Articles
- Page Path
- HOME > Acute Crit Care > Volume 37(1); 2022 > Article
-
Original Article
Infection Nosocomial meningitis in intensive care: a 10-year retrospective study and literature review -
Sofia R. Valdoleiros1
 , Cristina Torrão2
, Cristina Torrão2 , Laura S. Freitas3
, Laura S. Freitas3 , Diana Mano2
, Diana Mano2 , Celina Gonçalves2
, Celina Gonçalves2 , Carla Teixeira2,4,5
, Carla Teixeira2,4,5
-
Acute and Critical Care 2022;37(1):61-70.
DOI: https://doi.org/10.4266/acc.2021.01151
Published online: January 26, 2022
1Department of Infectious Diseases, Centro Hospitalar Universitário de São João, Porto, Portugal
2Department of Intensive Care, Centro Hospitalar Universitário do Porto, Porto, Portugal
3Department of Emergency, Centro Hospitalar de Entre o Douro e Vouga, Santa Maria da Feira, Portugal
4Instituto de Ciências Biomédicas Abel Salazar, Universidade do Porto, Porto, Portugal
5CriticalMed Cintesis, Centro de Investigação em Tecnologias e Serviços de Saúde, Faculdade de Medicina da Universidade do Porto, Porto, Portugal
- Corresponding author: Sofia R. Valdoleiros, Department of Infectious Diseases, Centro Hospitalar Universitário de São João, Alameda Prof. Hernâni Monteiro, 4200-319 Porto, Portugal Tel: +351-222-077-500, E-mail: sofia.valdoleiros@chsj.min-saude.pt
Copyright © 2022 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Nosocomial meningitis is a medical emergency that requires early diagnosis, prompt initiation of therapy, and frequent admission to the intensive care unit (ICU).
-
Methods
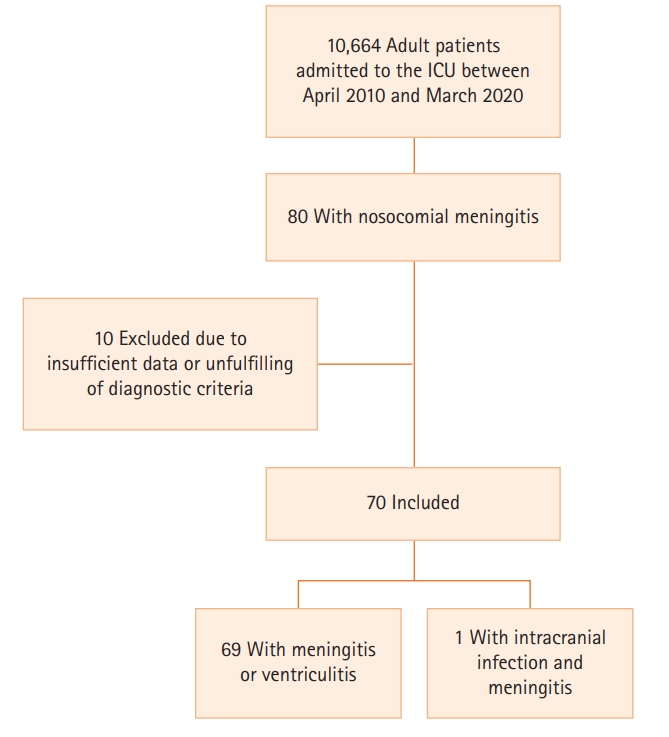
- A retrospective study was conducted in adult patients diagnosed with nosocomial meningitis who required admission to the ICU between April 2010 and March 2020. Meningitis/ventriculitis and intracranial infection were defined according to Centers for Disease Control and Prevention guidelines.
-
Results
- An incidence of 0.75% of nosocomial meningitis was observed among 70 patients. The mean patient age was 59 years and 34% were ≥65 years. Twenty-two percent of patients were in an immunocompromised state. A clear predisposing factor for nosocomial meningitis (traumatic brain injury, basal skull fracture, brain hemorrhage, central nervous system [CNS] invasive procedure or device) was present in 93% of patients. Fever was the most frequent clinical feature. A microbiological agent was identified in 30% of cases, of which 27% were bacteria, with a predominance of Gram-negative over Gram-positive. Complications developed in 47% of cases, 24% of patients were discharged with a Glasgow coma scale <14, and 37% died. There were no clear clinical predictors of complications. Advanced age (≥65 years old) and the presence of complications were associated with higher hospital mortality.
-
Conclusions
- Nosocomial meningitis in critical care has a low incidence rate but high mortality and morbidity. In critical care patients with CNS-related risk factors, a high level of suspicion for meningitis is warranted, but diagnosis can be hindered by several confounding factors.
INTRODUCTION
MATERIALS AND METHODS
RESULTS
DISCUSSION
KEY MESSAGES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conceptualization: SRV, CT, LSF, CG, CT. Data curation: SRV, CT, LSF, DM. Formal analysis: SRV, CT, LSF, DM, CG. Methodology: SRV, CT, LSF, DM. Project administration: SRV, CT. Validation: SRV, CT, CG. Visualization: SRV, CT. Writing–original draft: SRV, CT, LSF, DM. Writing–review & editing: SRV, CT, LSF, CG, CT.
NOTES
Acknowledgments
SUPPLEMENTARY MATERIALS
Supplementary Table 1.
| Treatment | Value |
|---|---|
| Empirical antimicrobial therapy | |
| Ceftazidime plus vancomycin | 42 (60) |
| Meropenem plus vancomycin | 22 (31.4) |
| Route of therapy | |
| Intravenous only | 69 (98.6) |
| Intravenous and intraventricular | 1 (1.4) |
| Duration of antibiotic therapy (day)a | 18.0±8.3 |
| Other therapeutic measure | |
| Corticoid | 13 (19.4) |
| Intracranial hypertension management | |
| Antiedematous therapy | 8 (11.4) |
| Barbiturate coma therapy | 2 (2.8) |
| CSF shunt | 11 (15.7) |
| Craniectomy | 2 (2.9) |
| Neurosurgery for infection control | 4 (5.7) |
| Removal of CNS device | 2 (2.9) |
| Closure of CSF fistula | 1 (1.4) |
| Variable |
Any complication |
Non-alert at dischargea |
30-Day mortality and hospital mortality |
|||
|---|---|---|---|---|---|---|
| OR (95% CI) | P-value | OR (95% CI) | P-value | OR (95% CI) | P-value | |
| Demographics and underlying disease | ||||||
| Age ≥65 yr | 0.71 (0.26–1.93) | 0.51 | 1.30 (0.28–5.73) | 0.71 | 8.89 (2.42–32.68) | 0.001 |
| Male | 1.06 (0.40–2.82) | 0.90 | 7.60 (0.89–64.62) | 0.04 | 0.86 (0.27–2.76) | 1 |
| Previous partially dependent | 0.53 (0.09–3.12) | 0.68 | 0.80 (0.69–0.91) | 1 | 9.64 (1.57–59.32) | 0.02 |
| Immunocompromised | 0.80 (0.29–2.19) | 0.67 | 1.98 (0.51–7.68) | 0.47 | 1.03 (0.31–3.46) | 1 |
| Clinical feature | ||||||
| Fever at diagnosis | 0.16 (0.01–1.1) | 0.06 | 1.28 (1.11–1.48) | 0.57 | 0.65 (0.11–3.74) | 0.64 |
| GCS at diagnosis <8 pointsb | 0.68 (0.23–2.00) | 0.48 | 4.09 (0.83–20.14) | 0.71 | 0.94 (0.27–3.22) | 1 |
| New focal neurological deficit at diagnosisb | 5.67 (0.56–57.23) | 0.16 | - | 0.08 | 3.94 (0.55–28.11) | 0.3 |
| Complication (any) | NA | NA | 0.45 (0.11–1.92) | 0.33 | 2.78 (0.83–9.24) | 0.09 |
| Hospital stay (mean) | 2 (–37 to 13) | 0.34 | 35 (–94 to 61) | 0.65 | 30.4 (12.7–48.05) | 0.05 |
| Laboratory findings | ||||||
| C-reactive protein ≥100 mg/L | 1.29 (0.50–3.31) | 0.60 | 1.00 (0.26–3.77) | 1 | 0.95 (0.30–2.99) | 0.93 |
| CSF white cell ≥100 per μl | 1.10 (0.33–3.61) | 0.88 | 3.12 (0.36–27.50) | 0.42 | 0.79 (0.18–3.40) | 0.71 |
| CSF glucose level <0.30 g/L | 0.99 (0.24–4.09) | 1 | 3.56 (0.66–19.11) | 0.15 | 1.31 (0.24–7.29) | 0.67 |
| CSF protein level ≥1 g/L | 1.52 (0.55–4.17) | 0.42 | 0.51 (0.13–1.96) | 0.49 | 1.42 (0.38–5.31) | 0.75 |
| Concomitant bacteremia | 1.15 (0.33–3.98) | 0.83 | 0.63 (0.07–5.88) | 1 | 3.43 (0.90–13.02) | 0.11 |
| Negative CSF culture | 0.56 (0.20–1.58) | 0.27 | 0.78 (0.18–3.52) | 0.71 | 0.27 (0.08–0.89) | 0.05 |
| Treatment | ||||||
| Meropenem plus vancomycinc | 3.15 (1.08–9.22) | 0.03 | 0.96 (0.21–4.30) | 1 | 2.25 (0.63–8.06) | 0.31 |
| Antibiotic treatment >14 daysd | 2.84 (0.91–8.86) | 0.07 | 1.48 (0.29–7.54) | 0.70 | 1.05 (0.19–5.74) | 1 |
| Steroid therapy | 3.54 (0.97–12.96) | 0.05 | 4.34 (0.93–20.30) | 0.07 | 2.22 (0.56–8.82) | 0.26 |
| Antiedematous therapy (including barbiturates) | - | 0.001 | 1.41 (0.24–8.16) | 0.65 | - | 0.19 |
| CSF shunt | 1.42 (0.39–5.18) | 0.59 | 0.45 (0.05–4.04) | 0.67 | 0.79 (0.15–4.10) | 1 |
| Neurology consult | 0.92 (0.34–2.48) | 0.87 | 5.25 (1.29–21.40) | 0.03 | 1.37 (0.42–4.44) | 0.60 |
| Neurosurgery consult | 1.69 (0.45–6.40) | 0.44 | 1.58 (0.17–14.66) | 1 | 0.40 (0.10–1.61) | 0.23 |
- 1. Robinson CP, Busl KM. Meningitis and encephalitis management in the ICU. Curr Opin Crit Care 2019;25:423-29.ArticlePubMed
- 2. Kurtaran B, Kuscu F, Ulu A, Inal AS, Komur S, Kibar F, et al. The causes of postoperative meningitis: the comparison of Gram-negative and Gram-positive pathogens. Turk Neurosurg 2018;28:589-96.ArticlePubMed
- 3. Hussein K, Bitterman R, Shofty B, Paul M, Neuberger A. Management of post-neurosurgical meningitis: narrative review. Clin Microbiol Infect 2017;23:621-8.ArticlePubMed
- 4. van de Beek D, Drake JM, Tunkel AR. Nosocomial bacterial meningitis. N Engl J Med 2010;362:146-54.ArticlePubMed
- 5. Baer ET. Post-dural puncture bacterial meningitis. Anesthesiology 2006;105:381-93.ArticlePubMed
- 6. Baltas I, Tsoulfa S, Sakellariou P, Vogas V, Fylaktakis M, Kondodimou A. Posttraumatic meningitis: bacteriology, hydrocephalus, and outcome. Neurosurgery 1994;35:422-6.PubMed
- 7. Choi D, Spann R. Traumatic cerebrospinal fluid leakage: risk factors and the use of prophylactic antibiotics. Br J Neurosurg 1996;10:571-5.ArticlePubMed
- 8. Bullock MR, Chesnut R, Ghajar J, Gordon D, Hartl R, Newell DW, et al. Surgical management of depressed cranial fractures. Neurosurgery 2006;58(3 Suppl):S56-60.ArticlePubMed
- 9. Hoogmoed J, van de Beek D, Coert BA, Horn J, Vandertop WP, Verbaan D. Clinical and laboratory characteristics for the diagnosis of bacterial ventriculitis after aneurysmal subarachnoid hemorrhage. Neurocrit Care 2017;26:362-70.ArticlePubMed
- 10. Frontera JA, Fernandez A, Schmidt JM, Claassen J, Wartenberg KE, Badjatia N, et al. Impact of nosocomial infectious complications after subarachnoid hemorrhage. Neurosurgery 2008;62:80-7.ArticlePubMed
- 11. Tunkel AR, Glaser CA, Bloch KC, Sejvar JJ, Marra CM, Roos KL, et al. The management of encephalitis: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis 2008;47:303-27.ArticlePubMed
- 12. Busl KM. Nosocomial infections in the neurointensive care unit. Neurol Clin 2017;35:785-807.ArticlePubMed
- 13. Zarrouk V, Vassor I, Bert F, Bouccara D, Kalamarides M, Bendersky N, et al. Evaluation of the management of postoperative aseptic meningitis. Clin Infect Dis 2007;44:1555-9.ArticlePubMed
- 14. Schade RP, Schinkel J, Roelandse FW, Geskus RB, Visser LG, van Dijk JM, et al. Lack of value of routine analysis of cerebrospinal fluid for prediction and diagnosis of external drainage-related bacterial meningitis. J Neurosurg 2006;104:101-8.ArticlePubMed
- 15. Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 2008;36:309-32.ArticlePubMed
- 16. Governale LS, Fein N, Logsdon J, Black PM. Techniques and complications of external lumbar drainage for normal pressure hydrocephalus. Neurosurgery 2008;63(4 Suppl 2):379-84.ArticlePubMed
- 17. Palabiyikoglu I, Tekeli E, Cokca F, Akan O, Unal N, Erberktas I, et al. Nosocomial meningitis in a university hospital between 1993 and 2002. J Hosp Infect 2006;62:94-7.ArticlePubMed
- 18. Dizbay M, Tunçcan OG, Arman D, Baş S, Gürsoy A, Aktaş F. Factors associated with mortality in nosocomial central nervous system infections. ANKEM Derg 2011;25:6-11.Article
- 19. Srihawan C, Castelblanco RL, Salazar L, Wootton SH, Aguilera E, Ostrosky-Zeichner L, et al. Clinical characteristics and predictors of adverse outcome in adult and pediatric patients with healthcare-associated ventriculitis and meningitis. Open Forum Infect Dis 2016;3:ofw077. ArticlePubMedPMC
- 20. Lu CH, Chang WN, Chuang YC, Chang HW. The prognostic factors of adult gram-negative bacillary meningitis. J Hosp Infect 1998;40:27-34.ArticlePubMed
- 21. Ozer B, Ozbakıs Akkurt BC, Duran N, Onlen Y, Savas L, Turhanoglu S. Evaluation of nosocomial infections and risk factors in critically ill patients. Med Sci Monit 2011;17:PH17-22. ArticlePubMed
- 22. Kim HI, Kim SW, Park GY, Kwon EG, Kim HH, Jeong JY, et al. The causes and treatment outcomes of 91 patients with adult nosocomial meningitis. Korean J Intern Med 2012;27:171-9.ArticlePubMedPMC
- 23. Wang KW, Chang WN, Huang CR, Tsai NW, Tsui HW, Wang HC, et al. Post-neurosurgical nosocomial bacterial meningitis in adults: microbiology, clinical features, and outcomes. J Clin Neurosci 2005;12:647-50.ArticlePubMed
- 24. Logigan C, Mihalache D, Dorneanu O, Turcu T. Study of nosocomial bacillary meningitis admitted in the Clinic of Infectious Diseases Iaşi on a 20 years period. Rev Med Chir Soc Med Nat Iasi 2009;113:721-6.PubMed
- 25. Dimitriu SM, Luca V, Turcu T, Scurtu C, Prisecaru L. Acute nosocomial bacterial meningitis: apropos 27 cases. Rev Med Chir Soc Med Nat Iasi 1992;96:19-25.
- 26. Commichau C, Scarmeas N, Mayer SA. Risk factors for fever in the neurologic intensive care unit. Neurology 2003;60:837-41.ArticlePubMed
- 27. Stocchetti N, Rossi S, Zanier ER, Colombo A, Beretta L, Citerio G. Pyrexia in head-injured patients admitted to intensive care. Intensive Care Med 2002;28:1555-62.ArticlePubMed
- 28. Shi YJ, Zheng GH, Qian LY, Qsman RA, Li GG, Zhang GJ. Longitudinal analysis of risk factors for clinical outcomes of enterobacteriaceae meningitis/encephalitis in post-neurosurgical patients: a comparative cohort study during 2014-2019. Infect Drug Resist 2020;13:2161-70.PubMedPMC
- 29. Erdem I, Hakan T, Ceran N, Metin F, Akcay SS, Kucukercan M, et al. Clinical features, laboratory data, management and the risk factors that affect the mortality in patients with postoperative meningitis. Neurol India 2008;56:433-7.ArticlePubMed
- 30. Tunkel AR, Hartman BJ, Kaplan SL, Kaufman BA, Roos KL, Scheld WM, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis 2004;39:1267-84.ArticlePubMed
- 31. Federico G, Tumbarello M, Spanu T, Rosell R, Iacoangeli M, Scerrati M, et al. Risk factors and prognostic indicators of bacterial meningitis in a cohort of 3580 postneurosurgical patients. Scand J Infect Dis 2001;33:533-7.ArticlePubMed
- 32. Weisfelt M, van de Beek D, Spanjaard L, de Gans J. Nosocomial bacterial meningitis in adults: a prospective series of 50 cases. J Hosp Infect 2007;66:71-8.ArticlePubMed
- 33. Chouhdari A, Ebrahimzadeh K, Rezaei O, Samadian M, Sharifi G, Hajiesmaeili M. Investigating related factors with mortality rate in patients with postoperative meningitis: One longitudinal follow up study in Iran. Iran J Neurol 2018;17:82-5.PubMedPMC
References
Figure & Data
References
Citations

- Procalcitonin As Diagnostic Tool for CNS Infections—Overall, Not Good Enough (Yet?)*
Michael A. Pizzi, Katharina M. Busl
Critical Care Medicine.2024; 52(1): 163. CrossRef - A retrospective analysis of 20,178 adult neurological infection admissions to United Kingdom critical care units from 2001 to 2020
Joseph Donovan, Abena Glover, John Gregson, Andrew W. Hitchings, Emma C. Wall, Robert S. Heyderman
BMC Infectious Diseases.2024;[Epub] CrossRef - Clinical Characteristics, Treatment, and Outcomes of Veterans with Cerebrospinal Fluid Culture Positive for Gram-Negative Rod Bacteria: A Retrospective Analysis over 18 Years in 125 Veterans Health Administration Hospitals
Shinya Hasegawa, Eiyu Matsumoto, Jennifer R. Carlson, Hiroyuki Suzuki
Current Microbiology.2024;[Epub] CrossRef - Confronting multidrug-resistantKlebsiellameningitis after mid-clival Cerebrospinal Fluid leak repair: a therapeutic odyssey
Anbarasi Madoure, Dharanya Gopalakrishnan Srinivasan, Tejaswi Mishra, Lokesh Kumar Penubarthi
BMJ Case Reports.2024; 17(6): e257872. CrossRef - Glioma grade and post-neurosurgical meningitis risk
Sakke Niemelä, Jarmo Oksi, Jussi Jero, Eliisa Löyttyniemi, Melissa Rahi, Jaakko Rinne, Jussi P. Posti, Dan Laukka
Acta Neurochirurgica.2024;[Epub] CrossRef - Bacterial meningitis in adults: a retrospective study among 148 patients in an 8-year period in a university hospital, Finland
Sakke Niemelä, Laura Lempinen, Eliisa Löyttyniemi, Jarmo Oksi, Jussi Jero
BMC Infectious Diseases.2023;[Epub] CrossRef - Bacterial meningitis in children with an abnormal craniocerebral structure
Jiali Pan, Wei Xu, Wenliang Song, Tao Zhang
Frontiers in Pediatrics.2023;[Epub] CrossRef - Fieber in der Intensivmedizin
Jan-Hendrik Naendrup, Boris Böll, Jorge Garcia Borrega
Intensivmedizin up2date.2023; 19(01): 17. CrossRef - Neurosurgical management of penetrating brain injury during World War I: A historical cohort
Rayan Fawaz, Mathilde Schmitt, Philémon Robert, Nathan Beucler, Jean-Marc Delmas, Nicolas Desse, Aurore Sellier, Arnaud Dagain
Neurochirurgie.2023; 69(3): 101439. CrossRef - Etiology and Outcomes of Healthcare-Associated Meningitis and Ventriculitis—A Single Center Cohort Study
Hana Panic, Branimir Gjurasin, Marija Santini, Marko Kutlesa, Neven Papic
Infectious Disease Reports.2022; 14(3): 420. CrossRef - Healthcare-associated central nervous system infections
Mariachiara Ippolito, Antonino Giarratano, Andrea Cortegiani
Current Opinion in Anaesthesiology.2022; 35(5): 549. CrossRef

 KSCCM
KSCCM

 PubReader
PubReader ePub Link
ePub Link Cite
Cite