Abstract
- Effective use of noninvasive ventilation in patients with chronic obstructive pulmonary disease is well-known. However, noninvasive ventilation in patients presenting with altered sensorium and severe acidosis (pH <7.1) has been rarely described. Invasive mechanical ventilation is associated with high mortality in coronavirus disease 2019 (COVID-19), and use of noninvasive ventilation over invasive ventilation is an area of investigation. We report a case of COVID-19-induced acute exacerbation of chronic obstructive pulmonary disease in a 66-year-old male. His past medical history included obstructive sleep apnea, hypertension, cor pulmonale, atrial fibrillation, and amiodarone-induced hypothyroidism. On presentation, he had acute hypercapnic respiratory failure, severe acidosis (partial pressure of carbon dioxide [PCO2], 147 mm Hg; pH, 7.06), and altered mentation. The patient was successfully managed with noninvasive ventilation, avoiding endotracheal intubation, invasive ventilation, and related complications. Although precarious, a trial of noninvasive ventilation can be considered in COVID-19-induced acute exacerbation of chronic obstructive pulmonary disease with hypercapnic respiratory failure, severe acidosis, and altered mentation.
-
Keywords: case report; chronic obstructive pulmonary disease; coma; COVID-19; noninvasive ventilation
Noninvasive ventilation (NIV) is beneficial as a first‐line intervention in patients with acute hypercapnic respiratory failure (AHRF) secondary to acute exacerbation of chronic obstructive pulmonary disease (AECOPD). NIV is likely to reduce endotracheal intubation (ETI), intensive care unit (ICU) admission, morbidity, and mortality [1]. Altered consciousness, especially if severely depressed the need for immediate ETI and is considered a contraindication to use of NIV as it can predispose a patient to aspiration [2]. Although an association between the level of consciousness with NIV failure or in-hospital mortality in patients with acute hypoxic respiratory failure has not been found [3], severe acidosis (pH < 7.2) predicted prolonged NIV in severe AECOPD [4]. Despite the advantages of NIV, and even though its proper use has shown rapid clinical improvement in patients, some clinicians question the choice of NIV over invasive mechanical ventilation (IMV). For example, some reports predict failure of NIV at pH <7.25, and NIV is not widely used as a first-line modality of ventilation in such conditions [4,5]. We report a case of hypercapnic coma with severe respiratory acidosis in a patient with coronavirus disease 2019 (COVID-19)-induced AECOPD in which NIV was successfully used as first-line respiratory therapy.
CASE REPORT
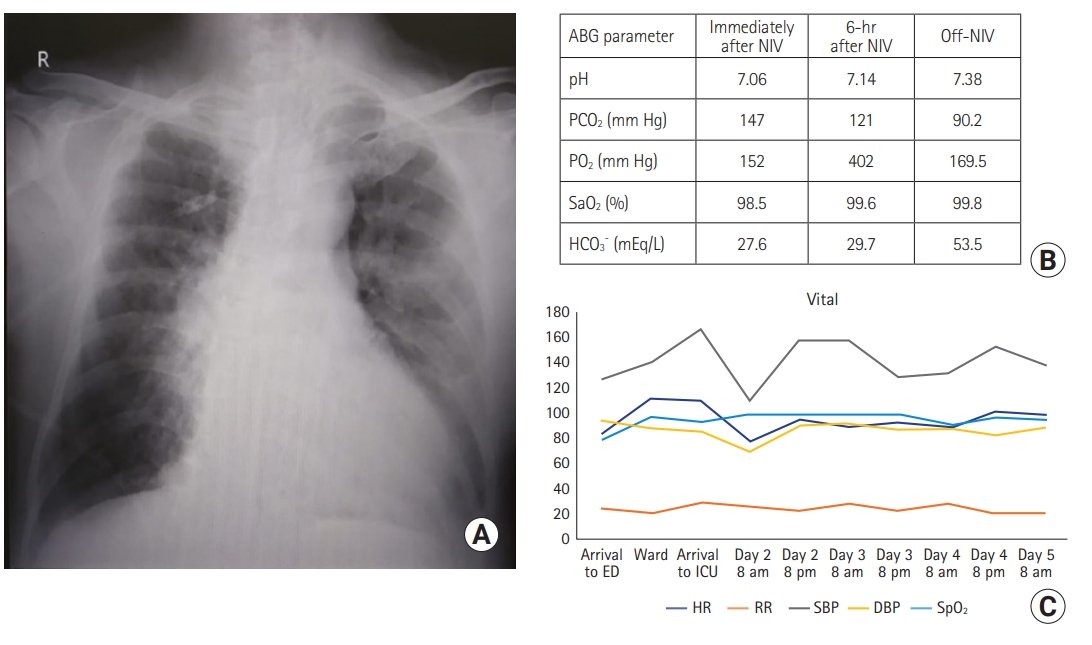
Informed consent for publication was obtained from the patient. In our institute, ethical approval is not required for case reports. A 66-year-old male with no history of fever or travel was admitted to an outside institution with dyspnea and occasional cough. He tested positive for COVID-19 and was referred to our hospital. His past medical history included past smoker (80 pack-years), obstructive sleep apnea (OSA) for 40 years, and COPD for 10 years. He had been on home NIV, metered dose inhaler, and oxygen therapy for the previous 6 years. He also had suffered hypertension, cor-pulmonale, atrial fibrillation on amiodarone, and amiodarone-induced hypothyroidism for the previous 2 years. On arrival to the emergency department, he was conscious, tachypneic, and hemodynamically stable but desaturated. His oxygen saturation as measured by pulse oximetry (SpO2) improved to 94% on oxygen via a simple facemask at 10 L/min. Although the patient’s oxygen saturation level was maintained throughout the evening, he became drowsy (Glasgow coma scale [GCS] E2V3M4) in the night due to carbon dioxide retention (arterial blood gas [ABG]: partial pressure of carbon dioxide [pCO2], 134 mm Hg; pH, 7.0). Therefore, he was transferred to the COVID-19 ICU and immediately put on NIV. Electrocardiogram showed new-onset premature ventricular contractions. ABG reports immediately after initiating NIV, at 6 hours, and off NIV are shown in the table within the Figure 1B. Chest X-ray revealed mild COVID-19-related changes. After NIV for 24 hours, the patient’s sensorium (GCS E4V5M6) and overall clinical condition improved. He was shifted to oxygen via face mask at 6 L/min, which was later reduced to oxygen via nasal prongs at 4 L/min with SpO2 97%. However, considering his history of home NIV and OSA, NIV support was continued at night. He was afebrile throughout. Laboratory evaluations revealed a total leucocyte count within 4 hours of admission at 7,900/dl, and C-reactive protein on day 2 of admission was 88 mg/L. The patient was treated conservatively with the following empirical broad-spectrum antibiotics and medications: piperacillin/tazobactam 4.5 g intravenous injection, azithromycin 500 mg oral tablet, hydroxychloroquine 400 mg oral tablet twice on the first day and 200 mg twice a day for four days, dexamethasone 6 mg once per day, multivitamins, vitamin C, zinc, and one low-molecular-weight heparin 0.6 ml subcutaneous injection. The following cardiovascular medications were administered: amiodarone, metoprolol, amlodipine, and torsemide. Finally, the following respiratory medications were administered: metered dose inhaler tiotropium two puffs (9 µg each) OD, N-acetyl cysteine oral tablet, and oral doxofylline along with other supportive measures. After admission to the ICU, he remained afebrile, and his vital signs remained stable (Figure 1). His COVID-19 reverse transcription-polymerase chain reaction report returned negative, and he was discharged to the COVID-19 ward in stable condition on the fourth hospital day. The patient was subsequently discharged home on his chronic medication and home-NIV at night.
DISCUSSION
In the present case, NIV helped avoid ETI and its associated complications. A significant number of clinicians do not initiate treatment with NIV for fear of increased morbidity due to contraindications of NIV use, inexperience, unavailability of NIV, etc. [6]. In our case, the patient’s routine use of NIV at home during the night bolstered our decision to initiate NIV. In our opinion, carbon dioxide retention led to narcosis and altered consciousness, and improvement of the clinical condition and ABG can be attributed almost entirely to early use of NIV in the critical care unit. High-flow nasal cannula was not used in this case because it is primarily intended for improving oxygenation, and its superiority over NIV in managing AHRF is not established [7].
NIV achieves the same physiological response and rapid gas exchange as conventional mechanical ventilation [8,9]. Improvement in gas exchange, as evidenced by a reduction in the PCO2 in the blood and improvement in pH within a few hours of starting NIV, predicts success in patients who are cooperative and tolerant of the NIV interface and have control over their airway secretions [10]. Moreover, the time, resources, and expertise required to initiate NIV are relatively less than those for performing IMV.
The literature indicates that COPD acts as a risk factor for severe COVID-19 and associated mortality [11]. Although coronaviruses can cause AECOPD, whether COVID-19 can precipitate AECOPD or not is under investigation [12]. However, acute exacerbation, which is defined as acute changes in symptoms needing a change in treatment, is often a clinical diagnosis [13]. Before his infection with COVID-19, the patient in the present case was stable on medications and home NIV, and the AECOPD leading to AHRF was precipitated by COVID-19 infection. Despite limited evidence of NIV use in patients with blood pH <7.25 and GCS 9/15, our patient did well. Close monitoring in the ICU was a crucial part of our management that allowed deference of IMV and its related complications. Nevertheless, the benefits of NIV over ETI and IMV should be weighed in each patient. When deciding whether to start NIV and continue the same for 24 hours, we considered certain factors, such as patient history and initial clinical condition, our ICU setup, and the team's ability to continuously monitor the patient and intervene both immediately and rapidly in case of clinical deterioration. These considerations allowed us to successfully use NIV as first-line therapy in a hypercapnic, comatose, and severely acidotic AECOPD patient with multiple comorbidities and COVID-19 infection in whom ETI and IMV would have been performed otherwise. Recently, the use of NIV in altered consciousness is gaining acceptability [3,14], and our case supports the effectiveness of NIV, even in COVID-19-induced AECOPD.
With appropriate patient selection that is guided by detailed history, physical examination, and proper investigations, NIV can successfully be used in comatose AHRF caused by AECOPD.
NOTES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conceptualization: JAP, HMRK. Data curation: all authors. Formal analysis: HMRK. Methodology: all authors. Writing–original draft: JAP, MPK, PPN. Writing–review & editing: HMRK.
Figure 1.(A) Mild ground-glass opacities left>right lung field with chronic obstructive pulmonary disease-related changes. (B) The arterial blood gas (ABG) parameters and (C) vital signs over time. NIV: noninvasive ventilation; pCO2: partial pressure of carbon dioxide; pO2: arterial partial pressure of oxygen; SaO2: peripheral oxygen saturation; HCO3: bicarbonate; ED: emergency department; ICU: intensive care unit; HR: heart rate; RR: respiratory rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; SpO2: oxygen saturation as measured by pulse oximetry.

References
- 1. Osadnik CR, Tee VS, Carson-Chahhoud KV, Picot J, Wedzicha JA, Smith BJ. Non-invasive ventilation for the management of acute hypercapnic respiratory failure due to exacerbation of chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2017;7:CD004104. ArticlePubMed
- 2. Addala D, Shrimanker R, Davies MG. Non-invasive ventilation: initiation and initial management. Br J Hosp Med (Lond) 2017;78:C140-4.ArticlePubMed
- 3. Kogo M, Nagata K, Ito J, Sato Y, Teraoka S, Kato R, et al. Noninvasive positive pressure ventilation for the treatment of acute hypoxic respiratory failure with altered consciousness level. Eur Respir J 2016;48(Suppl 60):PA3573. Article
- 4. Steriade AT, Johari S, Sargarovschi N, Necula D, Tudose CE, Ionita D, et al. Predictors of outcome of noninvasive ventilation in severe COPD exacerbation. BMC Pulm Med 2019;19:131. ArticlePubMedPMC
- 5. Ozsancak Ugurlu A, Habesoglu MA. Epidemiology of NIV for acute respiratory failure in COPD patients: results from the international surveys vs. the "real world". COPD 2017;14:429-38.ArticlePubMed
- 6. Sweet DD, Naismith A, Keenan SP, Sinuff T, Dodek PM. Missed opportunities for noninvasive positive pressure ventilation: a utilization review. J Crit Care 2008;23:111-7.ArticlePubMed
- 7. Lee HW, Choi SM, Lee J, Park YS, Lee CH, Yoo CG, et al. Reduction of PaCO2 by high-flow nasal cannula in acute hypercapnic respiratory failure patients receiving conventional oxygen therapy. Acute Crit Care 2019;34:202-11.ArticlePubMedPMC
- 8. Meduri GU, Conoscenti CC, Menashe P, Nair S. Noninvasive face mask ventilation in patients with acute respiratory failure. Chest 1989;95:865-70.ArticlePubMed
- 9. Schreiber A, Fusar Poli B, Bos LD, Nenna R. Noninvasive ventilation in hypercapnic respiratory failure: from rocking beds to fancy masks. Breathe (Sheff) 2018;14:235-7.ArticlePubMedPMC
- 10. Meduri GU, Abou-Shala N, Fox RC, Jones CB, Leeper KV, Wunderink RG. Noninvasive face mask mechanical ventilation in patients with acute hypercapnic respiratory failure. Chest 1991;100:445-54.ArticlePubMed
- 11. Alqahtani JS, Oyelade T, Aldhahir AM, Alghamdi SM, Almehmadi M, Alqahtani AS, et al. Prevalence, severity and mortality associated with COPD and smoking in patients with COVID-19: a rapid systematic review and meta-analysis. PLoS One 2020;15:e0233147.ArticlePubMedPMC
- 12. Simons SO, Hurst JR, Miravitlles M, Franssen FM, Janssen DJ, Papi A, et al. Caring for patients with COPD and COVID-19: a viewpoint to spark discussion. Thorax 2020;75:1035-9.ArticlePubMed
- 13. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: 2019 report [Internet]. Global Initiative for Chronic Obstructive Lung. Disease; 2019 [cited 2021 Jan 14]. Available from: https://goldcopd.org/wp-content/uploads/2018/11/GOLD-2019-v1.7-FINAL-14Nov2018-WMS.pdf.
- 14. Gonçalves G, Saeed H, Abdelrahim ME, Harb HS, Madney YM, Eng K, et al. Non-invasive ventilation in patients with an altered level of consciousness: a clinical review and practical insights. Adv Respir Med 2020;88:233-44.ArticlePubMed
Citations
Citations to this article as recorded by

- Lipid Emulsion-Mediated Improvement of Hemodynamic Depression Caused by Amlodipine Toxicity
Ju-Tae Sohn
Pediatric Emergency Care.2023; 39(3): 205. CrossRef
 , Habib Md Reazaul Karim
, Habib Md Reazaul Karim , Manu P Kesavankutty
, Manu P Kesavankutty , Porika Prashanth Nayak
, Porika Prashanth Nayak




 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite