Abstract
-
Background
- In this study, we explored whether awake prone position (PP) can impact prognosis of severe hypoxemia coronavirus disease 2019 (COVID-19) patients.
-
Methods
- This was a prospective observational study of severe, critically ill adult COVID-19 patients admitted to the intensive care unit. Patients were divided into two groups: group G1, patients who benefited from a vigilant and effective PP (>4 hours minimum/24) and group G2=control group. We compared demographic, clinical, paraclinical and evolutionary data.
-
Results
- Three hundred forty-nine patients were hospitalized during the study period, 273 met the inclusion criteria. PP was performed in 192 patients (70.3%). The two groups were comparable in terms of demographic characteristics, clinical severity and modalities of oxygenation at intensive care unit (ICU) admission. The mean PaO2/FIO2 ratios were 141 and 128 mm Hg, respectively (P=0.07). The computed tomography scan was comparable with a critical >75% in 48.5% (G1) versus 54.2% (G2). The median duration of the daily PP session was 13±7 hours per day. The average duration of spontaneous PP days was 7 days (4–19). Use of invasive ventilation was lower in the G1 group (27% vs. 56%, P=0.002). Healthcare-associated infections were significantly lower in G1 (42.1% vs. 82%, P=0.01). Duration of total mechanical ventilation and length of ICU stay were comparable between the two groups. Mortality was significantly higher in G2 (64% vs. 28%, P=0.02).
-
Conclusions
- Our study confirmed that awake PP can improve prognosis in COVID-19 patients. Randomized controlled trials are needed to confirm this result.
-
Keywords: COVID-19; intensive care unit; patient outcomes; prone position; respiratory distress
INTRODUCTION
Since December 2019 and the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) outbreak in China, a large number of cases have been reported worldwide. Although the majority of patients show mild symptoms, coronavirus disease 2019 (COVID-19) was responsible for severe illness characterized by progressive hypoxemic respiratory failure. Some patients had rapidly developed acute respiratory distress syndrome (ARDS) and other systemic complications followed by multiple organs failure and need for invasive mechanical ventilation and intensive care unit (ICU) admission [1-3]. In patients who are intubated for moderate to severe ARDS, prone position (PP) was proposed as an adjuvant non respiratory tool allowing to improve oxygenation and reduce mortality. Its benefit was mainly reported highly hypoxemic patients [4]. Recently, awake PP has been proposed to enhance oxygenation in non-intubated patients with ARDS even those with severe COVID-19 [5] and it has been recently incorporated into guidelines and expert consensus statements [6,7] and has been identified as a research priority by the Surviving Sepsis Research Committee [8]. In this context, we aimed to assess if awake PP reduces the rate death or intubation in patients with severe COVID-19 acute hypoxemic respiratory failure who require respiratory support.
MATERIALS AND METHODS
Study Design and Setting
This study was approved by the Institutional Review Board of the Regional Hospital of Zaghouan (No. 32/2020) with a written informed consent for patients.
We conducted a prospective, observational and single-center study from March 2020 to September 2022 in the ICU of Regional Hospital of Zaghouan. All patients admitted to the ICU diagnosed with acute hypoxemic respiratory failure due to COVID-19 and confirmed by a reverse transcription polymerase chain reaction test were eligible. Acute hypoxemic respiratory failure was defined as a requirement of respiratory support with high-flow nasal cannula or non-invasive ventilation with the ratio of the partial pressure of arterial oxygen to the fraction of inspired oxygen (PaO2/FiO2) ≤300 mm Hg. Awake PP was explained to every patient and they were encouraged to spend as much time in PP as they could tolerate. Target time in PP was 4 to 12 hours per day divided into three sessions which were lasting 4 to 6 hours. PP was performed one hour after meals to avoid gastrointestinal side effects.
We excluded patients who were unable to awake PP, hemodynamically unstable, pregnant women, or those who were intubated during the first 24 hours. However, patients who refused the PP were used as a control group. Demographical, clinical, radiological, and laboratory information were recorded, including comorbidities, respiratory variables, and details of treatments administered for COVID-19 on the day of admission and during ICU stay.
All patients received antibiotics if the inflammatory parameters or computed tomography (CT) lesions showed suspicion of bacterial surinfection, vitamins, heparin and corticoids (dexamethasone 6 mg/day). Remdesivir and hydroxychloroquine were not prescribed in our study.
Oxygen therapy was initiated with face mask at 5 L/min and the flow rate was titrated to reach the target oxygen saturation as measured by pulse oximetry (SpO2) >94%. If the target SpO2 was not achieved then non-rebreathing mask at 10 to 15 L/min was considered. Non-invasive ventilation was started if PaO2/FiO2 <200 mm Hg or patients presented signs of respiratory distress such as retractions or accessory muscle use. Mechanical invasive ventilation was considered in refractory hypoxemic patient (PaO2/FiO2 <150 mm Hg with non-invasive ventilation) or in presence of other associated distress.
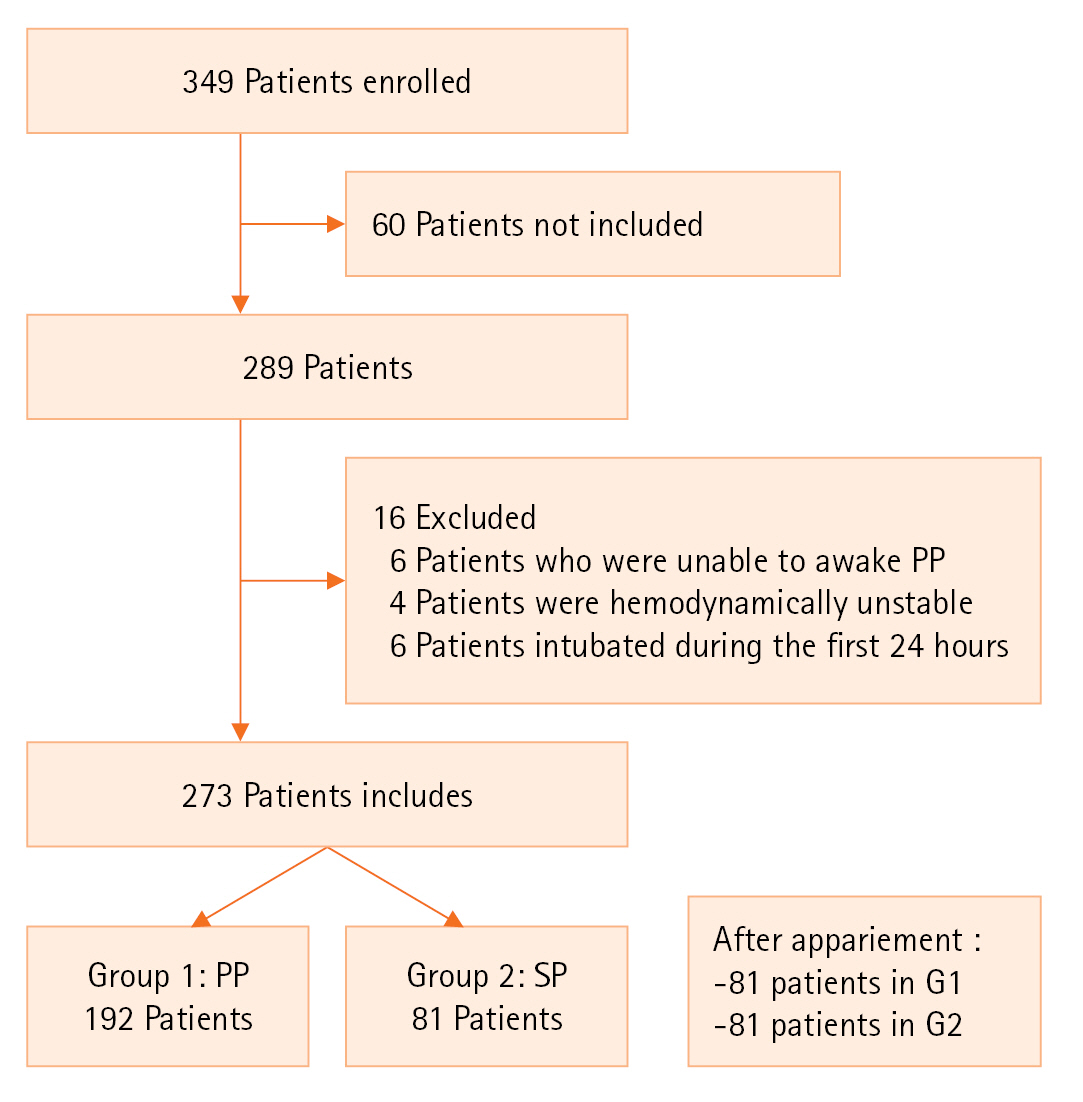
Included patients were divided into two groups: (1) PP group: patients who benefit of awake vigilant and effective PP (>8 hours minimum/24) and (2) supine position (SP) group:supine group. A total of 349 patients were enrolled in the study period, among them, 273 patients were initially included: 192 patients in SP (70.3%) and 81 patients in PP (29.6%). The two groups were secondary matched according to demographic, clinical, and biological features. Two groups were divided G1 (PP) included 81 patients and G2 (SP) included 81 patients. The primary endpoint was ICU mortality. The secondary outcome were need for mechanical ventilation and length of hospital stay (Figure 1).
Definitions
Awake PP
PP in non-intubated patients, has been attempted for patients with acute hypoxemic respiratory failure [1].
Hemodynamically unstable
It is an acute circulatory failure, due to an imbalance between oxygen intake and needs (O2), not spontaneously reversible, resulting in tissue hypoxia by [2]: either a tissue perfusion defect causing a lack of O2 intake (cellular hypoxia) and/or a defect in adenosine triphosphate production (energy deficiency).
Acute renal failure
It is a decrease in rapid-onset glomerular filtration rate resulting in uremia and life-threatening short-term water-electrolyte disorders [3].
Healthcare-associated infections
Healthcare-associated infections acquired during the process of receiving health care that was not present during the time of admission. They may occur in different areas of healthcare delivery, such as in hospitals, long-term care facilities, and ambulatory settings, and may also appear after discharge [4].
Statistical Modeling
Continuous variables were reported as mean (standard deviation) or median (interquartile range) depending on the distribution of data. We conducted both unadjusted and multivariable logistic regression models to investigate the effect of both dosages on primary and secondary endpoints. The multivariable model was adjusted by potential confounding factors identified at baseline [5].
RESULTS
Demographic Characteristic
The median age of our population was 58 years with sex ratio 1.48. Comorbidities was resumed in Table 1.
Clinical Characteristics
The mean Acute Physiology and Chronic Health Evaluation (APACHE) II and Simplified Acute Physiology Score (SAPS) II severity scores were 7.7±4.4 and 24±7.4, respectively. Mainly symptoms presented at the ICU admission were resumed in Table 2. The laboratory results are shown in Table 3. The acid-base profile for both groups showed no significant differences. During ICU stay, all patients received corticosteroids, for a median time of 8 days (interquartile range, 6–10 days). In all patients, the initially corticosteroid prescribed was dexamethasone (12–24 mg/day). Moreover, 256 patients (97.4%) received heparins, 50 patients (18.3%) received preventive coagulation doses. The two groups had received the same medications. The CT scan was comparable with a critical >75% to 48.5% (G1) versus 54.2% (G2). The median duration of the daily PP session was 13±7 hours per day. The average duration of spontaneous PP days was 7 days [4–19].
Evolutive Findings
In our study, application of an awake PP was associated with a significant reduction in the need of mechanical invasive ventilation, healthcare-associated infections and acute renal failure (Table 4). Mechanical ventilation maintained for a median of 12 days for PP group versus 11 days for SP group (P=0.7) without difference between the two groups.
Mortality
In our study, we founded a higher rate of mortality in the SP group (28% vs 64%, P=0.02). The median of length of ICU stays for group PP (11 days vs. 14 days, P=0.06).
Multivariate Analysis
All factors correlated with prognosis identified in univariate analysis were included in multivariate analysis models constructed by top-down stepwise method. Ultimately, the independent factors correlated with mortality were lack of awake PP (odds ratio [OR], 3.23; 95% confidence interval [CI],1.6–4.2), occurrence of healthcare-associated infections (OR, 4.54; 95% CI, 3.24–10; P=0.02), septic shock (OR, 32.2; 95% CI, 2.89–100; P=0.04) and acute renal failure (OR, 18.86; 95% CI, 2.46–142; P=0.01) (Table 5).
DISCUSSION
Our study confirmed that awake PP can improve severe hypoxemia and respiratory failure in COVID-19 patients with spontaneous breathing. In fact, awake PP in these patients was associated with significant reduce in need to invasive mechanical ventilation and mortality compared with SP.
This positive effect seems to be related to direct relationship on chest compliance, gas exchange and hemodynamic effects. In fact, overall compliance of the chest wall is influenced by the flexibility of its three anatomical boundaries: anterior, posterior, and abdominal. In the SP, changes in compliance are most influenced by the abdomen and anterior chest wall, whereas in the PP, the posterior chest and abdomen are key determinants [7]. In ARDS patients, lung compliance is mainly determined by the degree of ventilation opening of the lungs. Notably, specific lung compliance was similar in ARDS patients and healthy individuals, suggesting that surfactant changes or early fibrosis were not major in altering lung mechanical properties [8]. It can be seen that any change in lung compliance is mainly due to the opening of new lung units and/or the mechanical improvement of the opened units to achieve a more favorable position on the volume-pressure curve [9]. In the PP, this beneficial displacement can occur by promoting a uniform distribution of total stress and strain [10]. The most striking change observed on CT scans when changing from supine to PPs is a dorsal to ventral redistribution of density [11]. Impact of PP on improving gas exchange is also determined by several effects. The first element is the amount of tissue that is open to ventilation and perfusion during the respiratory cycle. If recruitment of the dorsal lung exceeds that of the ventral lung, oxygenation should improve with unchanged perfusion distribution. The second element is the homogeneity of inflation. In homogeneity is related to the uneven distribution of ventilation. Since perfusion remains nearly constant, more uniform ventilation results in a more uniform ventilation to perfusion ratio distribution, which is reflected in reduced venous admixture and reduced dead space. Local changes in chest wall compliance may also contribute to improved oxygenation [10]. Due to the reduced compliance of the anterior chest wall and the curvature of the diaphragm, the distribution of tidal volumes actually shifts toward the posterior paraventral region of the lungs, where ventilation is usually absent in the SP. This improvement is a result of reduced heterogeneity in shunt and ventilator perfusion, as the anatomically cone-like lung fits into its cylindrical thorax, deforming less when the patient is prone or supine [11]. This reduces atelectasis in the dorsal lung region, where shunts are preferentially distributed in ARDS [12].
Although expert clearly recommend employment of PP in severe ARDS invasively ventilated, its place in critically ill COVID-19 patients non intubated still debated. The streamlined Surviving Sepsis Campaign Guidelines on the operation of grown-ups with coronavirus disease 2019 in the ICU started that there is inadequate substantiation to issue a recommendation on use of awake PP in non-intubated grown-ups with severe COVID-19 [13]. There were several confounding factors, duration, frequency of PP and type of oxygen supplementation ways weren't formalized across all included studies.
In the same way, World Health Organisation COVID-19 Clinical Management Living Guidance suggested a tentative recommendation for awake PP of oppressively ill COVID-19 cases taking supplemental oxygen (including high-inflow nasal oxygen)[14]. Lately streamlined "British Thoracic Society/Intensive Care Society: respiratory care in patients with acute hypoxaemic respiratory failure associated with COVID-19" also suggested PP or side displacing for COVID-19 cases in the respiratory support pathway [15]. A recent expert agreement for the operation of COVID-19 affiliated acute respiratory failure concluded that awake PP considered to ameliorate oxygenation and it should be used when supplemental oxygenation is needed to maintain SpO₂ >90 [16]. Numerous studies had evaluated benefit of awake PP in ARDS COVID-19 [16-20]. Elharrar et al had conducted a prospective, single-center study of PP applied in hypoxemic ARF treated with high-flow oxygen therapy [17]. Among the tolerant cases, PaO2 increased from 73.6 mm Hg before to 94.9 mm Hg during PP. Only three cases maintained better oxygenation 6–12 hours after resupination [18].
In COVID-19 hospitalized cases other analogous studies have reported advanced oxygenation during PP [18]. A methodical review and meta-analysis evaluated the effect, timing, and populations that might profit from awake proning regarding oxygenation, mortality, and tracheal intubation compared with SP in hypoxemic acute respiratory failure. This study concluded that a PP can ameliorate oxygenation among non-intubated patients with acute hypoxemic respiratory failure when applied for at least 4 hours. Awake proning appears safe, but generalization of this results needs more randomized controlled trials. The results of feasibility and safety studies will potentially help in the design of larger definitive trials to address this crucial knowledge gap [19]. Methodical review and meta-analysis of non-randomized trials demonstrated that PP enhanced PaO₂/FiO₂ with better SpO₂ than SP in COVID-19 cases. It also reported that the mortality rate was lower in those who entered prone ventilation [20].
Efficacy of PP combined with high-flow oxygen therapy remedy or non-invasive ventilation was lately reported in small cohorts ofnon-infectious and contagious non–COVID-19 ARDS cases [21]. Bahloul et al. [22] demonstrated in a prospective experimental study of severe, critically ill adult COVID-19 cases admitted to ICU, that the early operation of PP can ameliorate hypoxemia in severe hypoxemia COVID-19 patients.
This study presents some limitations. First, It was a single-center one. Second, feasibility of PP was not admitted for all patients and finally number of patients was small.
We supposed that awake PP could improves oxygenation during related ARDS COVID-19 non-intubated patients. Due to its relative ease of use, awake PP can reduce need for invasive mechanical and decrease mortality. Association of PP to non-invasive ventilation or high-flow nasal cannula is suggested to improve respiratory status. Randomized controlled trials are needed to confirm the beneficial effects of PP in COVID-19 patients with spontaneous breathing.
KEY MESSAGES
▪ Application of awake prone position (PP) for more than 8 hours/24 can improve prognostic in critically ill coronavirus disease 2019 (COVID-19) patients.
▪ Early application of awake PP seems to reduce the use of invasive mechanical ventilation.
▪ Awake PP seem to reduce the incidence of healthcare-associated infections.
▪ Randomized controlled trials are needed to confirm the beneficial effects of awake PP in COVID-19 patients.
NOTES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
FUNDING
None.
-
AUTHOR CONTRIBUTIONS
Conceptualization: KBI, IS. Methodology: KBl, IS, Formal analysis: KBI, IS, FE, IT, BBD. Data curation: KBI, IS, TM . Visualization: TM. Project administration: KBI, TM. Funding acquisition: KBS, NBS. Writing – original draft: KBI, IS. Writing – review & editing: TM, KBI, IS.
Acknowledgments
None.
Figure 1.Flowchart of population. PP: prone position; SP: supine position.

Table 1.The clinical presentation of the study group on hospital admission
|
Variable |
Population (n=273) |
PP (n=192) |
SP (n=81) |
P-value |
|
Age (yr) |
58 (28–92) |
55 (28–60) |
56 (28–75) |
0.69 |
|
Sex ratio (M/F) |
1.5 |
1.4 |
1.2 |
0.70 |
|
BMI (kg/m2) |
29 (21–58) |
33 (21–58) |
29 (22–50) |
0.01 |
|
Presence of comorbidities |
124 (45.7) |
83 (43.7) |
32 (39.6) |
0.07 |
|
Arterial hypertension |
96 (35.5) |
66 (34.8) |
29 (36.1) |
0.81 |
|
Diabetes |
87 (31.9) |
61 (31.6) |
26 (32.1) |
0.09 |
|
Cardiac comorbidities |
43 (16.1) |
25 (13) |
18 (22.2) |
0.06 |
|
History of PE |
3 (1.2) |
2 (1.0) |
1 (1.2) |
0.07 |
|
Dyslipidemia |
52 (19.4) |
31 (16.1) |
21 (25.9) |
0.06 |
|
Chronic respiratory failure |
57 (21.2) |
34 (17.7) |
23 (28.3) |
0.07 |
|
Asthma |
10 (3.7) |
7 (3.6) |
3 (3.7) |
0.92 |
|
COPD |
19 (7.1) |
15 (7.8) |
4 (4.9) |
0.07 |
|
CKD |
2 (0.8) |
1 (0.5) |
1 (1.2) |
0.81 |
|
Hypothyroidism |
12 (3.4) |
10 (5.2) |
2 (2.4) |
0.74 |
Table 2.Clinical parameter of the study group on admission
|
Variable |
Total |
PP |
SP |
P-value |
|
Temperature (°C) |
36.7±2.5 |
36.8±2.7 |
36.5±0.2 |
0.67 |
|
RR (cycles/min) |
29.7±6.1 |
30±5.5 |
29±1.6 |
0.73 |
|
PaO2/FIO2 ratio |
132±2 |
141±1 |
128±2 |
0.07 |
|
CR (bpm) |
88±17 |
92±17 |
88±9 |
0.23 |
|
Glasgow scale score |
14±1 |
14±1 |
14±1 |
0.67 |
|
Dyspnea |
261 (95.8) |
182 (94.7) |
79 (97.5) |
0.91 |
|
Fever |
238 (87.4) |
178 (92.7) |
60 (74.1) |
0.12 |
|
Cough |
271 (77.9) |
190 (98.9) |
81 (100) |
0.70 |
Table 3.Paraclinical characteristics of the study group
|
Variable |
Total |
PP |
SP |
P-value |
|
Leucocytes (103/μl) |
9,808±55 |
10,302±6,359 |
9,066±4,047 |
0.402 |
|
Lymphocytes (103/μl) |
990±110 |
1,125±349 |
787±550 |
0.251 |
|
CRP (mg/L) |
122±87 |
148±87 |
85±75 |
0.006 |
|
Creatinine (mmol/dl) |
90±65 |
93±70 |
86±62 |
0.806 |
|
Natremia (mEq/L) |
133±16 |
135±6 |
130±24 |
0.196 |
|
Kaliemia (mEq/L) |
4.0±0.5 |
4.1±0.6 |
3.9±0.4 |
0.248 |
|
Hemoglobin (mg/dl) |
12.2±1.9 |
12.3±2.1 |
11.7±0.7 |
0.305 |
|
Platelets (103/μl) |
246±81 |
236±77 |
328±73 |
0.058 |
|
PH |
7.41±0.12 |
7.41±0.05 |
7.47±0.01 |
0.749 |
|
PaO2/FiO2 (mm Hg) |
124±44 |
126±44 |
104±54 |
0.673 |
Table 4.The evolutive findings of the study group
|
Parameter |
PP (n=192) |
SP (n=81) |
P-value |
|
PEEP (cm H2O), mean±SD |
6.9±2 |
8±1.8 |
0.017 |
|
Needed of IMV (%) |
27 |
56 |
0.002 |
|
PaO2/FIO2
|
280 |
195 |
0.02 |
|
Nosocomial infection (%) |
42.1 |
82 |
0.01 |
|
VAP |
26.6 |
54.4 |
0.002 |
|
UTI |
6.6 |
15.1 |
0.68 |
|
Fungal infection |
0 |
2.3 |
0.91 |
|
TE |
20 |
5.8 |
0.06 |
|
Acute renal failure (%) |
25 |
34 |
<0.001 |
|
Delay of IMV (day) |
12 |
11 |
0.82 |
|
Mortality (%) |
28 |
64 |
0.02 |
Table 5.Independent factor of mortality of the study group
|
Factor |
P-value |
OR |
Minimum |
Maximum |
|
Lack of awake PP |
0.03 |
3.23 |
1.61 |
4.22 |
|
Nosocomial infection |
0.02 |
4.54 |
3.24 |
10.01 |
|
Septic shock |
0.04 |
32.21 |
2.89 |
100.02 |
|
Acute renal failure |
0.01 |
18.86 |
2.46 |
142.01 |
References
- 1. Li J, Luo J, Pavlov I, Perez Y, Tan W, Roca O, et al. Awake prone positioning for non-intubated patients with COVID-19-related acute hypoxaemic respiratory failure: a systematic review and meta-analysis. Lancet Respir Med 2022;10:573-83.PubMedPMC
- 2. Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med 2013;369:840-51.ArticlePubMed
- 3. Lopes JA, Jorge S. The RIFLE and AKIN classifications for acute kidney injury: a critical and comprehensive review. Clin Kidney J 2013;6:8-14.ArticlePubMed
- 4. Sikora A, Zahra F. Nosocomial infections [Internet]. StatPearls Publishing. 2023;[cited 2023 Jun 1]. Available from: https://pubmed.ncbi.nlm.nih.gov/32644738/.
- 5. Scaravilli V, Grasselli G, Castagna L, Zanella A, Isgrò S, Lucchini A, et al. Prone positioning improves oxygenation in spontaneously breathing nonintubated patients with hypoxemic acute respiratory failure: a retrospective study. J Crit Care 2015;30:1390-4.ArticlePubMed
- 6. Chalmers JD, Crichton ML, Goeminne PC, Cao B, Humbert M, Shteinberg M, et al. Management of hospitalised adults with coronavirus disease 2019 (COVID-19): European Respiratory Society living guideline. Eur Respir J 2021;57:2100048. ArticlePubMedPMC
- 7. Nasa P, Azoulay E, Khanna AK, Jain R, Gupta S, Javeri Y, et al. Expert consensus statements for the management of COVID-19-related acute respiratory failure using a Delphi method. Crit Care 2021;25:106. ArticlePubMedPMCPDF
- 8. Serpa Neto A, Checkley W, Sivakorn C, Hashmi M, Papali A, Schultz MJ, et al. Pragmatic recommendations for the management of acute respiratory failure and mechanical ventilation in patients with COVID-19 in low- and middle-income countries. Am J Trop Med Hyg 2021;104(3_Suppl):60-71.PMC
- 9. Gattinoni L, Pesenti A. The concept of “baby lung”. Intensive Care Med 2005;31:776-84.ArticlePubMedPDF
- 10. Gattinoni L, Pelosi P, Vitale G, Pesenti A, D'Andrea L, Mascheroni D. Body position changes redistribute lung computed-tomographic density in patients with acute respiratory failure. Anesthesiology 1991;74:15-23.ArticlePubMedPDF
- 11. Bone RC. The ARDS lung. New insights from computed tomography. JAMA 1993;269:2134-5.ArticlePubMed
- 12. Albert RK, Leasa D, Sanderson M, Robertson HT, Hlastala MP. The prone position improves arterial oxygenation and reduces shunt in oleic-acid-induced acute lung injury. Am Rev Respir Dis 1987;135:628-33.PubMed
- 13. Albert RK, Hubmayr RD. The prone position eliminates compression of the lungs by the heart. Am J Respir Crit Care Med 2000;161:1660-5.ArticlePubMed
- 14. Lamm WJ, Graham MM, Albert RK. Mechanism by which the prone position improves oxygenation in acute lung injury. Am J Respir Crit Care Med 1994;150:184-93.ArticlePubMed
- 15. Sartini C, Tresoldi M, Scarpellini P, Tettamanti A, Carcò F, Landoni G, et al. Respiratory parameters in patients with COVID-19 after using noninvasive ventilation in the prone position outside the intensive care unit. JAMA 2020;323:2338-40.ArticlePubMedPMC
- 16. Coppo A, Bellani G, Winterton D, Di Pierro M, Soria A, Faverio P, et al. Feasibility and physiological effects of prone positioning in non-intubated patients with acute respiratory failure due to COVID-19 (PRON-COVID): a prospective cohort study. Lancet Respir Med 2020;8:765-74.ArticlePubMedPMC
- 17. Caputo ND, Strayer RJ, Levitan R. Early self-proning in awake, non-intubated patients in the emergency department: a single ED’s experience during the COVID-19 pandemic. Acad Emerg Med 2020;27:375-8.ArticlePubMedPMCPDF
- 18. Elharrar X, Trigui Y, Dols AM, Touchon F, Martinez S, Prud’homme E, et al. Use of prone positioning in nonintubated patients with COVID-19 and hypoxemic acute respiratory failure. JAMA 2020;323:2336-8.ArticlePubMedPMC
- 19. Despres C, Brunin Y, Berthier F, Pili-Floury S, Besch G. Prone positioning combined with high-flow nasal or conventional oxygen therapy in severe COVID-19 patients. Crit Care 2020;24:256. ArticlePubMedPMCPDF
- 20. Paul V, Patel S, Royse M, Odish M, Malhotra A, Koenig S. Proning in non-intubated (PINI) in times of COVID-19: case series and a review. J Intensive Care Med 2020;35:818-24.ArticlePubMedPMCPDF
- 21. Chua EX, Zahir SM, Ng KT, Teoh WY, Hasan MS, Ruslan SR, et al. Effect of prone versus supine position in COVID-19 patients: a systematic review and meta-analysis. J Clin Anesth 2021;74:110406. ArticlePubMedPMC
- 22. Bahloul M, Kharrat S, Hafdhi M, Maalla A, Turki O, Chtara K, et al. Impact of prone position on outcomes of COVID-19 patients with spontaneous breathing. Acute Crit Care 2021;36:208-14.ArticlePubMedPMCPDF
Citations
Citations to this article as recorded by

 , Fatma Essafi1,2, Imen Talik1, Najla Ben Slimene1,2, Ines Sdiri1, Boudour Ben Dhia1, Takoua Merhbene1,2
, Fatma Essafi1,2, Imen Talik1, Najla Ben Slimene1,2, Ines Sdiri1, Boudour Ben Dhia1, Takoua Merhbene1,2 



 KSCCM
KSCCM

 PubReader
PubReader ePub Link
ePub Link Cite
Cite