Abstract
-
Background
- Concerns regarding positive-pressure-ventilation for the treatment of coronavirus disease 2019 (COVID-19) hypoxemia led the search for alternative oxygenation techniques. This study aimed to assess one such method, dual oxygenation, i.e., the addition of a reservoir mask (RM) on top of a high-flow nasal cannula (HFNC).
-
Methods
- In this retrospective cohort study, the records of all patients hospitalized with COVID-19 during 2020–2022 were reviewed. Patients over the age of 18 years with hypoxemia necessitating HFNC were included. Exclusion criteria were positive-pressure-ventilation for any indication other than hypoxemic respiratory failure, transfer to another facility while still on HFNC and “do-not-intubate/resuscitate” orders. The primary outcome was mortality within 30 days from the first application of HFNC. Secondary outcomes were intubation and admission to the intensive care unit.
-
Results
- Of 659 patients included in the final analysis, 316 were treated with dual oxygenation and 343 with HFNC alone. Propensity for treatment was estimated based on background diagnoses, laboratories and vital signs upon admission, gender and glucocorticoid dose. Inverse probability of treatment weighted regression including age, body mass index, Sequential Organ Failure Assessment (SOFA) score and respiratory rate oxygenation index showed treatment with dual oxygenation to be associated with lower 30-day mortality (adjusted hazard ratio, 0.615; 95% confidence interval, 0.469–0.809). Differences in the secondary outcomes did not reach statistical significance.
-
Conclusions
- Our study suggests that the addition of RM on top of HFNC may be associated with decreased mortality in patients with severe COVID-19 hypoxemia.
-
Keywords: COVID-19; hypoxia; pneumonia
INTRODUCTION
Hypoxemic respiratory failure is the most feared complication of novel coronavirus (coronavirus disease 2019 [COVID-19]) pneumonia [1]. Affecting more than 14% of patients hospitalized with COVID-19, this is the leading cause of both intensive care unit (ICU) admissions and mortality in this population [2]. Positive pressure ventilation—both invasive and noninvasive—has been long accepted as the most effective method of improving oxygenation [3]. Nonetheless, various concerns regarding the availability of these treatments (including shortages in ventilators and skilled operators), have lead the quest for alternative methods of oxygenation [4], aimed at alleviating the burden of invasive and noninvasive positive pressure ventilation and theirs potential complications.
One such method, often denominated as “dual oxygenation” and vastly implemented in our tertiary care medical center, is the addition of a reservoir mask (RM) on top of a high-flow nasal cannula (HFNC). HFNC has been extensively shown to improve oxygenation by reducing effective dead space ventilation and markedly increasing the fraction of inspired oxygen (FiO2) [5], as well as significantly increasing the end expiratory pressure [6]. This modality was also shown to reduce 30-day mortality compared to other noninvasive measures [7]. Some alterations to the flow rate and oxygen concentrations of HFNC [8], the effect of manipulating the delivery device [9], opening to shutting the patient’s mouth [6] or the addition of a surgical mask on top of the HFNC have been empirically evaluated [10]. This study aimed to evaluate the effects of dual oxygenation, i.e., the addition of RM on top of HFNC aiming to increase the amount of oxygen delivered to the alveoli in the attempt to avoid the need for invasive ventilation, compared to HFNC alone, on clinically significant outcome measures in the treatment of COVID-19 associated hypoxemic respiratory failure.
MATERIALS AND METHODS
This human study was approved by the Institutional Ethics Committee of Rambam Medical Center (No. 0228-21-RMC-D). The requirement for consent was waived by the ethics committee due to the retrospective nature of this study.
This human study was approved by our Institutional Ethics Committee - approval: 0228-21-RMC-D. The requirement for consent was waived by the ethics committee due to the retrospective nature of this study.
This retrospective cohort study aimed to compare the treatment of severely hypoxemic COVID-19 patients with either HFNC alone (control) or dual oxygenation (HFNC with the addition of a RM). The study was conducted in Rambam Healthcare Campus, a tertiary level care 1,100 beds medical center, situated in Haifa, Israel. The Electronic Health Registry files of all patients hospitalized between January 1, 2020, and December 31, 2022, in any one of eight dedicated COVID-19 wards were reviewed. To be included, a patient had to have had (1) a positive polymerase chain reaction test for COVID-19 performed within 2 days of hospital admission, (2) severe hypoxemia requiring the use of HFNC, defined as pulse oxygen saturation (SpO2) persistently below 90% despite maximal flow (15 L/min) on RM, and (3) 18 years of age or older. Exclusion criteria were (1) positive pressure ventilation (either noninvasive or invasive) initiated for any reason other than refractory hypoxemia (e.g., general anesthesia), (2) transfer to another facility while still on HFNC, and (3) patients with do-not-resuscitate (DNR) or do-not-intubate (DNI) orders at any point during the hospitalization.
All patients were continuously monitored by pulse-oximetry. For HFNC, a flow of 40 L/min heated to 37 ℃ and humidified to 100% was used, at FiO2 of 40%–100%, titrated to achieve capillary SpO2 of 90%–95%. RM was added via a non-rebreather mask at a fixed oxygen flow of 15 L/min at ambient temperature. Invasive mechanical intubation was performed at the attending physician’s discretion, mostly because of decreasing SpO2 despite maximal non-invasive respiratory support. The primary outcome was mortality within 30 days from the first application of HFNC. Secondary outcomes were the initiation of invasive intermittent positive pressure ventilation (i.e., intubation) and admission to an ICU.
Statistical Analysis
Standard descriptive statistics were used to summarize population characteristics. Survival analysis was performed using the Gehan-Breslow-Wilcoxon method and visualized by means of a Kaplan-Meier survival plot. Cox regression was performed for multivariate survival analysis. Propensity for the treatment group was calculated by logistic regression, based on a priori selected variables. Inverse probability of treatment weighting was selected due to the similar sample sizes of the intervention (dual) and control (HFNC alone) groups. A two-sided P <0.05 was considered statistically significant for all tests. Assuming patients treated with dual oxygenation to comprise at least 40% of the analyzed population the minimal total sample size needed to detect a relative hazard of 0.8 or lower (with β=0.2) for the primary outcome of 30 days mortality was calculated to be 657. Statistical analysis was performed using IBM SPSS software ver. 24.0 (IBM Corp.).
RESULTS
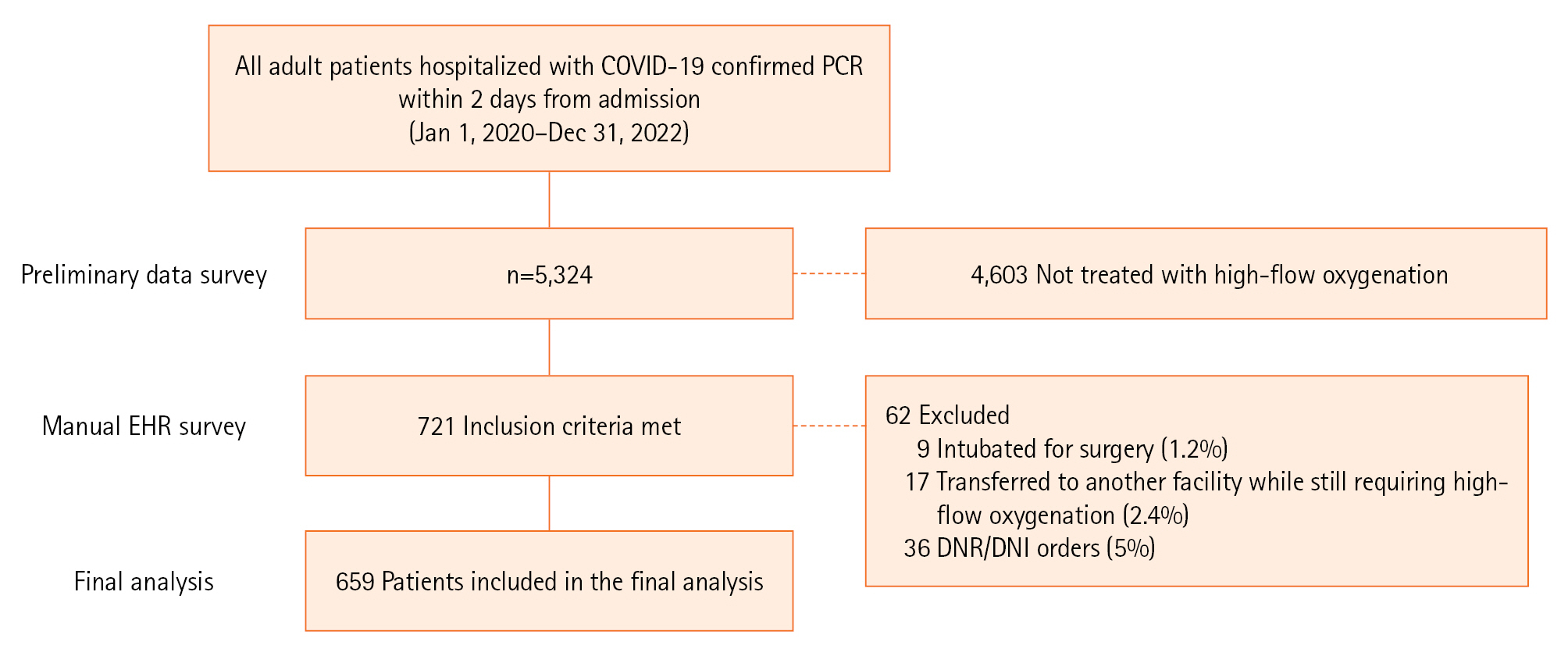
Of 5,324 patients hospitalized with COVID-19 during the study period, 721 (13.5%) were treated with HFNC. Nine patients (1.2%) were excluded for being intubated for surgery, 17 (2.4%) were transferred to another facility while still requiring high-flow oxygenation, and 36 (5%) had DNR or DNI orders. A total of 659 patients were included in the final analysis: 343 (52%) were treated with HFNC alone (controls) and 316 patients (48%) were treated with dual oxygenation (the combination of HFNC and RM), as summarised in Figure 1. The characteristics of both groups are presented in Table 1.
The background diagnoses of solid and hematological malignancies, heart failure and dyslipidemia were significantly (P<0.05) more prevalent in the dual oxygenation group. Patients treated with dual oxygenation had higher mean white blood cells count (5.6±12.9 vs. 3.8±7.4 ×103/μl, P=0.019), blood lactate (0.7±1.0 vs. 1.0±1.1 mMol/L P<0.001) and C-reactive protein levels (6.6±9.9 vs. 9.1±10.7 mg/dl P=0.002). The respiratory rate oxygenation (ROX) index measured at 6 hours after the initial application of HFNC was significantly higher in the dual oxygenation group (4.7±1.4 vs. 4.4±1.1, P=0.003). There were no significant differences between the two groups in the lowest Sequential Organ Failure Assessment (SOFA) score during hospitalization, as well as length of treatment or total dose of corticosteroids administered. Rates of, time to and duration of noninvasive bilevel positive airway pressure support, days spent using HFNC, and body mass indexes were similar in both groups.
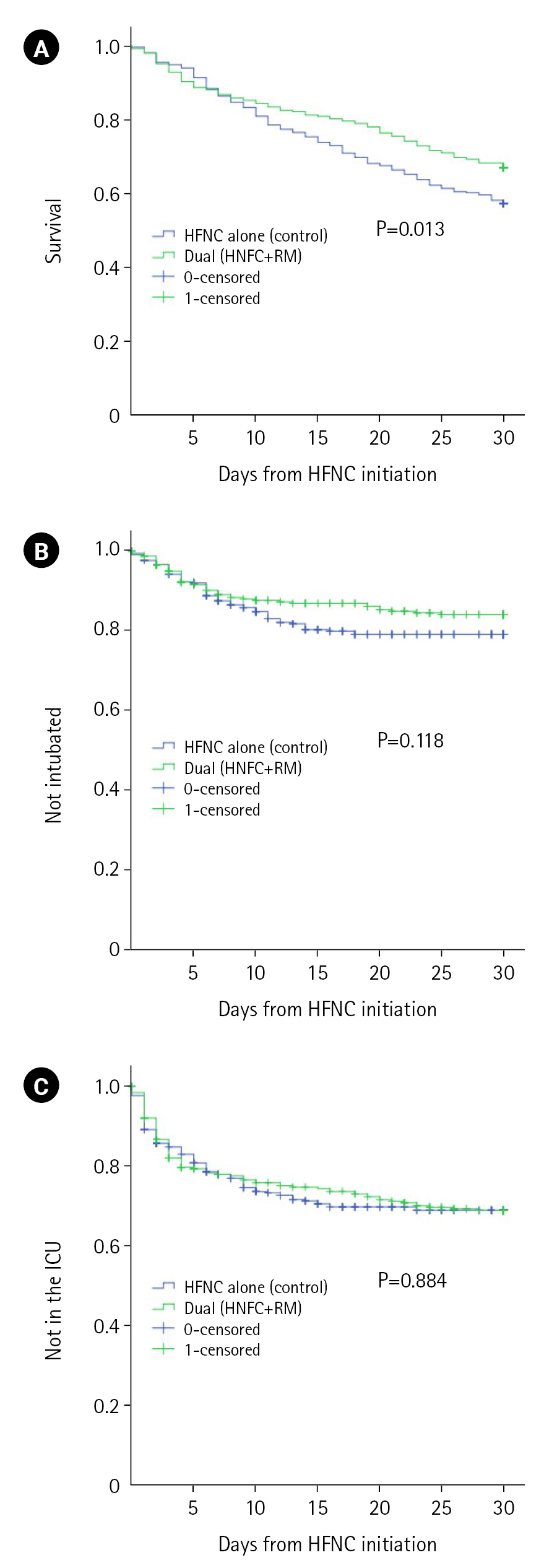
At 30 days from the initial application of HFNC, the primary outcome of all-cause mortality was significantly lower in the dual oxygenation group (104 [32.9%] vs. 146 [42.6%], P=0.011). The lower proportion of invasive positive pressure ventilation in the dual oxygenation group within 30 days from HFNC initiation (46 [14.6%] vs. 65 [19%], P=0.132) did not reach statistical significance. Rates of ICU admission within 30 days from HFNC initiation were similar between groups (93 [29.3%] and 100 [29.2%], P=0.938). Survival analysis of both primary and secondary outcomes are visualized Figure 2.
Assessing for potential confounders, propensity for the treatment group (dual vs. control) was scored using a set of antecedently selected independent variables. These included gender, background diagnoses (solid and hematological malignancy, hypertension, heart failure, ischemic heart disease, atrial fibrillation, dyslipidemia, diabetes, dialysis, smoking, asthma, and chronic obstructive pulmonary disease), laboratories upon admission (neutrophil and lymphocyte counts, lactate, C-reactive protein, D-dimer, bilirubin and serum creatinine), vital signs on presentation (pulse, blood pressure, SpO2, respiratory rate and temperature) and the total dose of glucocorticoids administered. This scoring was used to inversely weight the probability of treatment group when analyzing the primary outcome of overall survival at 30 days. Next, Cox proportional weighted hazard model was constructed. Age, body mass index, highest SOFA score during hospitalization and ROX index at 6 hours after the initiation of HFNC were included in the model. Dual oxygenation remained an independent predictor of 30 days mortality (adjusted hazard ratio, 0.615; 95% confidence interval, 0.469–0.809), as presented in Supplementary Table 1. The inverse probability of treatment weighted survival is depicted in Figure 3.
DISCUSSION
Our study demonstrated a decreased 30 days mortality in patients treated with the addition of RM on top of HFNC. This association remained unchanged after accounting for multiple potential confounders, including age, smoking status, body mass index, underlying diseases (and particularly chronic lung diseases), laboratories and vital signs at presentation and severity as reflected by the SOFA score and ROX index. Several mechanistic explanations could be offered to elucidate these results.
First, flow addition might be of importance. While 40 L/min is much higher than the typical minute ventilation, this ventilatory volume is inspired over a period of time that is significantly shorter, leading to high-peak inspiratory flows (PIF). Patients in respiratory failure demonstrated PIFs of over 42 L/min on average and up to 120 L/min [11]. Conversely, the O2 infrastructure in our hospital only allows for up to 40 L/min flow of pure oxygen, lower than the maximal flow of most available HFNC devices, that support minute flow of up to 60 L/min. In other words, 15 L/min of RM added to the HFNC of 40 L/min (increasing flow by 37.5) may be significant in view of the high PIF in respiratory failure [12,13]. In a recent bench and healthy volunteers model, increasing the flow from 40 to 60 also increased the positive end expiratory pressure, which might explain improved oxygenation [6].
Second, not unlike the addition of a surgical mask (shown in a small study to improve oxygenation) [10] or simply adding a venturi mask with no flow [14], the RM adds a physical barrier. All high flow devices in our center rely on nasal prongs for delivery. Shifts to the hemoglobin dissociation curve [15], and the rise of PaCO2 [16] have all been suggested to explain these findings. It seems, nonetheless, that the most likely mechanism is the decrease of room air entrainment in the presence of a physical barrier. PEEP was significantly lowered when the subjects opened their mouths [6]—a posture typical of the air hungry patient. This was shown in volunteers subjected to strenuous exercise to simulate the increased ventilation in severe respiratory failure (shown to breathe through their mouth) [17]. Partial arterial pressure of O2 was significantly increased (with PaCO2 unchanged) when a barrier was applied [10]. FIO2 delivery was shown to decrease significantly in severe respiratory failure, a finding attributed to room air entrainment, in a recent meta-analysis [18]. It seems the simple barrier of the RM helps funnel the high flow from the nasal prongs to the mouth, contributing especially in severe respiratory distress where patients are more likely to breathe with their mouth open.
The decrease in room air entrainment might be even more significant when a highly compliant reservoir is added [9]. The large diameter and proximity of the reservoir bag minimize the resistance of this source of effectively pure oxygen during inspiration [7]. Since, as discussed above, the limiting factor in respiratory failure seems to be flow rather than volume, even the modest volume (but high flow) reservoir bag could help address the increased PIF needs of patients with respiratory failure.
This study has several important limitations. Firstly, the retrospective design inherently raises the risk of biases, particularly since no randomization was performed and no strict protocol detailing the criteria for adding RM to HFNC was followed. As a result, criteria for adding RM or ICU admission varied greatly between physicians and during various phases of the pandemic, as did criteria for intubation or ICU admission. Secondly, no consistent data was available regarding important physiological parameters (oxygenation, respiratory effort, etc.) immediately before and after the addition of RM, leaving our inference of the mechanism by which this intervention improves oxygenation to be very limited, relying on previous much smaller studies. Similarly, we could not ascertain the extent and duration to which the patient’s mouth was open. Lastly, mortality in this cohort was high. While sadly not atypical of the pandemic, such rates are not expected in all respiratory failure patients.
In conclusion, this study demonstrates the potential efficacy of the addition of RM on top of HFNC in COVID-19 patients with severe hypoxemia, resulting in improved overall survival. This cheap, widely available, and safe treatment may warrant further studies.
KEY MESSAGES
▪ We investigated a simple, readily available, safe, and yet not sufficiently researched oxygenation method: the addition of a reservoir mask on top of high flow oxygen cannula.
▪ Our data suggest this rudimentary intervention can be associated with decreased 30-day mortality.
▪ These findings could be extremely important for clinicians and incentivize further studies.
NOTES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
FUNDING
None.
-
AUTHOR CONTRIBUTIONS
Conceptualization: IG, RZ, EF. Methodology: IG. Formal analysis: IG. Data curation: IG, KH. Visualization: IG. Project administration: EF. Writing–original draft: IG, EF. Writing–review & editing: IG, RZ, KH, AN, EF.
Acknowledgments
None.
SUPPLEMENTARY MATERIALS
Supplementary materials can be found via https://doi.org/10.4266/acc.2023.00451.
Figure 1.Flowchart of the study. COVID-19: coronavirus disease 2019; PCR: polymerase chain reaction; DNR: do-not-resuscitate; DNI: do-not-intubate.
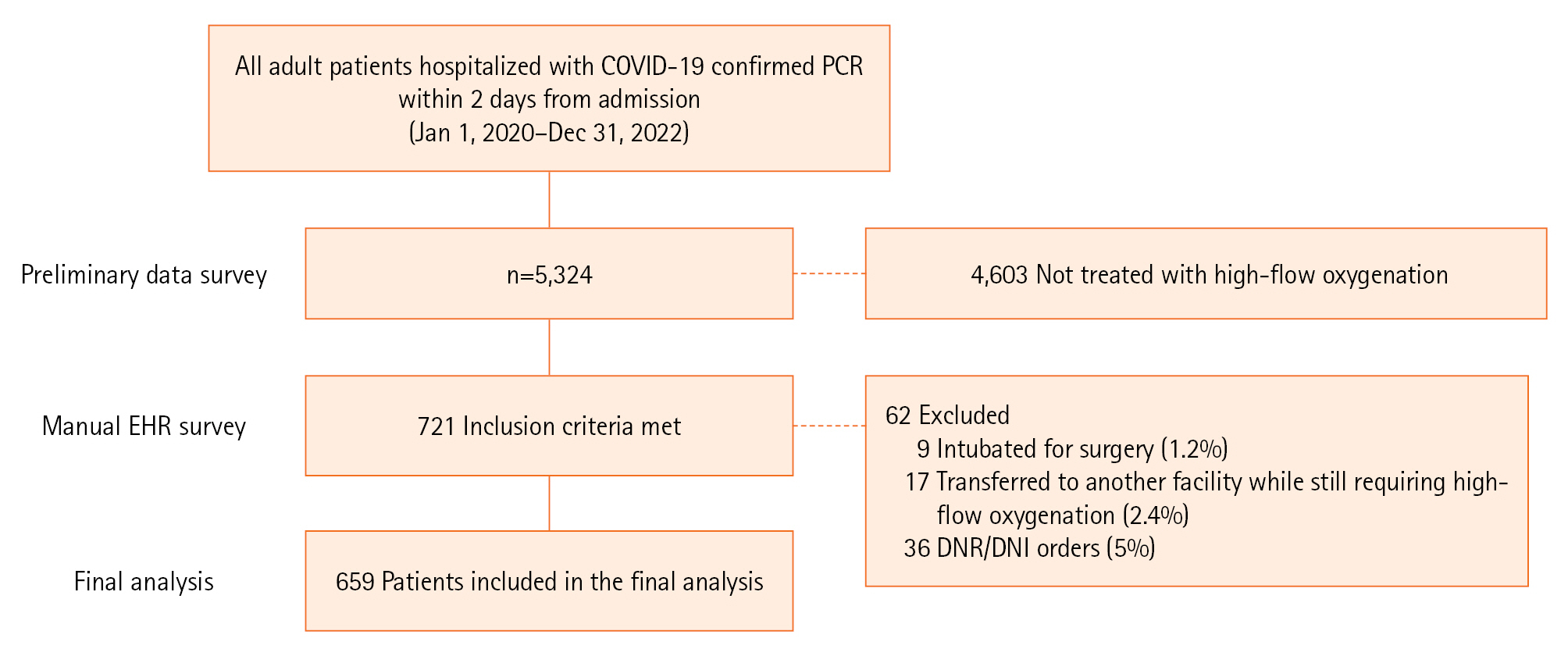
Figure 2.Kaplan-Mayer survival curves from first high-flow nasal cannula (HFNC) to death (A), the initiation of invasive positive pressure ventilation (i.e., intubation) (B), and admission to the intensive care unit (ICU) (C). RM: reservoir mask.
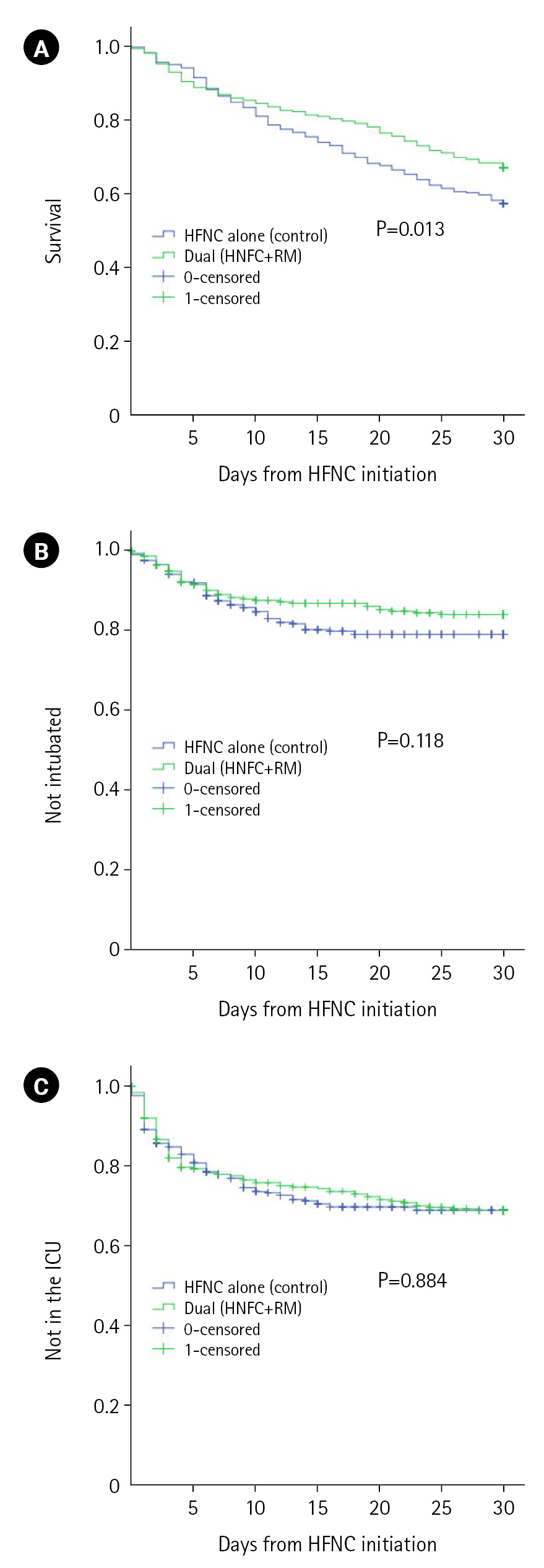
Figure 3.Propensity score weighted Cox regression for 30 days survival. Predicting variables included respiratory support method (dual vs. high-flow nasal cannula [HFNC] alone), age, respiratory rate oxygenation (ROX) index, Sequential Organ Failure Assessment score, and body mass index. RM: reservoir mask.
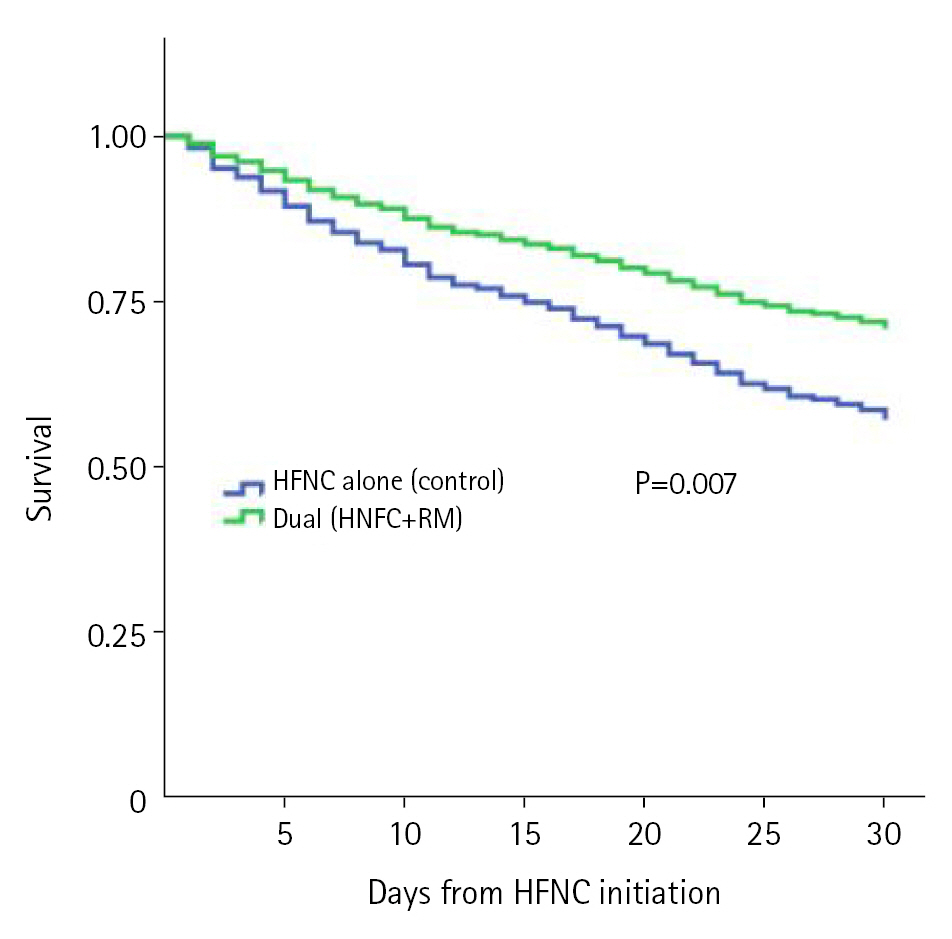
Table 1.Study patients characteristics
|
Variable |
HFNC alone (control, n=343) |
Dual (HFNC+RM, n=316) |
P-value |
|
Female |
131 (38.2) |
110 (34.8) |
0.368 |
|
Age (yr) |
66.5±15.6 |
68.0±15.7 |
0.198 |
|
History |
|
|
|
|
Solid malignancy |
72 (20.9) |
93 (29.4) |
0.012 |
|
Hematological malignancy |
24 (7.0) |
39 (12.3) |
0.020 |
|
Heart failure |
103 (30.0) |
135 (42.7) |
0.001 |
|
Hemodialysis |
21 (6.1) |
32 (10.1) |
0.059 |
|
COPD |
31 (9.0) |
23 (7.3) |
0.411 |
|
Asthma |
13 (3.8) |
17 (5.4) |
0.328 |
|
Smoking |
43 (12.5) |
48 (15.2) |
0.324 |
|
Atrial fibrillation |
50 (14.6) |
58 (18.4) |
0.191 |
|
Dyslipidemia |
146 (42.6) |
165 (52.2) |
0.013 |
|
Hypertension |
82 (23.9) |
109 (34.5) |
0.003 |
|
Ischemic heart disease |
90 (26.2) |
98 (31.0) |
0.175 |
|
Diabetes mellitus |
135 (39.4) |
131 (41.5) |
0.584 |
|
Laboratory results upon admission |
|
|
|
|
White blood cells (×103/μl) |
3.8±7.4 |
5.6±12.9 |
0.019 |
|
Neutrophiles (×103/μl) |
6.9±4.2 |
7.2±5.3 |
0.563 |
|
Lymphocytes (×103/μl) |
1.9±14.2 |
2.3±13.3 |
0.779 |
|
Hemoglobin (mg/dl) |
12.0±2.3 |
12.3±2.3 |
0.153 |
|
Lactate (mMol/L) |
0.7±1.0 |
1.0±1.1 |
<0.001 |
|
C-reactive protein (mg/dl) |
6.6±9.9 |
9.1±10.7 |
0.002 |
|
D-dimer (mg/ml) |
1,321±1,083 |
1,489±1,182 |
0.198 |
|
Bilirubin (mg/dl) |
0.6±0.4 |
0.6±0.6 |
0.420 |
|
Serum creatinine (mg/dl) |
1.5±1.5 |
1.4±1.4 |
0.511 |
|
Glucocorticoid length of treatment (day) |
5.9±3.8 |
6.0±3.7 |
0.882 |
|
Glucocorticoid cumulative dose (prednisone equivalent in mg) |
237±153 |
238±148 |
0.879 |
|
Highest SOFA score during hospitalization |
4.7±1.7 |
4.7±1.8 |
0.983 |
|
Highest ROX index within 12 hours from HFNC initiation |
4.4±1.1 |
4.7±1.4 |
0.003 |
|
Body mass index (kg/m2) |
29.0±5.8 |
29.6±6.2 |
0.240 |
|
Day on HFNC |
13 (4 to 22) |
13 (4 to 24) |
0.266 |
|
BiPAP |
84 (24.5) |
68 (21.5) |
0.366 |
|
Day to BiPAP initiation |
0 (0 to 4) |
–1 (–2 to 2) |
0.389 |
|
Day on BiPAP 3 |
3 (1 to 6) |
3 (1 to 5) |
0.635 |
|
Deceased 30 days after first application of HFNC |
146 (42.6) |
104 (32.9) |
0.011 |
|
Intubated 30 days after first application of HFNC |
65 (19) |
46 (14.6) |
0.132 |
|
Admitted to the ICU within 30 days after first application of HFNC |
100 (29.2) |
93 (29.3) |
0.938 |
References
- 1. Macedo A, Gonçalves N, Febra C. COVID-19 fatality rates in hospitalized patients: systematic review and meta-analysis. Ann Epidemiol 2021;57:14-21.ArticlePubMedPMC
- 2. Izcovich A, Ragusa MA, Tortosa F, Lavena Marzio MA, Agnoletti C, Bengolea A, et al. Prognostic factors for severity and mortality in patients infected with COVID-19: a systematic review. PLoS One 2020;15:e0241955.ArticlePubMedPMC
- 3. Groff P, Ferrari R. Non-invasive respiratory support in the treatment of acute hypoxemic respiratory failure secondary to COVID-19 related pneumonia. Eur J Intern Med 2021;86:17-21.ArticlePubMedPMC
- 4. Crimi C, Pierucci P, Renda T, Pisani L, Carlucci A. High-flow nasal cannula and COVID-19: a clinical review. Respir Care 2022;67:227-40.ArticlePubMed
- 5. Godoy DA, Graneros N, Oyarzabal L, Murillo-Cabezas F. High-flow oxygen therapy in acute hypoxemic respiratory failure secondary to COVID-19 pneumonia. Med Intensiva (Engl Ed) 2021;45:506-8.ArticlePMC
- 6. Vieira F, Bezerra FS, Coudroy R, Schreiber A, Telias I, Dubo S, et al. High-flow nasal cannula compared with continuous positive airway pressure: a bench and physiological study. J Appl Physiol (1985) 2022;132:1580-90.Article
- 7. Gur I, Zalts R, Azzam M, Hussein K, Neuberger A, Fuchs E. Dual oxygenation in the treatment of hypoxemic respiratory failure: a retrospective cohort study. Isr Med Assoc J 2023;25:595-600.
- 8. Leeies M, Flynn E, Turgeon AF, Paunovic B, Loewen H, Rabbani R, et al. High-flow oxygen via nasal cannulae in patients with acute hypoxemic respiratory failure: a systematic review and meta-analysis. Syst Rev 2017;6:202. ArticlePubMedPMCPDF
- 9. Duprez F, Bruyneel A, Machayekhi S, Droguet M, Bouckaert Y, Brimioulle S, et al. The double-trunk mask improves oxygenation during high-flow nasal cannula therapy for acute hypoxemic respiratory failure. Respir Care 2019;64:908-14.ArticlePubMed
- 10. Montiel V, Robert A, Robert A, Nabaoui A, Marie T, Mestre NM, et al. Surgical mask on top of high-flow nasal cannula improves oxygenation in critically ill COVID-19 patients with hypoxemic respiratory failure. Ann Intensive Care 2020;10:125. ArticlePubMedPMCPDF
- 11. Mälberg J, Hadziosmanovic N, Smekal D. Physiological respiratory parameters in pre-hospital patients with suspected COVID-19: a prospective cohort study. PLoS One 2021;16:e0257018.ArticlePubMedPMC
- 12. Duprez F, de Terwangne C, Bellemans V, Poncin W, Reychler G, Sorgente A, et al. High-flow nasal cannula therapy, factors affecting effective inspired oxygen fraction: an experimental adult bench model. J Clin Monit Comput 2022;36:1441-8.ArticlePubMedPDF
- 13. Mauri T, Alban L, Turrini C, Cambiaghi B, Carlesso E, Taccone P, et al. Optimum support by high-flow nasal cannula in acute hypoxemic respiratory failure: effects of increasing flow rates. Intensive Care Med 2017;43:1453-63.ArticlePubMedPDF
- 14. Dogani B, Månsson F, Resman F, Hartman H, Tham J, Torisson G. The application of an oxygen mask, without supplemental oxygen, improved oxygenation in patients with severe COVID-19 already treated with high-flow nasal cannula. Crit Care 2021;25:319. ArticlePubMedPMCPDF
- 15. Born P, Castro R. A combination of Bohr and Haldane effects provide a physiologic explanation for the increase in arterial oxygen saturation when a face mask is added to a high-flow nasal cannula in severely hypoxemic COVID-19 patients. Crit Care 2021;25:395. ArticlePubMedPMCPDF
- 16. Swenson ER. Does inspiration of exhaled CO2 explain improved oxygenation with a face mask plus high-flow nasal cannula oxygen in severe COVID-19 infection? Crit Care 2021;25:343. ArticlePubMedPMCPDF
- 17. Ritchie JE, Williams AB, Gerard C, Hockey H. Evaluation of a humidified nasal high-flow oxygen system, using oxygraphy, capnography and measurement of upper airway pressures. Anaesth Intensive Care 2011;39:1103-10.ArticlePubMedPDF
- 18. Lee CC, Mankodi D, Shaharyar S, Ravindranathan S, Danckers M, Herscovici P, et al. High flow nasal cannula versus conventional oxygen therapy and non-invasive ventilation in adults with acute hypoxemic respiratory failure: a systematic review. Respir Med 2016;121:100-8.ArticlePubMed
Citations
Citations to this article as recorded by

 , Ronen Zalts1,2
, Ronen Zalts1,2 , Yaniv Dotan2
, Yaniv Dotan2 , Khitam Hussain1,2, Ami Neuberger1,2, Eyal Fuchs1,2
, Khitam Hussain1,2, Ami Neuberger1,2, Eyal Fuchs1,2







 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite