Articles
- Page Path
- HOME > Acute Crit Care > Volume 33(3); 2018 > Article
- Original Article Timing and Associated Factors for Sepsis-3 in Severe Trauma Patients: A 3-Year Single Trauma Center Experience
-
Seungwoo Chung
 , Donghwan Choi
, Donghwan Choi , Jayun Cho, Yo Huh
, Jayun Cho, Yo Huh , Jonghwan Moon
, Jonghwan Moon , Junsik Kwon
, Junsik Kwon , Kyoungwon Jung
, Kyoungwon Jung , John-Cook Jong Lee
, John-Cook Jong Lee , Byung Hee Kang
, Byung Hee Kang
-
Acute and Critical Care 2018;33(3):130-134.
DOI: https://doi.org/10.4266/acc.2018.00122
Published online: August 31, 2018
Department of Surgery, Ajou University School of Medicine, Suwon, Korea
- Corresponding author Byung Hee Kang Department of Surgery, Ajou University School of Medicine, 206 World cup-ro, Yeongtong-gu, Suwon 16499, Korea Tel: +82-31-219-7764 Fax: +82-31-219-7781 E-mail: kbhname@gmail.com
Copyright © 2018 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- We hypothesized that the recent change of sepsis definition by sepsis-3 would facilitate the measurement of timing of sepsis for trauma patients presenting with initial systemic inflammatory response syndrome. Moreover, we investigated factors associated with sepsis according to the sepsis-3 definition.
-
Methods
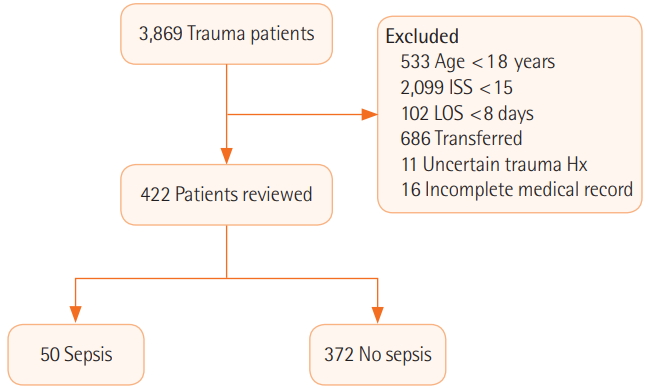
- Trauma patients in a single level I trauma center were retrospectively reviewed from January 2014 to December 2016. Exclusion criteria were younger than 18 years, Injury Severity Score (ISS) <15, length of stay <8 days, transferred from other hospitals, uncertain trauma history, and incomplete medical records. A binary logistic regression test was used to identify the risk factors for sepsis-3.
-
Results
- A total of 3,869 patients were considered and, after a process of exclusion, 422 patients were reviewed. Fifty patients (11.85%) were diagnosed with sepsis. The sepsis group presented with higher mortality (14 [28.0%] vs. 17 [4.6%], P<0.001) and longer intensive care unit stay (23 days [range, 11 to 35 days] vs. 3 days [range, 1 to 9 days], P<0.001). Multivariate analysis demonstrated that, in men, high lactate level and red blood cell transfusion within 24 hours were risk factors for sepsis. The median timing of sepsis-3 was at 8 hospital days and 4 postoperative days. The most common focus was the respiratory system.
-
Conclusions
- Sepsis defined by sepsis-3 remains a critical issue in severe trauma patients. Male patients with higher ISS, lactate level, and red blood cell transfusion should be cared for with caution. Reassessment of sepsis should be considered at day 8 of hospital stay or day 4 postoperatively.
INTRODUCTION
MATERIALS AND METHODS
RESULTS
DISCUSSION
KEY MESSAGES
Values are presented as median (interquartile range) or number (%). BP: blood pressure; ISS: Injury Severity Score; RTS: Revised Trauma Score; GCS: Glasgow Coma Scale; SIRS: systemic inflammatory response syndrome; RBC: red blood cell; FFP: fresh frozen plasma; ER: emergency room; SOFA: Sequential Organ Failure Assessment; ICU: intensive care unit; LOS: length of stay.
| Variable | Odds ratio (95% CI) | P-value |
|---|---|---|
| Male sex | 3.254 (1.009-10.493) | 0.048 |
| Lactate | 1.181 (1.028-1.357) | 0.019 |
| ISS | 1.062 (1.024-1.102) | 0.001 |
| RBC transfusion | 1.061 (1.028-1.094) | <0.001 |
- 1. Sauaia A, Moore FA, Moore EE, Moser KS, Brennan R, Read RA, et al. Epidemiology of trauma deaths: a reassessment. J Trauma 1995;38:185-93.ArticlePubMed
- 2. Kisat M, Villegas CV, Onguti S, Zafar SN, Latif A, Efron DT, et al. Predictors of sepsis in moderately severely injured patients: an analysis of the National Trauma Data Bank. Surg Infect (Larchmt) 2013;14:62-8.ArticlePubMedPMC
- 3. Osborn TM, Tracy JK, Dunne JR, Pasquale M, Napolitano LM. Epidemiology of sepsis in patients with traumatic injury. Crit Care Med 2004;32:2234-40.ArticlePubMed
- 4. Wafaisade A, Lefering R, Bouillon B, Sakka SG, Thamm OC, Paffrath T, et al. Epidemiology and risk factors of sepsis after multiple trauma: an analysis of 29,829 patients from the Trauma Registry of the German Society for Trauma Surgery. Crit Care Med 2011;39:621-8.ArticlePubMed
- 5. Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016;315:801-10.ArticlePubMedPMC
- 6. Vincent JL, Moreno R, Takala J, Willatts S, De Mendonça A, Bruining H, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 1996;22:707-10.ArticlePubMed
- 7. Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, et al. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med 2003;31:1250-6.ArticlePubMed
- 8. Park JH, Choi SH, Yoon YH, Park SJ, Kim JY, Cho HJ. Risk factors for sepsis in Korean trauma patients. Eur J Trauma Emerg Surg 2016;42:453-8.ArticlePubMedPDF
- 9. Donnelly JP, Safford MM, Shapiro NI, Baddley JW, Wang HE. Application of the Third International Consensus Definitions for Sepsis (Sepsis-3) Classification: a retrospective population-based cohort study. Lancet Infect Dis 2017;17:661-70.ArticlePubMedPMC
- 10. Shankar-Hari M, Harrison DA, Rubenfeld GD, Rowan K. Epidemiology of sepsis and septic shock in critical care units: comparison between sepsis-2 and sepsis-3 populations using a national critical care database. Br J Anaesth 2017;119:626-36.ArticlePubMedPDF
- 11. Besen BA, Romano TG, Nassar AP Jr, Taniguchi LU, Azevedo LC, Mendes PV, et al. Sepsis-3 definitions predict ICU mortality in a low-middle-income country. Ann Intensive Care 2016;6:107. ArticlePubMedPMCPDF
- 12. Lenz A, Franklin GA, Cheadle WG. Systemic inflammation after trauma. Injury 2007;38:1336-45.ArticlePubMed
- 13. Tschoeke SK, Ertel W. Immunoparalysis after multiple trauma. Injury 2007;38:1346-57.ArticlePubMed
- 14. Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med 2001;29:1303-10.ArticlePubMed
- 15. Martin GS, Mannino DM, Eaton S, Moss M. The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med 2003;348:1546-54.ArticlePubMed
- 16. Nair GB, Niederman MS. Year in review 2013. Critical care: respiratory infections. Crit Care 2014;18:572. ArticlePubMedPMCPDF
- 17. Quenot JP, Binquet C, Kara F, Martinet O, Ganster F, Navellou JC, et al. The epidemiology of septic shock in French intensive care units: the prospective multicenter cohort EPISS study. Crit Care 2013;17:R65. ArticlePubMedPMC
- 18. Rubano JA, Paccione MF, Rutigliano DN, Vosswinkel JA, Mc-Cormack JE, Huang EC, et al. Outcomes following prolonged mechanical ventilation: analysis of a countywide trauma registry. J Trauma Acute Care Surg 2015;78:289-94.ArticlePubMed
References
Figure & Data
References
Citations

- A biomarker panel of C-reactive protein, procalcitonin and serum amyloid A is a predictor of sepsis in severe trauma patients
Mei Li, Yan-jun Qin, Xin-liang Zhang, Chun-hua Zhang, Rui-juan Ci, Wei Chen, De-zheng Hu, Shi-min Dong
Scientific Reports.2024;[Epub] CrossRef - Identifying biomarkers deciphering sepsis from trauma-induced sterile inflammation and trauma-induced sepsis
Praveen Papareddy, Michael Selle, Nicolas Partouche, Vincent Legros, Benjamin Rieu, Jon Olinder, Cecilia Ryden, Eva Bartakova, Michal Holub, Klaus Jung, Julien Pottecher, Heiko Herwald
Frontiers in Immunology.2024;[Epub] CrossRef - The Road to Sepsis in Geriatric Polytrauma Patients—Can We Forecast Sepsis in Trauma Patients?
Cédric Niggli, Philipp Vetter, Jan Hambrecht, Hans-Christoph Pape, Ladislav Mica
Journal of Clinical Medicine.2024; 13(6): 1570. CrossRef - Defining Posttraumatic Sepsis for Population-Level Research
Katherine Stern, Qian Qiu, Michael Weykamp, Grant O’Keefe, Scott C. Brakenridge
JAMA Network Open.2023; 6(1): e2251445. CrossRef - Strategies for the treatment of femoral fractures in severely injured patients: trends in over two decades from the TraumaRegister DGU®
Felix M. Bläsius, Markus Laubach, Hagen Andruszkow, Philipp Lichte, Hans-Christoph Pape, Rolf Lefering, Klemens Horst, Frank Hildebrand
European Journal of Trauma and Emergency Surgery.2022; 48(3): 1769. CrossRef - Infectious Diseases-Related Emergency Department Visits Among Non-Elderly Adults with Intellectual and Developmental Disabilities in the United States: Results from the National Emergency Department Sample, 2016
Hussaini Zandam, Monika Mitra, Ilhom Akobirshoev, Frank S. Li, Ari Ne'eman
Population Health Management.2022; 25(3): 335. CrossRef - Patient, provider, and system factors that contribute to health care–associated infection and sepsis development in patients after a traumatic injury: An integrative review
Debbie Tan, Taneal Wiseman, Vasiliki Betihavas, Kaye Rolls
Australian Critical Care.2021; 34(3): 269. CrossRef - Accuracy of Procalcitonin Levels for Diagnosis of Culture-Positive Sepsis in Critically Ill Trauma Patients: A Retrospective Analysis
Aisha Bakhtiar, Syed Jawad Haider Kazmi, Muhammad Sohaib Asghar, Muhammad Nadeem Khurshaidi, Salman Mazhar, Noman A Khan, Nisar Ahmed, Farah Yasmin, Rabail Yaseen, Maira Hassan
Cureus.2021;[Epub] CrossRef - An Evaluation of the Effect of Performance Improvement and Patient Safety Program Implemented in a New Regional Trauma Center of Korea
Yo Huh, Junsik Kwon, Jonghwan Moon, Byung Hee Kang, Sora Kim, Jayoung Yoo, Seoyoung Song, Kyoungwon Jung
Journal of Korean Medical Science.2021;[Epub] CrossRef - The impact of infection complications after trauma differs according to trauma severity
Akira Komori, Hiroki Iriyama, Takako Kainoh, Makoto Aoki, Toshio Naito, Toshikazu Abe
Scientific Reports.2021;[Epub] CrossRef - Gene Expression–Based Diagnosis of Infections in Critically Ill Patients—Prospective Validation of the SepsisMetaScore in a Longitudinal Severe Trauma Cohort
Simone Thair, Caspar Mewes, José Hinz, Ingo Bergmann, Benedikt Büttner, Stephan Sehmisch, Konrad Meissner, Michael Quintel, Timothy E. Sweeney, Purvesh Khatri, Ashham Mansur
Critical Care Medicine.2021; 49(8): e751. CrossRef - Immunometabolic signatures predict risk of progression to sepsis in COVID-19
Ana Sofía Herrera-Van Oostdam, Julio E. Castañeda-Delgado, Juan José Oropeza-Valdez, Juan Carlos Borrego, Joel Monárrez-Espino, Jiamin Zheng, Rupasri Mandal, Lun Zhang, Elizabeth Soto-Guzmán, Julio César Fernández-Ruiz, Fátima Ochoa-González, Flor M. Trej
PLOS ONE.2021; 16(8): e0256784. CrossRef - Sepsis in Trauma: A Deadly Complication
Fernanda Mas-Celis, Jimena Olea-López, Javier Alberto Parroquin-Maldonado
Archives of Medical Research.2021; 52(8): 808. CrossRef - New automated analysis to monitor neutrophil function point-of-care in the intensive care unit after trauma
Lillian Hesselink, Roy Spijkerman, Emma de Fraiture, Suzanne Bongers, Karlijn J. P. Van Wessem, Nienke Vrisekoop, Leo Koenderman, Luke P. H. Leenen, Falco Hietbrink
Intensive Care Medicine Experimental.2020;[Epub] CrossRef

 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite


