Articles
- Page Path
- HOME > Acute Crit Care > Volume 38(3); 2023 > Article
-
Original Article
Pediatrics Impact of the COVID-19 pandemic on diabetic ketoacidosis management in the pediatric intensive care unit -
Fevzi Kahveci1
 , Buse Önen Ocak2
, Buse Önen Ocak2 , Emrah Gün1
, Emrah Gün1 , Anar Gurbanov1
, Anar Gurbanov1 , Hacer Uçmak1
, Hacer Uçmak1 , Ayşen Durak Aslan2
, Ayşen Durak Aslan2 , Ayşegül Ceran3
, Ayşegül Ceran3 , Hasan Özen1
, Hasan Özen1 , Burak Balaban1
, Burak Balaban1 , Edin Botan1
, Edin Botan1 , Zeynep Şıklar3
, Zeynep Şıklar3 , Merih Berberoğlu3
, Merih Berberoğlu3 , Tanıl Kendirli1
, Tanıl Kendirli1
-
Acute and Critical Care 2023;38(3):371-379.
DOI: https://doi.org/10.4266/acc.2023.00038
Published online: August 31, 2023
1Division of Pediatric Intensive Care, Department of Pediatrics, Ankara University Faculty of Medicine, Ankara, Türkiye
2Department of Pediatrics, Ankara University Faculty of Medicine, Ankara, Türkiye
3Division of Pediatric Endocrinology, Department of Pediatrics, Ankara University Faculty of Medicine, Ankara, Türkiye
- Corresponding author: Fevzi Kahveci Division of Pediatric Intensive Care, Department of Pediatrics, Ankara University Faculty of Medicine, Ankara 06100, Türkiye Tel: +90-532-0631836 Email: fevziikahvecii@gmail.com
Copyright © 2023 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Diabetic ketoacidosis (DKA) is a common endocrine emergency in pediatric patients. Early presentation to health facilities, diagnosis, and good management in the pediatric intensive care unit (PICU) are crucial for better outcomes in children with DKA.
-
Methods
- This was a single-center, retrospective cohort study conducted between February 2015 and January 2022. Patients with DKA were divided into two groups according to pandemic status and diabetes diagnosis.
-
Results
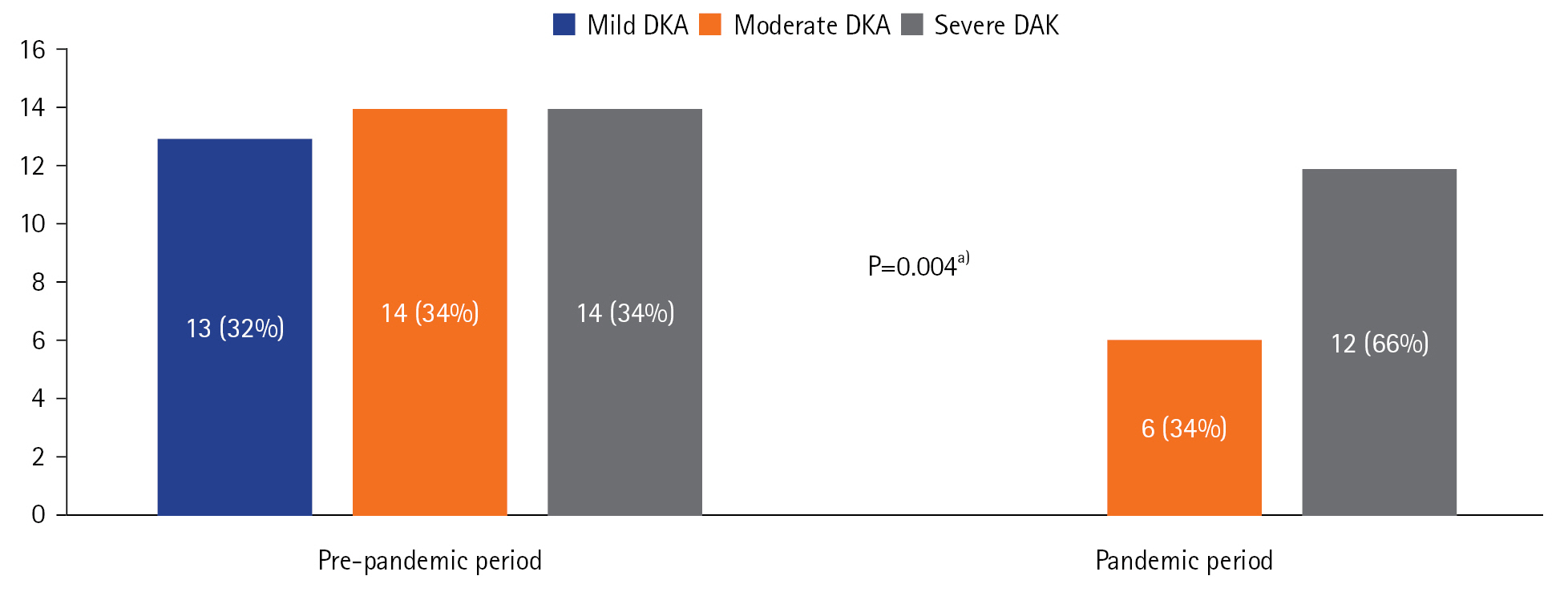
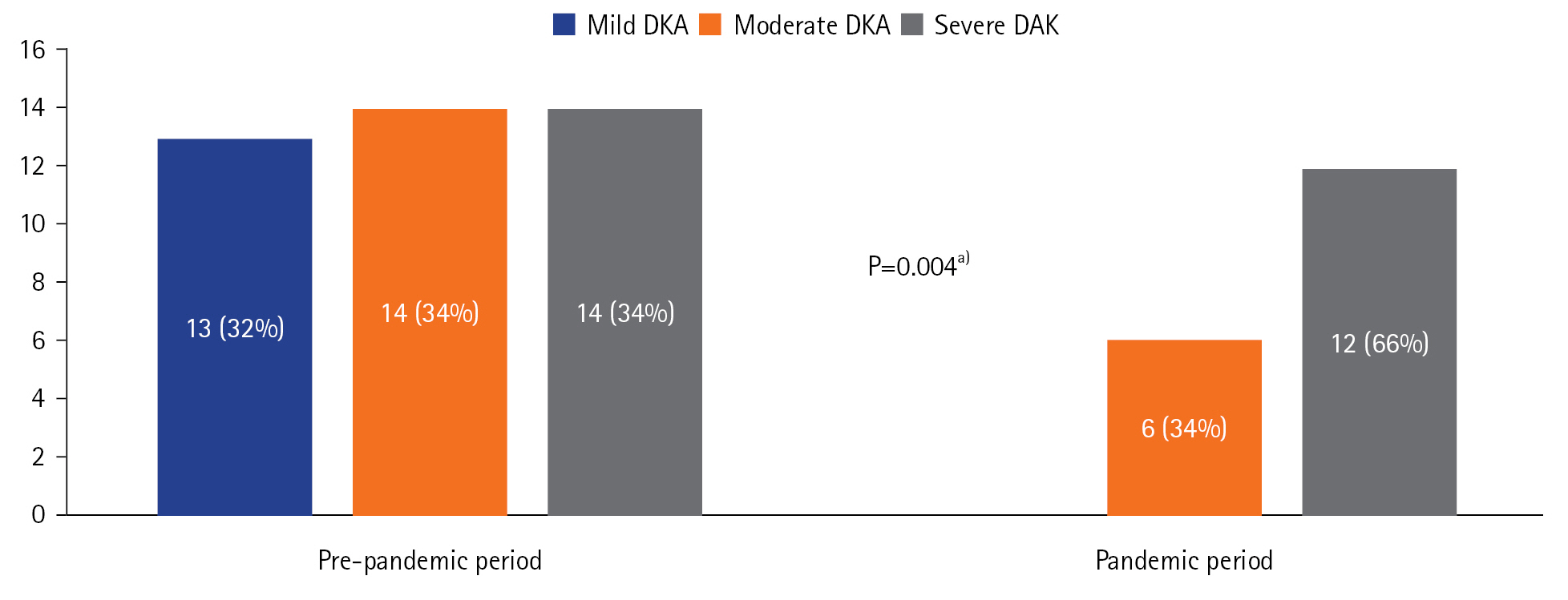
- The study enrolled 59 patients, and their mean age was 11±5 years. Forty (68%) had newly diagnosed type 1 diabetes mellitus (T1DM), and 61% received follow-up in the pre-pandemic period. Blood glucose, blood ketone, potassium, phosphorus, and creatinine levels were significantly higher in the new-onset T1DM group compared with the previously diagnosed group (P=0.01, P=0.02, P<0.001, P=0.01, and P=0.08, respectively). In patients with newly diagnosed T1DM, length of PICU stays were longer than in those with previously diagnosed T1DM (28.5±8.9 vs. 17.3±6.7 hours, p<0.001). The pandemic group was compared with pre-pandemic group, there was a statistically significant difference in laboratory parameters of pH, HCO3, and lactate and also Pediatric Risk of Mortality (PRISM) III score. All patients survived, and there were no neurologic sequelae.
-
Conclusion
- Patients admitted during the pandemic period were admitted with more severe DKA and had higher PRISM III scores. During the pandemic period, there was an increase in the incidence of DKA in the participating center compared to that before the pandemic.
INTRODUCTION
MATERIALS AND METHODS
RESULTS
DISCUSSION
KEY MESSAGES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
FUNDING
None.
-
AUTHOR CONTRIBUTIONS
Conceptualization: FK, AG, AC, HO, EB. Methodology: FK, EG, ADA, AC, BB, EB. Validation: FK. Formal analysis: FK, EG, AG, HU, BB, ZS, MB. Data curation: FK, BOO, EG, HU, ADA, AC, HO, EB, ZS, TK. Project administration: ZS, MB, TK. Writing–original draft: FK. Writing–review & editing: ZŞ, TK.
NOTES
Acknowledgments
| Parameter | Total patient |
Diabetes diagnosis status |
P-value | |
|---|---|---|---|---|
| Previously diagnosed (n=19) | Newly diagnosed (n=40) | |||
| Female | 37 (62.7) | 12 (20.3) | 25 (42.4) | 0.96a) |
| Age (yr) | 11±5 | 14.3±2.5 | 11.4±3.1 | <0.001b) |
| Diabetes diagnosis age (yr) | 8.98±4.3 | 8.3±3.7 | 9.2±4.6 | 0.47c) |
| Height (cm) | 149±26.4 | 161.5±15 | 152±15 | <0.001b) |
| BMI (kg/m2) | 16.8±4.6 | 20.0±3.7 | 16.5±5.2 | 0.003b) |
| Height z-score | 0.38 (–6.6 to 4.0) | 0.14 (–3.2 to 1.6) | 0.54 (–6.6 to 4.0) | 0.21b) |
| BMI z-score | –0.28 (–4.3 to 3.1) | 0.03 (–2.5 to 2.2) | –0.66 (–4.3 to 3.1) | 0.15b) |
| Symptom duration (day) | 7.0±10.3 | 2.5±3.4 | 14.0±11.0 | <0.001b) |
| PICU length of stay (hr) | 25.0±9.7 | 17.3±6.7 | 28.5±8.9 | <0.001c) |
| Hospital stay (day) | 9.0±4.0 | 5.0±2.8 | 9.0±3.4 | <0.001b) |
| GCS | 15.0±2.4 | 13.5±2.6 | 15.0±2.5 | 0.39b) |
| PRISM III score | 8.0±2.1 | 8.0±2.3 | 8.0±2.1 | 0.80b) |
| Pandemic status (pre-pandemic), yes | 41 (69.5) | 12 (20.3) | 29 (49.2) | 0.46a) |
| Total saline bolus (ml/kg) | 10.0±7.2 | 10.0±4.2 | 10.0±6.2 | 0.23b) |
| Using crystalline insulin (hr) | 20.0±8.8 | 17.8±14 | 20.7±5.1 | 0.24c) |
| Polydipsia, yes | 27 (45.8) | 3 (5.1) | 24 (40.7) | <0.001a) |
| Polyuria, yes | 27 (45.8) | 3 (5.1) | 24 (40.7) | <0.001a) |
| Vomiting, yes | 23 (38.9) | 13 (22.0) | 10 (16.9) | <0.001a) |
| Stomachache, yes | 7 (11.9) | 6 (10.2) | 1 (1.7) | 0.002a) |
| Blood glucose (mg/dl) | 466±129 | 517±102 | 431±132 | 0.01c) |
| Sodium (mmol/L) | 134.3±4.2 | 134.0±4.6 | 134±4 | 0.92c) |
| Potassium (mmol/L) | 4.2±0.8 | 4.7±0.7 | 4.0±0.8 | <0.001c) |
| Phosphorus (mg/dl) | 3.7±1.5 | 4.9±1.9 | 3.5±1.2 | 0.01b) |
| Creatine (mg/dl) | 0.8±0.3 | 0.9±0.3 | 0.7±0.3 | 0.08c) |
| Blood ketone (mmol/L) | 6.2±1.1 | 6.0±1.2 | 6.9±1.0 | 0.02b) |
Values are presented as number (%), mean±standard deviation, or median (range).
DKA: diabetic ketoacidosis; BMI: body mass index; PICU: pediatric intensive care unit; GCS: Glasgow coma score; PRISM: Pediatric Risk of Mortality.
a) Chi-square test;
b) Mann-Whitney U-test;
c) Independent samples t-test.
| Parameter | Total patients |
Pandemic status |
P-value | |
|---|---|---|---|---|
| Pre-pandemic (n=41) | Pandemic (n=18) | |||
| Newly diagnosed | 40 (100) | 29 (72.5) | 11 (27.5) | 0.33a) |
| Female | 37 (62.7) | 25 (42.2) | 12 (20.3) | 0.67a) |
| Age (yr) | 11±5 | 11±5 | 12±5 | 0.45b) |
| Diabetes diagnosis age (yr) | 9±4 | 9±4 | 9±4 | 0.74b) |
| Height (cm) | 149±27 | 143±26 | 148±27 | 0.51b) |
| BMI (kg/m2) | 16.8±4.6 | 17.9±4.3 | 18.2±5.4 | 0.94b) |
| Height z-score | 0.39 (–6.6 to 4.0) | 0.1 (–6.1 to 4.0) | 0.5 (–6.6 to 3.9) | 0.66c) |
| BMI z-score | –0.4±1.7 | –0.3±1.6 | –0.8±1.9 | 0.36b) |
| Body weight (kg) | 40±19 | 38±19 | 42±20 | 0.53b) |
| Symptom duration (day) | 7.0±10.3 | 7.0±9.9 | 6.0±11.4 | 0.54c) |
| PICU length of stay (hr) | 25.0±9.7 | 25.5±10.1 | 23.6±8.9 | 0.51b) |
| Hospital stay (day) | 9±4 | 9±4 | 8±4 | 0.18c) |
| GCS | 15.0±2.4 | 13.5±2.5 | 12.8±2.3 | 0.22b) |
| PRISM III score | 8.7±2.1 | 8.0±2.0 | 10.0±2.0 | 0.02c) |
| Total saline bolus (ml/kg) | 10±7 | 10±8 | 10±6 | 0.79c) |
| Blood glucose (mg/dl) | 466±129 | 452±139 | 491±103 | 0.35b) |
| HbA1c (%) | 11.8±3.3 | 11.8±3.4 | 11.8±3.0 | 0.91b) |
| pH | 7.10±0.13 | 7.13±0.13 | 7.03±0.10 | 0.01c) |
| pCO2 | 21.3±11.3 | 21.5±12.7 | 17.2±5.4 | 0.09b) |
| HCO3 | 7.0±3.8 | 8.4±4 | 4.2±2.4 | 0.01c) |
| Lactate | 2.0±1.4 | 2.4±1.7 | 1.9±1.3 | 0.01b) |
| Leukocyte (×109/L) | 14,584±8,017 | 16,672±9,445 | 12,970±7,087 | 0.003b) |
| Neutrophil (×109/L) | 10,047±7,823 | 12,300±8,853 | 8,211±6,915 | 0.02b) |
Values are presented as number (%), mean±standard deviation, or median (range).
DKA: diabetic ketoacidosis; BMI: body mass index; PICU: pediatric intensive care unit; GCS: Glasgow coma score; PRISM: Pediatric Risk of Mortality.
a) Chi-square test;
b) Independent samples t-test;
c) Mann-Whitney U-test.
- 1. von Saint Andre-von Arnim A, Farris R, Roberts JS, Yanay O, Brogan TV, Zimmerman JJ. Common endocrine issues in the pediatric intensive care unit. Crit Care Clin 2013;29:335-58.ArticlePubMed
- 2. Sherry NA, Levitsky LL. Management of diabetic ketoacidosis in children and adolescents. Paediatr Drugs 2008;10:209-15.ArticlePubMed
- 3. Ganesh R, Suresh N, Ramesh J. Diabetic ketoacidosis in children. Natl Med J India 2006;19:155-8.PubMed
- 4. Jayashree M, Singhi S. Diabetic ketoacidosis: predictors of outcome in a pediatric intensive care unit of a developing country. Pediatr Crit Care Med 2004;5:427-33.ArticlePubMed
- 5. Wolfsdorf JI, Allgrove J, Craig ME, Edge J, Glaser N, Jain V, et al. ISPAD Clinical Practice Consensus Guidelines 2014: diabetic ketoacidosis and hyperglycemic hyperosmolar state. Pediatr Diabetes 2014;15 Suppl 20:154-79.PubMed
- 6. Castellanos L, Tuffaha M, Koren D, Levitsky LL. Management of diabetic ketoacidosis in children and adolescents with type 1 diabetes mellitus. Paediatr Drugs 2020;22:357-67.ArticlePubMedPDF
- 7. Del Pozo P, Aránguiz D, Córdova G, Scheu C, Valle P, Cerda J, et al. Clinical profile of children with diabetic ketoacidosis in fifteen years of management in a Critical Care Unit. Rev Chil Pediatr 2018;89:491-8.PubMed
- 8. Çocuk Endokrinoloji ve Diyabet Derneği. Child diabetes group diabetic ketoacidosis (DKA) treatment and follow-up guide [Internet]. Çocuk Endokrinoloji ve Diyabet Derneği; 2016 [cited 2023 May 10]. Available from : https://www.cocukendokrindiyabet.org/uploads/dokumanlar/9UtrSqWB3LMvcHyPLJw9.pdf.
- 9. Pollack MM, Patel KM, Ruttimann UE. PRISM III: an updated Pediatric Risk of Mortality score. Crit Care Med 1996;24:743-52.PubMed
- 10. Lopes CL, Pinheiro PP, Barberena LS, Eckert GU. Diabetic ketoacidosis in a pediatric intensive care unit. J Pediatr (Rio J) 2017;93:179-84.ArticlePubMed
- 11. Baloch SH, Ibrahim PM, Lohano PD, Gowa MA, Mahar S, Memon R. Pediatric risk of mortality III score in predicting mortality among diabetic ketoacidosis patients in a pediatric ıntensive care unit. Cureus 2021;13:e19734.PubMedPMC
- 12. Alfayez OM, Aldmasi KS, Alruwais NH, Bin Awad NM, Al Yami MS, Almohammed OA, et al. Incidence of diabetic ketoacidosis among pediatrics with type 1 diabetes prior to and during COVID-19 pandemic: a meta-analysis of observational studies. Front Endocrinol (Lausanne) 2022;13:856958. ArticlePubMedPMC
- 13. Apicella M, Campopiano MC, Mantuano M, Mazoni L, Coppelli A, Del Prato S. COVID-19 in people with diabetes: understanding the reasons for worse outcomes. Lancet Diabetes Endocrinol 2020;8:782-92.ArticlePubMedPMC
- 14. Chee YJ, Ng SJH, Yeoh E. Diabetic ketoacidosis precipitated by Covid-19 in a patient with newly diagnosed diabetes mellitus. Diabetes Res Clin Pract 2020;164:108166. ArticlePubMedPMC
- 15. Li J, Wang X, Chen J, Zuo X, Zhang H, Deng A. COVID-19 infection may cause ketosis and ketoacidosis. Diabetes Obes Metab 2020;22:1935-41.ArticlePubMedPMCPDF
- 16. Rubino F, Amiel SA, Zimmet P, Alberti G, Bornstein S, Eckel RH, et al. New-onset diabetes in Covid-19. N Engl J Med 2020;383:789-90.ArticlePubMedPMC
- 17. Glaser N, Barnett P, McCaslin I, Nelson D, Trainor J, Louie J, et al. Risk factors for cerebral edema in children with diabetic ketoacidosis. N Engl J Med 2001;344:264-9.ArticlePubMed
- 18. Lawrence SE, Cummings EA, Gaboury I, Daneman D. Population-based study of incidence and risk factors for cerebral edema in pediatric diabetic ketoacidosis. J Pediatr 2005;146:688-92.ArticlePubMed
- 19. Ghetti S, Kuppermann N, Rewers A, Myers SR, Schunk JE, Stoner MJ, et al. Cognitive function following diabetic ketoacidosis in children with new-onset or previously diagnosed type 1 diabetes. Diabetes Care 2020;43:2768-75.ArticlePubMedPMCPDF
- 20. Durward A, Ferguson LP, Taylor D, Murdoch IA, Tibby SM. The temporal relationship between glucose-corrected serum sodium and neurological status in severe diabetic ketoacidosis. Arch Dis Child 2011;96:50-7.ArticlePubMed
- 21. Lone SW, Siddiqui EU, Muhammed F, Atta I, Ibrahim MN, Raza J. Frequency, clinical characteristics and outcome of diabetic ketoacidosis in children with type-1 diabetes at a tertiary care hospital. J Pak Med Assoc 2010;60:725-9.PubMed
- 22. Chambers MA, Mecham C, Arreola EV, Sinha M. Increase in the number of pediatric new-onset diabetes and diabetic ketoacidosis cases during the COVID-19 pandemic. Endocr Pract 2022;28:479-85.ArticlePubMedPMC
References
Figure & Data
References
Citations

- Covid 19 and diabetes in children: advances and strategies
Zhaoyuan Wu, Jinling Wang, Rahim Ullah, Minghao Chen, Ke Huang, Guanping Dong, Junfen Fu
Diabetology & Metabolic Syndrome.2024;[Epub] CrossRef

 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite