlePubMed
- 38. Hoste EA, Damen J, Vanholder RC, Lameire NH, Delanghe JR, Van den Hauwe K, et al. Assessment of renal function in recently admitted critically ill patients with normal serum creatinine. Nephrol Dial Transplant 2005;20:747-53.ArticlePubMed
- 39. Kwong YD, Chen S, Bouajram R, Li F, Matthay MA, Mehta KM, et al. The value of kinetic glomerular filtration rate estimation on medication dosing in acute kidney injury. PLoS One 2019;14:e0225601. ArticlePubMedPMC
- 40. Seelhammer TG, Maile MD, Heung M, Haft JW, Jewell ES, Engoren M. Kinetic estimated glomerular filtration rate and acute kidney injury in cardiac surgery patients. J Crit Care 2016;31:249-54.ArticlePubMed
- 41. Bairy M, See FH, Lim RS. Using the kinetic estimating glomerular filtration rate equation for estimating glomerular filtration rate and detecting acute kidney injury: a pilot study. Nephron 2018;140:231-9.ArticlePubMed
Citations
Citations to this article as recorded by

- SWOT and Root Cause Analyses of Antimicrobial Resistance to Oral Antimicrobial Treatment of Cystitis
Pradeep Tyagi, Shachi Tyagi, Laurence Stewart, Scott Glickman
Antibiotics.2024; 13(4): 328. CrossRef - Prospective Comparison of Urinary Measured Creatinine Clearance With eGFR and Cystatin C Based Cis-eGFR, Including Kinetic eGFR in the Immediate Post-transplant Period With Prompt Allograft Function
Ashokkumar Jain, Umar Farooq, Nasrollah Ghahramani, Deborah Daoud, Eileen Swartz, Christopher Hamilton, Alireza Vafaei Sadr, Thomas Butler
Transplantation Proceedings.2024;[Epub] CrossRef - Estimated glomerular filtration rates are higher when creatinine‐based equations are compared with a cystatin C‐based equation in coronavirus disease 2019
Anders O. Larsson, Michael Hultström, Robert Frithiof, Miklos Lipcsey, Ulf Nyman, Mats B. Eriksson
Acta Anaesthesiologica Scandinavica.2023; 67(2): 213. CrossRef - Selection of appropriate reference creatinine estimate for acute kidney injury diagnosis in patients with severe trauma
Kangho Lee, Dongyeon Ryu, Hohyun Kim, Sungjin Park, Sangbong Lee, Chanik Park, Gilhwan Kim, Sunhyun Kim, Nahyeon Lee
Acute and Critical Care.2023; 38(1): 95. CrossRef
 , Fatemeh Heydarpour2
, Fatemeh Heydarpour2 , Hasanali Karimpour3
, Hasanali Karimpour3 , Foroud Shahbazi1
, Foroud Shahbazi1





 KSCCM
KSCCM

 PubReader
PubReader ePub Link
ePub Link Cite
Cite