Abstract
-
Background
-
Stenotrophomonas maltophilia has been increasingly recognized as an opportunistic pathogen associated with high morbidity and mortality. Data on the prognostic factors associated with S. maltophilia pneumonia in patients admitted to intensive care unit (ICU) are lacking.
-
Methods
- We conducted a retrospective analysis of data from 117 patients with S. maltophilia pneumonia admitted to the ICUs of two tertiary referral hospitals in South Korea between January 2011 and December 2022. To assess risk factors associated with in-hospital mortality, multivariable logistic regression analyses were performed.
-
Results
- The median age of the study population was 71 years. Ventilator-associated pneumonia was 76.1% of cases, and the median length of ICU stay before the first isolation of S. maltophilia was 15 days. The overall in-hospital mortality rate was 82.1%, and factors independently associated with mortality were age (odds ratio [OR], 1.05; 95% confidence interval [CI], 1.00–1.09; P=0.046), Sequential Organ Failure Assessment (SOFA) score (OR, 1.21; 95%; CI, 1.02–1.43; P=0.025), corticosteroid use (OR, 4.19; 95% CI, 1.26–13.91; P=0.019), and polymicrobial infection (OR, 0.22; P=0.009). However, the impact of appropriate antibiotic therapy on mortality was insignificant. In a subgroup of patients who received appropriate antibiotic therapy (n=58), antibiotic treatment modality-related variables, including combination or empirical therapy, also showed no significant association with survival.
-
Conclusions
- Patients with S. maltophilia pneumonia in ICU have high mortality rates. Older age, higher SOFA score, and corticosteroid use were independently associated with increased in-hospital mortality, whereas polymicrobial infection was associated with lower mortality. The effect of appropriate antibiotic therapy on prognosis was insignificant.
-
Keywords: anti-bacterial agents; intensive care units; pneumonia; risk factors; Stenotrophomonas maltophilia
INTRODUCTION
Stenotrophomonas maltophilia is a Gram-negative multidrug-resistant bacterium that has emerged as a significant opportunistic pathogen of global concern [1]. S. maltophilia infections primarily present as a respiratory tract infection, with significant morbidity in vulnerable patients, including immunocompromised and debilitated patients [2]. Varying degrees of mortality have been reported in patients with S. maltophilia pneumonia, with rates reaching as high as 70% [2]. Owing to intrinsic resistance to various antibiotics, limitations in antimicrobial susceptibility testing (AST), and lack of clinical data, the treatment of S. maltophilia infection is a significant challenge for clinicians [3].
Factors commonly encountered in patients admitted to intensive care unit (ICU), such as severe illness, invasive procedures, and exposure to broad-spectrum antibiotics, have been reported to be associated with ICU-acquired S. maltophilia pneumonia, which is a significant risk factor for ICU death [4]. Previous studies have shown that the frequency of S. maltophilia pneumonia in ICU tends to increase over time [4]; moreover, in a recent study on patients with pneumonia in ICUs of medical centers in the United States, S. maltophilia was one of the top six commonly identified pathogens [5]. Additionally, there has been an increase in resistance rates to drugs that have classically shown good susceptibility [3]. Despite this emerging clinical relevance, S. maltophilia has been considerably less studied than other Gram-negative bacteria [3], and clinical data on S. maltophilia pneumonia in the ICU setting are lacking.
In the present study, we aimed to retrospectively analyze patients with S. maltophilia pneumonia in the ICU to describe their clinical features and investigate factors, including antibiotic therapy, associated with mortality.
MATERIALS AND METHODS
This study was approved by the Institutional Review Board of each institution, which waived the requirement for informed consent owing to the retrospective nature of the study.
Study Design and Participants
This retrospective study was conducted at two tertiary referral centers, the Kyungpook National University Hospital and the Kyungpook National University Chilgok Hospital, in Daegu, Korea. Information on consecutive adult patients admitted to the ICUs, including both medical and surgical ICUs, at two hospitals between January 1, 2011, and December 31, 2022, was collected, and patients with S. maltophilia cultured from respiratory specimens during their ICU stay were selected. The exclusion criteria included patients with no pneumonia events throughout their stay in the ICU, those who died or were discharged on the day of specimen collection, those with inadequate specimens, and those with S. maltophilia colonization (Figure 1). Clinical features were compared by dividing patients into two groups based on the occurrence of in-hospital death, which is the outcome of interest in the current study.
Definitions
S. maltophilia pneumonia was diagnosed when there was a positive microbiologic culture from a respiratory specimen and concurrent clinical and radiological signs consistent with pneumonia, defined as new or progressive lung infiltrates on chest radiograph plus at least two of the following clinical criteria: (1) body temperature of >38.0 °C or <36.0 °C, (2) white blood cell count of ≥12,000/mm3 or ≤4,000/mm3, and (3) macroscopically purulent tracheal aspirate or sputum, referring to some of the criteria proposed by U.S. Centers for Disease Control and Prevention [6]. Cases with a positive respiratory sample but not meeting the abovementioned pneumonia criteria were considered as S. maltophilia colonization and excluded from the final analysis. Ventilator-associated pneumonia (VAP) was defined as pneumonia in patients who had received at least 48 hours of mechanical ventilation [7]. The respiratory specimen was considered to be adequate with the following threshold: (1) quantitative culture of bronchoalveolar lavage (≥104 colony-forming unit [CFU]/mL), (2) quantitative culture of bronchoscopic or endotracheal aspirates (≥105 CFU/mL), (3) semi-quantitative culture of endotracheal aspirates (moderate or higher), or (4) sputum with <10 epithelial cells in a low power field.
Immunosuppression was defined as patients with at least one of the following: neutropenia (absolute neutrophil count or total white blood cell count of <500/mm3), hematologic malignancy or human immunodeficiency virus positive with CD4 count of <200, a history of splenectomy, solid organ or hematopoietic stem cell transplant, cytotoxic chemotherapy, immunosuppressant, or receiving daily corticosteroid therapy with a dose of ≥20 mg of prednisone or equivalent for ≥14 days) [6,8]. Polymicrobial infection was defined as the detection of other bacteria at the same time as the first isolation of S. maltophilia or within 1 week thereafter. Corticosteroid administration was referred to as at least one dose of ≥10 mg of prednisone or equivalent within the same time frame (within 1 week of index culture collection).
Appropriate antibiotic therapy was defined as a case where at least one antibiotic showing sensitivity in the AST was administered for 48 hours or longer, and simultaneous administration of two or more antibiotics was referred to as combination therapy. Among patients who received appropriate antibiotic therapy, those who started treatment before S. maltophilia was first identified were defined as empiric therapy, and the remaining cases were defined as definitive therapy.
Data Collection
Patients’ baseline characteristics, including demographics, comorbid conditions, and laboratory test results, were obtained from electronic medical records. Prior antibiotic exposure within the last 90 days was investigated, and variables indicative of severity at the time of admission to ICU, including Acute Physiological and Chronic Health Evaluation (APACHE) II and Sequential Organ Failure Assessment (SOFA) scores, were reviewed. The use of vasopressors prior to S. maltophilia infection was recorded.
To evaluate the severity of illness at the beginning of S. maltophilia infection, SOFA scores on the day of index culture collection were examined. For several drugs, AST results of the identified strains were recorded. Information on the presence of polymicrobial infection and isolation of S. maltophilia from normally sterile sites, including blood, abdominal fluid, pleural fluid, and cerebrospinal fluid, was collected. Moreover, the frequency of corticosteroid administration was recorded.
In addition to appropriate antibiotic therapy, the following variables related to treatment were investigated: monotherapy; combination therapy; use of trimethoprim-sulfamethoxazole (TMP-SMX), levofloxacin, and minocycline; empiric therapy; definitive therapy; and time interval from S. maltophilia diagnosis to initiation of appropriate antibiotics.
Statistical Analysis
Continuous variables were expressed as medians (interquartile range [IQR]), whereas categorical variables were expressed as numbers (percentage). Comparisons between two groups were performed using Mann-Whitney U-tests for continuous variables and chi-square or Fisher’s exact tests for categorical variables. To investigate the independent factors associated with in-hospital death, a multivariable analysis was performed using a logistic regression model. Variables showing statistically significant differences between the groups in univariate analysis or those considered clinically significant were subjected to multivariable analysis, and the backward elimination method was used to define the independent risk factors. Statistical analysis was performed using RStudio (R version 4.2.1). Two-sided P-value of <0.05 was considered to indicate statistically significant.
RESULTS
Initially, 336 patients with a positive S. maltophilia respiratory culture during their ICU stay were identified. Among them, those with no pneumonia events throughout their ICU stay (n=39), those who died or were discharged on the day of culture collection (n=11), those with inadequate specimens (n=84), and those with S. maltophilia colonization (n=85) were excluded. Subsequently, the data of 117 patients were analyzed. S. maltophilia was cultured from endotracheal aspirate (n=107), bronchoalveolar lavage (n=5), and sputum (n=5), with 4 cases additionally displaying concurrent bacteremia. The overall in-hospital mortality rate was 82.1% (n=96). The median duration of mechanical ventilation for all patients was 22 days, with no significant difference between the in-hospital survivors and nonsurvivors (median [IQR], 28 days [12–40] vs. 20 days [9–35]; P=0.146).
Baseline Characteristics
The baseline and clinical characteristics of patients are presented in Table 1. The median age of the study population was 71 years, which was significantly lower in survivors than in nonsurvivors (median [IQR], 64 years [55–72] vs. 72 years [65–80]; P=0.027). Comorbid conditions and prior antibiotic exposure were comparable between the two groups. The median APACHE II and SOFA scores of all patients at ICU admission were 17 and 8, respectively, with no significant difference between the two groups. Initial laboratory test results were similar between the two groups, except for the levels of lactic acid (1.4 mmol/L [1.2–3.1] vs. 3.0 mmol/L [1.7–6.0]; P=0.033) (Table 2).
Variables Related to S. maltophilia Infection and Treatment Details
Variables related to S. maltophilia infection and its treatment details are displayed in Table 3. VAP accounted for 76.1% (n=89) of all patients, and the median length of ICU stay before the first isolation of S. maltophilia was 15 days, with no significant difference between the groups. Additionally, SOFA scores on the day of the first S. maltophilia culture collection were comparable between the groups. Survivors had a significantly higher frequency of polymicrobial infection than nonsurvivors (n=15 [71.4%] vs. n=32 [33.3%]; P=0.003).
Among patients with corticosteroid administration, the number of survivors was significantly lower than that of nonsurvivors (n=5 [23.8%] vs. n=50 [52.1%]; P=0.035). No significant differences were observed between the two groups in terms of antibiotic treatment modality-related variables and appropriate antibiotic therapy.
Antibiotic Susceptibility
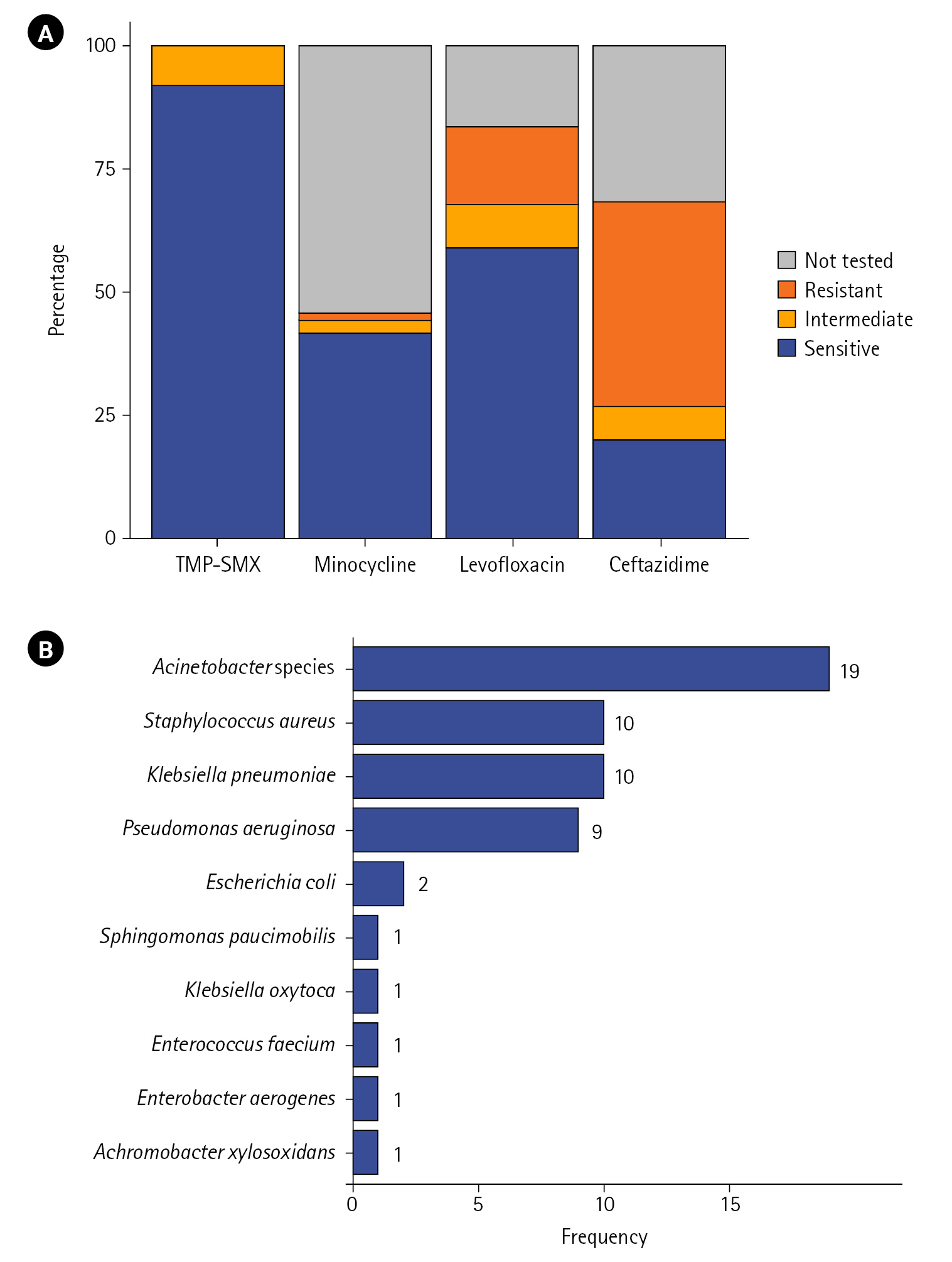
For the four main drugs, AST results of 115 patients are depicted in Figure 2A. The proportion of cases susceptible to TMP–SMX was the highest at 92.2%, followed by that of cases susceptible to levofloxacin (59.1%), minocycline (41.7%), and ceftazidime (20%). The proportion of cases showing resistant or intermediate results were for ceftazidime and levofloxacin was 48.7% and 24.4%, respectively, which tended to be higher than that of cases showing resistant or intermediate results for TMP–SMX (7.8%) and minocycline (4.3%).
Polymicrobial Infections
In 40.2% (n=47) of patients, other microorganisms were identified at the time of S. maltophilia isolation or within the first week of isolation. The distribution of coisolates is depicted in Figure 2B. Acinetobacter species showed the highest frequency (n=19), followed by Staphylococcus aureus (n=10), Klebsiella pneumoniae (n=10), and Pseudomonas aeruginosa (n=9). Escherichia coli was isolated in two cases, and Sphingomonas paucimobilis, Klebsiella oxytoca, Enterococcus faecium, Enterobacter aerogenes, and Achromobacter xylosoxidans were isolated in one case each.
Risk Factors Associated with In-Hospital Mortality
The following factors were included in the multivariable analysis: age, APACHE II, vasopressor use, SOFA score on the day of index culture collection, polymicrobial infection, corticosteroid use, and appropriate antibiotic therapy. Consequently, age (odds ratio [OR], 1.05; 95% confidence interval [CI], 1.00–1.09; P=0.046), SOFA score (OR, 1.21; 95%; CI, 1.02–1.43; P=0.025), corticosteroid use (OR, 4.19; 95% CI, 1.26–13.91; P=0.019) and polymicrobial infection (OR, 0.22; 95% CI, 0.07–0.69; P=0.009) were independent factors associated with in-hospital death, whereas appropriate antibiotic therapy did not modify mortality (Table 4).
A multivariable analysis performed in a subgroup of patients who received appropriate antibiotic therapy (n=58) showed that vasopressor use (P=0.044), polymicrobial infection (P=0.025), and corticosteroid use (P=0.046) were associated with in-hospital mortality. However, additionally included antibiotic treatment modality-related variables such as combination, empirical treatment, time interval from S. maltophilia diagnosis to initiation of appropriate antibiotics, and use of specific antibiotics, had no significant effect on survival (Supplementary Table 1).
In addition, a subgroup analysis was conducted for patients receiving corticosteroids (n=55). At the start of corticosteroid treatment, methylprednisolone was the most frequently prescribed, accounting for over half (56.4%) of cases, and it was administered at a median daily dosage of 0.9 mg/kg. Septic shock was the most common reason for corticosteroid use, followed by acute respiratory distress syndrome and severe pneumonia (Supplementary Table 2). The same multivariable analysis was performed including the aforementioned factors in this subgroup, but no statistically significant prognostic factors were found (Supplementary Table 3).
DISCUSSION
In this cohort of 117 critically ill patients with S. maltophilia pneumonia, the in-hospital mortality rate was approximately 82%. Factors that were independently associated with in-hospital death were age, SOFA score, corticosteroid use, and polymicrobial infection. However, the effects of appropriate antibiotic therapy, as well as empiric or combination therapy, on mortality were not significant.
Previous studies on critically ill patients with S. maltophilia pneumonia have reported hospital or ICU mortality rates of approximately 50%–70% [9-13], which are mostly lower than those of our study patients. Although the high mortality rate in the present study cannot be clearly explained by our data alone, it may be attributed to the fact that our patients had a median age of 71 years, surpassing the median age range of 61 to 65 years in previous studies [9,11-13]. While not identified as a prognostic factor in our study, the comparatively lower proportion of patients (49.6%) receiving appropriate antibiotic therapy, in contrast to the approximately 60%–73% range reported in prior studies [9-11,13], could have also exerted an influence. Furthermore, the outcomes of the current study might have been adversely influenced by the incorporation of patients with S. maltophilia pneumonia, using comparatively stringent criteria that eliminated colonization cases. This is in contrast to earlier studies which encompassed not only S. maltophilia infections but also instances of colonization [10,12].
As a general treatment approach for S. maltophilia infections, antibiotics including TMP–SMX, minocycline, and levofloxacin, are recommended as monotherapy or combination therapy depending on severity; however, there are limited clinical data to strongly support a specific treatment strategy [14]. In particular, data on the impact of appropriate antibiotic therapy on the prognosis of relatively severe S. maltophilia pneumonia in ICU are lacking, and most studies show conflicting results [9,13,15]. In a study including patients with VAP caused by S. maltophilia, Ibn Saied et al. [13 [13], which is similar to our study finding, whereas other studies involving critically ill patients with S. maltophilia pneumonia showed a significant association between appropriate antibiotic therapy and reduced mortality [9,15]. Regarding antibiotic treatment modalities, neither empiric nor combination therapy had a significant effect on survival in the current study, which is consistent with several recent studies [9,11,13].
In agreement with previous studies [9,11,12], compared with other antibiotics, S. maltophilia strains in our study showed the highest percentage of susceptibility in TMP–SMX. Moreover, the study by Puech et al. proposed TMP-SMX as the only agent with a survival benefit in patients with VAP caused by S. maltophilia. However, our study did not identify a benefit in terms of clinical outcomes for any particular antibiotic. Since there are data showing favorable outcomes related to adequate antibiotics in critically ill patients with S. maltophilia pneumonia [9,15], the need for appropriate antibiotic therapy cannot be denied in clinical practice; however, our findings suggest a more conservative approach to antibiotic treatment strategy rather than preemptive administration of a specific antibiotic before confirming AST results or the combination of two or more drugs.
In our study, a higher SOFA score was one of the predictors of mortality. This finding is consistent with previous data, wherein the SOFA score on the day of pneumonia onset or S. maltophilia isolation was independently associated with mortality of S. maltophilia infection in the ICU setting [9,11,12]. These results, combined with the fact that antibiotic treatment targeting S. maltophilia did not modify the survival of our patients, may support the idea that S. maltophilia isolation represents a precarious underlying condition with a poor prognosis, rather than being highly virulent per se [11,16].
Corticosteroids were administered at a significantly higher rate to nonsurvivors compared to survivors in the present study. The effect of corticosteroids has not been reported in previous studies of critically ill patients with S. maltophilia pneumonia. Corticosteroid therapy could be considered an adjunctive treatment option in some severe cases for various indications [17-19]. However, the benefits and harms of corticosteroids seem to be inconsistent depending on the specific causative agent of pneumonia [20-22]. Based on our findings, we suggest that corticosteroids be used cautiously and only when essential in patients in the ICU with suspected or diagnosed S. maltophilia pneumonia. However, notably, this retrospective study may have a bias, including a higher use of corticosteroids in more severe cases such as shock and respiratory failure. Therefore, the significance of corticosteroid use in our findings requires caution in interpretation, and future well-designed prospective studies on this topic are warranted.
In our study, polymicrobial infection was another prognostic factor, associated with reduced in-hospital mortality. The reported proportion of polymicrobial infection in cohorts of critically ill patients with S. maltophilia is slightly higher than ours, ranging from 45% to 58% [9,11,12]. Its prognostic relevance seems to be inconsistent with our results, with some studies showing no significant difference between mono and polymicrobial infection in terms of mortality [9,11] and one study presenting a worse prognosis in patients with polymicrobial infection [12]. Given the heterogeneity in the type and distribution of coisolates in those studies [9,11,12] and the potential interactions between S. maltophilia and certain strains [23,24], clearly elucidating the significance of polymicrobial infection as a prognostic factor in critically ill patients with S. maltophilia based on the current small-scale study is difficult. Conversely, a recent study on multidrug-resistant Acinetobacter baumannii VAP reported a similar result to ours, which showed a lower mortality rate in the case of polymicrobial infection than that in monomicrobial, and the attenuation of virulence due to competition with coexisting pathogens in a polymicrobial setting was suggested as a possible explanation [25].
Taken together, our results suggest that when S. maltophilia pneumonia is suspected in an ICU environment, high-risk patients, including those with older age and high severity scores, should be carefully screened and reevaluated for excessive use of antibiotics or corticosteroids. Furthermore, considering the high mortality rate of such patients and unknown efficacy of appropriate antibiotic therapy, various measures for preventing S. maltophilia infection and transmission, including barrier precautions during patient care, antibiotic stewardship, and appropriate maintenance of hospital environment and medical equipment [2], should be meticulously implemented.
The present study has several limitations. Considering the retrospective nature of this study, there may have been biases that were not identified. First of all, it should be considered that not all of the S. maltophilia pneumonia defined in our study may represent true infections. Approximately 40% of cases in our study had polymicrobial infections, with coisolates varying in species and frequency. Even after adjustment for polymicrobial infection, detailed information on antibiotics susceptibility testing or targeted antibiotic therapy for coisolates was not obtained. Furthermore, in such cases with polymicrobial infections, it is difficult to determine which microorganism is the actual causative agent of pneumonia. This may have influenced our results, where appropriate antibiotic therapy was not associated with mortality. Second, the presence of acute illness in organs other than the lungs during ICU stay was not considered in our analysis. Third, although steroid treatment was a prognostic factor in this study, we evaluated steroid use in a relatively narrow window of time: within the first week of index culture collection. Corticosteroids administered during other periods may also have affected clinical outcomes. Another limitation is that this study was conducted with a limited number of patients at institutions located in a single region. In addition, the smaller size of the subgroups may have compromised the reliability of our results.
In conclusion, patients with S. maltophilia pneumonia in ICU settings have high mortality rates. Older age, higher SOFA score, and corticosteroid use were independently associated with increased in-hospital mortality, whereas polymicrobial infection was associated with lower mortality. The effect of appropriate antibiotic therapy on the prognosis was not significant.
KEY MESSAGES
▪ In critically ill patients with Stenotrophomonas maltophilia pneumonia, older age, higher Sequential Organ Failure Assessment (SOFA) score, and corticosteroid use were associated with increased mortality, whereas polymicrobial infection was associated with better prognosis.
▪ The effect of appropriate antibiotic therapy on mortality was insignificant.
NOTES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conceptualization: YHL, JL. Data curation: BY, SHC, JEP, HS. Formal analysis: WKL, SSY, SYL. Methodology: YHL, SIC, CHK. Writing – original draft: YHL. Writing – review & editing: JL, JYP.
-
FUNDING
None.
SUPPLEMENTARY MATERIALS
Supplementary materials can be found via https://doi.org/10.4266/acc.2023.00682.
Supplementary Table 1.
Variables associated with in-hospital death in patients receiving appropriate antibiotics (n=58) assessed via multivariable logistic regression analysis
acc-2023-00682-Supplementary-Table-1.pdf
Supplementary Table 3.
Variables associated with in-hospital death in patients receiving corticosteroids (n=55) assessed via multivariable logistic regression analysis
acc-2023-00682-Supplementary-Table-3.pdf
Acknowledgments
None.
Figure 1.Flowchart of the study. ICU: intensive care unit.
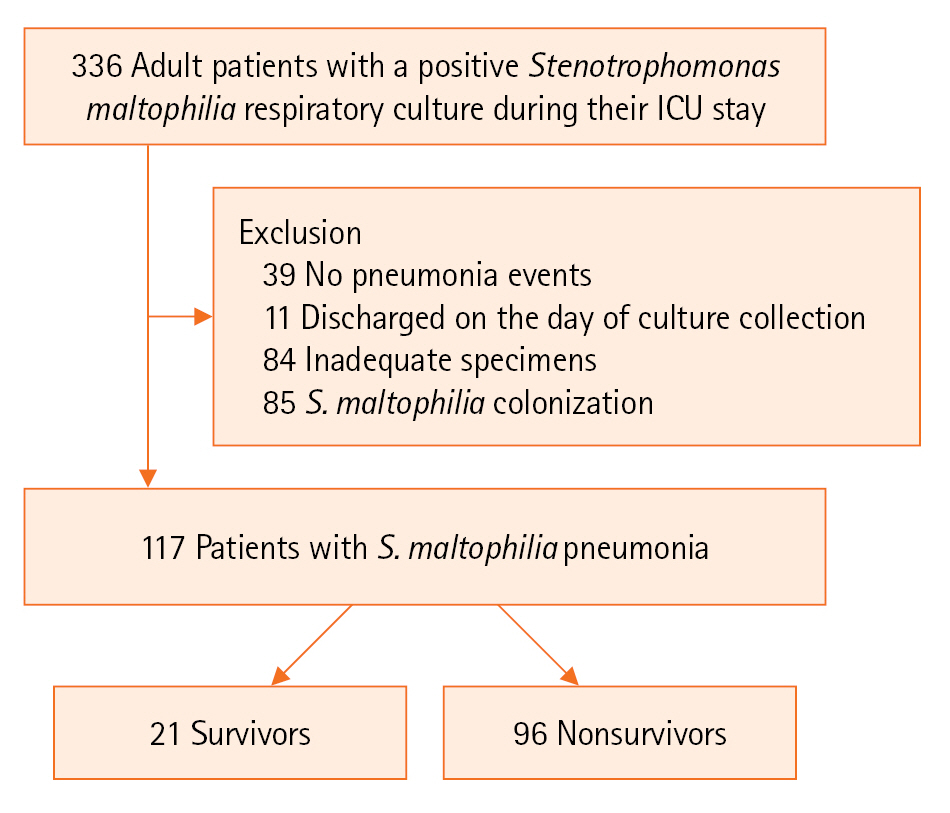
Figure 2.(A) Antibiotic susceptibility of Stenotrophomonas maltophilia strains isolated from the respiratory tract specimen of critically ill patients (n=115). Susceptibility results for each antibiotic are expressed as a percentage of all cases. (B) Frequency distribution of coisolates in patients with polymicrobial infection (n=47). Results are sorted by frequency, with duplicates allowed. TMP–SMX: trimethoprim–sulfamethoxazole.
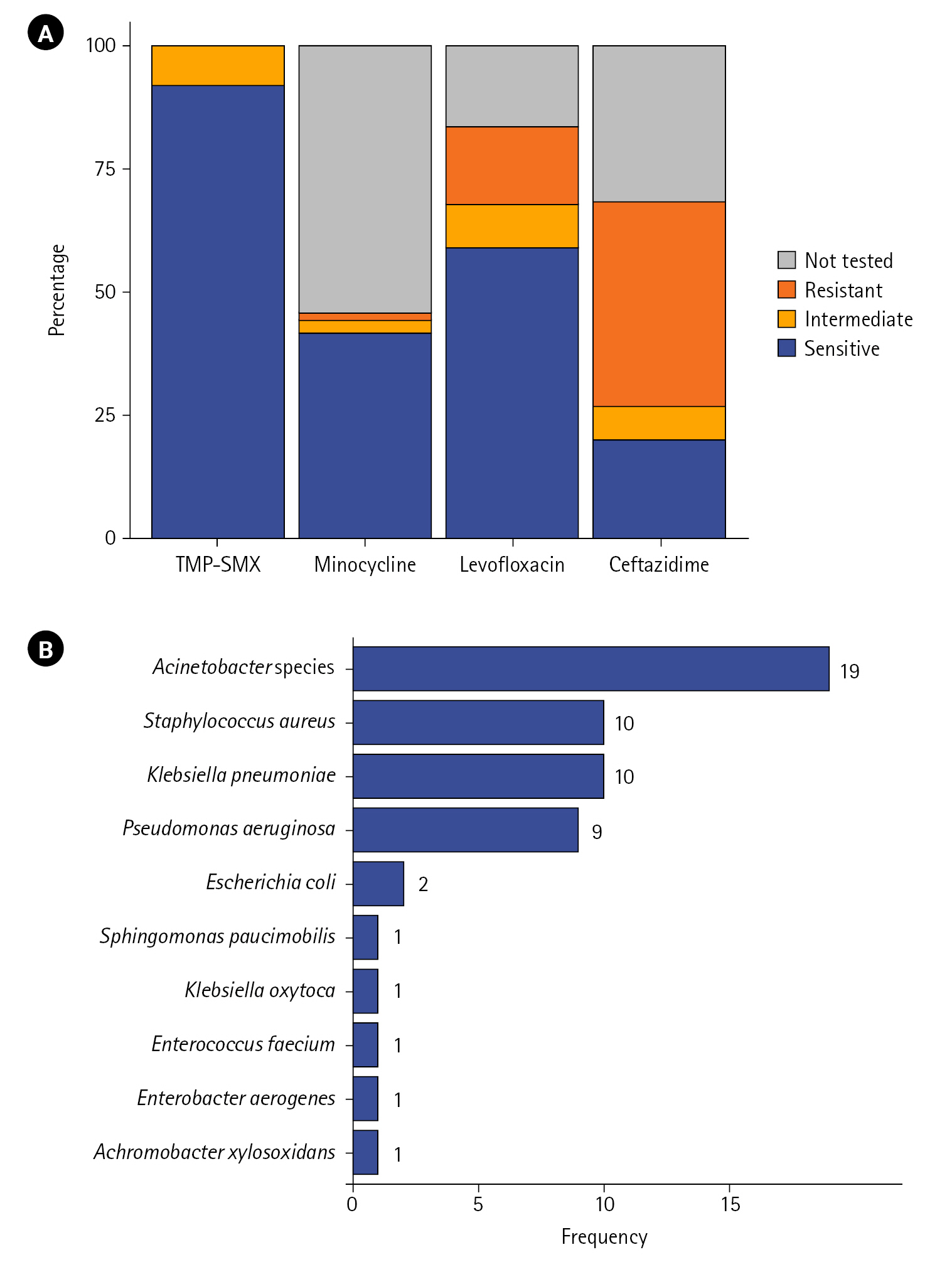
Table 1.Demographic and baseline characteristics
|
Variable |
Total (n=117) |
Survivor (n=21) |
Nonsurvivor (n=96) |
P-value |
|
Age (yr) |
71 (63–78) |
64 (55–72) |
72 (65–80) |
0.027 |
|
Male |
75 (64.1) |
13 (61.9) |
62 (64.6) |
>0.999 |
|
Body mass index (kg/m2) |
22.8 (19.8–25.0) |
22.5 (20.1–24.2) |
22.8 (19.8–25.1) |
0.577 |
|
Comorbid condition |
|
|
|
|
|
Chronic pulmonary disease |
18 (15.4) |
3 (14.3) |
15 (15.6) |
>0.999 |
|
Hypertension |
55 (47.0) |
10 (47.6) |
45 (46.9) |
>0.999 |
|
Diabetes mellitus |
39 (33.3) |
7 (33.3) |
32 (33.3) |
>0.999 |
|
Cardiovascular disease |
22 (18.8) |
6 (28.6) |
16 (16.7) |
0.339 |
|
Cerebrovascular disease |
14 (12.0) |
2 (9.5) |
12 (12.5) |
0.992 |
|
Chronic kidney disease |
20 (17.1) |
2 (9.5) |
18 (18.8) |
0.486 |
|
Chronic liver disease |
2 (1.7) |
0 |
2 (2.1) |
>0.999 |
|
Connective tissue disease |
3 (2.6) |
0 |
3 (3.1) |
0.953 |
|
Cancer |
28 (23.9) |
2 (9.5) |
26 (27.1) |
0.154 |
|
Immunosuppression |
20 (17.1) |
1 (4.8) |
19 (19.8) |
0.181 |
|
Previous antibiotic exposure |
|
|
|
|
|
Antipseudomonal penicillin or cephalosporin |
41 (35.0) |
5 (23.8) |
36 (37.5) |
0.348 |
|
Carbapenem |
34 (29.1) |
4 (19.0) |
30 (31.2) |
0.395 |
|
Aminoglycosides |
5 (4.3) |
2 (9.5) |
3 (3.1) |
0.473 |
|
Fluoroquinolones |
29 (24.8) |
4 (19.0) |
25 (26.0) |
0.694 |
|
Glycopeptides |
28 (23.9) |
3 (14.3) |
25 (26.0) |
0.389 |
|
Reason for ICU admission |
|
|
|
|
|
Medical condition |
106 (90.6) |
19 (90.5) |
87 (90.6) |
>0.999 |
|
Postoperative care |
11 (9.4) |
2 (9.5) |
9 (9.4) |
>0.999 |
|
Severity score on ICU admission |
|
|
|
|
|
APACHE II |
17 (14–22) |
16 (14–20) |
18 (14–23) |
0.362 |
|
SOFA |
8 (5–10) |
7 (4–9) |
8 (5–11) |
0.235 |
|
Vasopressor use |
102 (87.2) |
18 (85.7) |
84 (87.5) |
>0.999 |
Table 2.Initial laboratory findings
|
Variable |
n |
Total |
Survivor (n=21) |
Nonsurvivor (n=96) |
P-value |
|
White blood cell (´103/L) |
117 |
11.5 (7.6–16.3) |
11.2 (6.7–21.0) |
11.6 (7.8–15.6) |
0.607 |
|
Neutrophil–lymphocyte ratio |
116 |
11.9 (5.0–24.2) |
8.7 (5.3–32.9) |
13.4 (4.8–23.9) |
>0.999 |
|
Hematocrit (%) |
117 |
32.1 (28.1–36.9) |
33.0 (29.6–36.4) |
31.8 (27.8–37.0) |
0.486 |
|
C-reactive protein (mg/dl) |
115 |
9.3 (2.8–19.2) |
10.7 (1.4–19.1) |
9.2 (3.7–19.3) |
0.612 |
|
Albumin (g/dl) |
116 |
2.8 (2.5–3.2) |
2.6 (2.1–3.2) |
2.8 (2.5–3.2) |
0.143 |
|
Procalcitonin (mmol/L) |
64 |
0.8 (0.2–6.9) |
0.7 (0.3–6.2) |
0.8 (0.2–7.5) |
0.877 |
|
LDH (U/L) |
63 |
495 (323–750) |
445 (247–518) |
514 (335–886) |
0.105 |
|
Lactic acid (mmol/L) |
65 |
2.6 (1.6–5.5) |
1.4 (1.2–3.1) |
3.0 (1.7–6.0) |
0.033 |
|
D-dimer (μg/ml) |
70 |
7.3 (3.7–19.8) |
7.3 (5.4–10.9) |
7.5 (3.0–20.0) |
>0.999 |
|
Ferritin (ng/ml) |
24 |
839.7 (277.2–1,481.3) |
871.5 (689.7–923.0) |
709.2 (221.2–1,903.0) |
0.970 |
Table 3.Variables regarding Stenotrophomonas maltophilia infection and treatment details
|
Variable |
Total (n=117) |
Survivor (n=21) |
Nonsurvivor (n=96) |
P-value |
|
Ventilator-associated pneumonia |
89 (76.1) |
15 (71.4) |
74 (77.1) |
0.789 |
|
Length of ICU stay before S. maltophilia infection (day) |
15 (10–27) |
16 (10–30) |
15 (10–27) |
0.935 |
|
SOFA score on the day of index culture collection |
7 (5–10) |
7 (4–9) |
7 (5–12) |
0.131 |
|
Polymicrobial infection |
47 (40.2) |
15 (71.4) |
32 (33.3) |
0.003 |
|
S. maltophilia isolation from normally sterile sitesa)
|
10 (8.5) |
1 (4.8) |
9 (9.4) |
0.799 |
|
Corticosteroid use |
55 (57.0) |
5 (23.8) |
50 (52.1) |
0.035 |
|
Appropriate antibiotic therapy |
58 (49.6) |
10 (47.6) |
48 (50.0) |
>0.999 |
|
Monotherapy |
48 (41.0) |
9 (42.9) |
39 (40.6) |
>0.999 |
|
Combination therapy |
10 (8.5) |
1 (4.8) |
9 (9.4) |
0.799 |
|
Empiric therapy |
18 (15.4) |
2 (9.5) |
16 (16.7) |
0.626 |
|
Definitive therapy |
40 (34.2) |
8 (38.1) |
32 (33.3) |
0.871 |
|
Time interval between S. maltophilia isolation and antibiotic treatment (day) |
1 (0–4) |
1 (0–4) |
0 (0–4) |
0.417 |
|
TMP–SMX use |
20 (17.1) |
3 (14.3) |
17 (17.7) |
0.954 |
|
Levofloxacin use |
37 (31.6) |
6 (28.6) |
31 (32.3) |
0.942 |
|
Minocycline use |
18 (15.4) |
2 (9.5) |
16 (16.7) |
0.626 |
Table 4.Variables associated with in-hospital death assessed via multivariable logistic regression analysis
|
Variable |
Univariate analysis
|
Multivariable analysis
|
|
OR |
95% CI |
P-value |
OR |
95% CI |
P-value |
|
Age (yr) |
1.04 |
1.00–1.09 |
0.043 |
1.05 |
1.00–1.09 |
0.046 |
|
APACHE II score |
1.04 |
0.97–1.12 |
0.300 |
|
|
|
|
Vasopressor use |
1.17 |
0.25–4.14 |
0.800 |
|
|
|
|
SOFA score on the day of index culture collection |
1.13 |
0.99–1.30 |
0.082 |
1.21 |
1.02–1.43 |
0.025 |
|
Polymicrobial infection |
0.20 |
0.07–0.54 |
0.002 |
0.22 |
0.07–0.69 |
0.009 |
|
Corticosteroid use |
3.48 |
1.25–11.3 |
0.024 |
4.19 |
1.26–13.91 |
0.019 |
|
Appropriate antibiotic therapy |
1.10 |
0.43–2.87 |
0.800 |
|
|
|
References
- 1. Brooke JS. Advances in the microbiology of Stenotrophomonas maltophilia. Clin Microbiol Rev 2021;34:e0003019.ArticlePubMedPDF
- 2. Looney WJ, Narita M, Mühlemann K. Stenotrophomonas maltophilia: an emerging opportunist human pathogen. Lancet Infect Dis 2009;9:312-23.ArticlePubMed
- 3. Mojica MF, Humphries R, Lipuma JJ, Mathers AJ, Rao GG, Shelburne SA, et al. Clinical challenges treating Stenotrophomonas maltophilia infections: an update. JAC Antimicrob Resist 2022;4:dlac040. ArticlePubMedPMCPDF
- 4. Wang N, Tang C, Wang L. Risk factors for acquired Stenotrophomonas maltophilia pneumonia in intensive care unit: a systematic review and meta-analysis. Front Med (Lausanne) 2022;8:808391. ArticlePubMedPMC
- 5. Sader HS, Castanheira M, Mendes RE, Flamm RK. Frequency and antimicrobial susceptibility of Gram-negative bacteria isolated from patients with pneumonia hospitalized in ICUs of US medical centres (2015-17). J Antimicrob Chemother 2018;73:3053-9.ArticlePubMed
- 6. Centers for Disease Control and Prevention. National Healthcare Safety Network (NHSN) patient safety component manual: Chapter 6. Pneumonia (ventilator-associated [VAP] and non-ventilator-associated pneumonia [PNEU]) event [Internet]. Centers for Disease Control and Prevention. 2023;[cited 2023 May 7]. Available from: https://www.cdc.gov/nhsn/pdfs/pscmanual/pcsmanual_current.pdf.
- 7. Mietto C, Pinciroli R, Patel N, Berra L. Ventilator associated pneumonia: evolving definitions and preventive strategies. Respir Care 2013;58:990-1007.ArticlePubMed
- 8. Rubin LG, Levin MJ, Ljungman P, Davies EG, Avery R, Tomblyn M, et al. 2013 IDSA clinical practice guideline for vaccination of the immunocompromised host. Clin Infect Dis 2014;58:e44-100.ArticlePubMedPDF
- 9. Puech B, Canivet C, Teysseyre L, Miltgen G, Aujoulat T, Caron M, et al. Effect of antibiotic therapy on the prognosis of ventilator-associated pneumonia caused by Stenotrophomonas maltophilia. Ann Intensive Care 2021;11:160. ArticlePubMedPMCPDF
- 10. Nseir S, Di Pompeo C, Brisson H, Dewavrin F, Tissier S, Diarra M, et al. Intensive care unit-acquired Stenotrophomonas maltophilia: incidence, risk factors, and outcome. Crit Care 2006;10:R143. ArticlePubMedPMC
- 11. Guerci P, Bellut H, Mokhtari M, Gaudefroy J, Mongardon N, Charpentier C, et al. Outcomes of Stenotrophomonas maltophilia hospital-acquired pneumonia in intensive care unit: a nationwide retrospective study. Crit Care 2019;23:371. ArticlePubMedPMCPDF
- 12. Saugel B, Eschermann K, Hoffmann R, Hapfelmeier A, Schultheiss C, Phillip V, et al. Stenotrophomonas maltophilia in the respiratory tract of medical intensive care unit patients. Eur J Clin Microbiol Infect Dis 2012;31:1419-28.ArticlePubMedPDF
- 13. Ibn Saied W, Merceron S, Schwebel C, Le Monnier A, Oziel J, Garrouste-Orgeas M, et al. Ventilator-associated pneumonia due to Stenotrophomonas maltophilia: risk factors and outcome. J Infect 2020;80:279-85.ArticlePubMed
- 14. Tamma PD, Aitken SL, Bonomo RA, Mathers AJ, van Duin D, Clancy CJ. Infectious Diseases Society of America guidance on the treatment of AmpC β-lactamase-producing Enterobacterales, carbapenem-resistant Acinetobacter baumannii, and Stenotrophomonas maltophilia infections. Clin Infect Dis 2022;74:2089-114.ArticlePubMedPDF
- 15. Hanes SD, Demirkan K, Tolley E, Boucher BA, Croce MA, Wood GC, et al. Risk factors for late-onset nosocomial pneumonia caused by Stenotrophomonas maltophilia in critically ill trauma patients. Clin Infect Dis 2002;35:228-35.ArticlePubMed
- 16. Pathmanathan A, Waterer GW. Significance of positive Stenotrophomonas maltophilia culture in acute respiratory tract infection. Eur Respir J 2005;25:911-4.ArticlePubMed
- 17. Ceccato A, Russo A, Barbeta E, Oscanoa P, Tiseo G, Gabarrus A, et al. Real-world corticosteroid use in severe pneumonia: a propensity-score-matched study. Crit Care 2021;25:432. ArticlePubMedPMCPDF
- 18. Yoshihiro S, Hongo T, Ohki S, Kaneko T, Ishikawa J, Ihara S, et al. Steroid treatment in patients with acute respiratory distress syndrome: a systematic review and network meta-analysis. J Anesth 2022;36:107-21.ArticlePubMedPDF
- 19. Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med 2021;47:1181-247.PubMedPMC
- 20. Ding L, Huang H, Wang H, He H. Adjunctive corticosteroids may be associated with better outcome for non-HIV Pneumocystis pneumonia with respiratory failure: a systemic review and meta-analysis of observational studies. Ann Intensive Care 2020;10:34. ArticlePubMedPMCPDF
- 21. Moreno G, Rodríguez A, Reyes LF, Gomez J, Sole-Violan J, Díaz E, et al. Corticosteroid treatment in critically ill patients with severe influenza pneumonia: a propensity score matching study. Intensive Care Med 2018;44:1470-82.ArticlePubMedPMCPDF
- 22. RECOVERY Collaborative Group; Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, et al. Dexamethasone in hospitalized patients with Covid-19-preliminary report. N Engl J Med 2021;384:693-704.
- 23. McDaniel MS, Schoeb T, Swords WE. Cooperativity between Stenotrophomonas maltophilia and Pseudomonas aeruginosa during Polymicrobial Airway Infections. Infect Immun 2020;88:e00855-19.ArticlePubMedPMCPDF
- 24. Kataoka D, Fujiwara H, Kawakami T, Tanaka Y, Tanimoto A, Ikawa S, et al. The indirect pathogenicity of Stenotrophomonas maltophilia. Int J Antimicrob Agents 2003;22:601-6.ArticlePubMed
- 25. Adukauskiene D, Ciginskiene A, Adukauskaite A, Koulenti D, Rello J. Clinical features and outcomes of monobacterial and polybacterial episodes of ventilator-associated pneumonia due to multidrug-resistant Acinetobacter baumannii. Antibiotics (Basel) 2022;11:892. ArticlePubMedPMC
Citations
Citations to this article as recorded by

- Stenotrophomonas maltophilia Outbreak in an ICU: Investigation of Possible Routes of Transmission and Implementation of Infection Control Measures
Maria Luisa Cristina, Marina Sartini, Gianluca Ottria, Elisa Schinca, Giulia Adriano, Leonello Innocenti, Marco Lattuada, Stefania Tigano, David Usiglio, Filippo Del Puente
Pathogens.2024; 13(5): 369. CrossRef
 , Jaehee Lee1
, Jaehee Lee1 , Byunghyuk Yu2,3
, Byunghyuk Yu2,3 , Won Kee Lee4
, Won Kee Lee4 , Sun Ha Choi1
, Sun Ha Choi1 , Ji Eun Park1
, Ji Eun Park1 , Hyewon Seo1
, Hyewon Seo1 , Seung Soo Yoo1
, Seung Soo Yoo1 , Shin Yup Lee1
, Shin Yup Lee1 , Seung-Ick Cha1
, Seung-Ick Cha1 , Chang Ho Kim1
, Chang Ho Kim1 , Jae Yong Park1
, Jae Yong Park1







 KSCCM
KSCCM

 PubReader
PubReader ePub Link
ePub Link Cite
Cite