Abstract
- Myoclonic status epilepticus (MSE) is a sign of severe neurologic injury in cardiac arrest patients. To our knowledge, MSE has not been described as a result of prolonged hyperpyrexia. A 56-year-old man with coronavirus disease 2019 presented with acute respiratory distress syndrome, septic/hypovolemic shock, and presumed community-acquired pneumonia. Five days after presentation, he developed a sustained fever of 42.1°C that did not respond to acetaminophen or ice water gastric lavage. After several hours, he was placed on surface cooling. Three hours after fever resolution, new multifocal myoclonus was noted in the patient’s arms and trunk. Electroencephalography showed midline spikes consistent with MSE, which resolved with 40 mg/kg of levetiracetam. This case demonstrates that severe hyperthermia can cause cortical injury significant enough to trigger MSE and should be treated emergently using the most aggressive measures available. Providers should have a low threshold for electroencephalography in intubated patients with a recent history of hyperpyrexia.
-
Keywords: brain injury; coronavirus disease 2019; fever; hyperthermia; myoclonus; status epilepticus
Myoclonic status epilepticus (MSE) is most frequently associated with cardiac arrest and is often considered a sign of severe neurologic injury [1-3]. To our knowledge, MSE has not previously been described as a result of hyperpyrexia. Here we present the case of a man with coronavirus disease 2019 (COVID-19) who developed MSE following prolonged hyperpyrexia. This study was not obtained IRB approval because the patient was not enrolled in a study. Verbal consent to publish the case report was obtained from the patient's next of kin.
CASE REPORT
A 56-year-old man with a history of hypertension and poorly controlled diabetes mellitus complicated by chronic osteomyelitis presented to the hospital with 1 week of lethargy and poor appetite and 2 days of diarrhea. He was febrile at 38.3°C, had a heart rate of 150 to 180 bpm, and SpO2 of 87% on a non-rebreather. Nasopharyngeal polymerase chain reaction testing returned a positive result for COVID-19. One hour after hospital arrival, he became acutely dyspneic and hypoxic. He was intubated for hypoxemic respiratory failure and treated for acute respiratory distress syndrome, mixed septic/hypovolemic shock, and presumed community-acquired pneumonia. He was administered dexamethasone, remdesivir, ceftriaxone, vancomycin, and doxycycline.
Five days after his initial presentation, the patient became febrile at a maximum temperature of 42.1°C with new leukocytosis of 24,700/µL, up from 7,600/µL. He was given acetaminophen and ice water gastric lavage and transferred to surface cooling after several hours given a lack of response to the aforementioned measures. He sustained a fever above 41°C for 6 total hours. His creatine kinase concentration was 1,763 units/L, up from 301 units/L, and results of urinalysis and blood culture were unremarkable. Treatment was broadened to include vancomycin, ceftriaxone, and ampicillin for empiric central nervous system (CNS) coverage. Lumbar puncture performed during his fever showed an opening pressure of 25 mm Hg, three erythrocytes, two nucleated cells, a protein concentration of 40.7 mg/dL, and a glucose level of 143 mg/dl (serum glucose level, 211 mg/dl). Chest X-ray showed diffuse pulmonary opacities, unchanged from prior imaging. The lowest recorded blood oxygen level (SpO2) was 92%. His sputum culture ultimately grew Stenotrophomonas, and he was placed on a trimethoprim and sulfamethoxazole combination regimen.
Three hours after his fever resolved, the patient showed new multifocal myoclonus in his arms and trunk. He was unresponsive to painful stimuli. Arterial blood gas was notable at pH 7.4, partial pressure of carbon dioxide of 39 mm Hg, and partial pressure of oxygen of 130 mm Hg. Electroencephalography showed midline spikes consistent with MSE (Figure 1), which resolved with 40 mg/kg of levetiracetam. He was maintained on 750 mg of levetiracetam twice daily and was treated with acetaminophen for fever suppression and surface cooling with a target temperature of 36°C due to a concern for cortical injury related to his hyperpyrexia. Surface cooling was discontinued after 72 hours. Neuron-specific enolase peaked at 81.3 ng/ml the day after the fever and returned to within the normal range 2 days later.
Magnetic resonance imaging of the brain was not performed until 12 days post-fever due to patient instability and did not demonstrate any signs of cortical or metabolic injury (Figure 2). Forty days after his episode of MSE, the patient began following simple commands with his eyes, but he experienced no further neurologic recovery. His course was complicated by multiorgan failure and multiple brain infarcts, and he passed away 75 days after hospital admission.
DISCUSSION
MSE has been reported in diverse settings, including metabolic encephalopathy, degenerative CNS disorders, generalized epilepsy, and drug reactions [3,4]. However, it is most frequently associated with cardiac arrest [1-3]. MSE in cardiac arrest patients is considered a poor prognostic indicator and is associated with a high fatality rate [1-3]. Despite its grim prognosis, there are several case reports of recovery following MSE [3,5]. Unfortunately, our patient experienced minimal neurologic recovery and passed away following a prolonged hospital stay. MSE is frequently refractory to anti-epileptic therapy, which diverges from the experience in our patient, who responded well to levetiracetam [1-3].
Given his new leukocytosis at the time of fever and the sputum culture positive for Stenotrophomonas, our patient’s hyperpyrexia was likely secondary to pneumonia. Previous case reports have described MSE secondary to COVID-19 pneumonia and anoxia [3,5]. Although it is possible that the MSE in this patient was caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection or pneumonia, our patient’s normal SpO2 and arterial blood gas values together with lack of other metabolic derangements and the tight temporal correlation between fever and MSE onset all suggest that hyperpyrexia was the primary cause.
To our knowledge, MSE has not previously been described as a result of prolonged hyperpyrexia. Nonetheless, there is increasing recognition that hyperthermia of any etiology can cause neurologic injury [6,7]. Hyperthermia-induced injury is most common in the cerebellum but can affect many components of the CNS, including the basal ganglia, brainstem, and cerebral cortex [6]. The mechanism of neurologic injury is likely multifactorial. Hyperthermia has been shown to directly affect neurons through variable mechanisms, including denaturation, apoptosis, mitochondrial damage, and excitotoxicity from elevated levels of excitatory neurotransmitters in vitro and in animal models [6,7]. Other potential mechanisms demonstrated in animal and human models include disruption of the blood–brain barrier, changes in cerebral blood flow, and inflammatory cytokine response [6,8,9].
The pro-inflammatory cytokine response might be of particular importance in patients with COVID-19. Both infection by SARS-CoV-2 and hyperthermia cause a pro-inflammatory cascade that leads to the release of inflammatory cytokines, including tumor necrosis factor, interleukin (IL)-1, and IL-6 [6,8-10]. An elevated IL-6 concentration in particular is associated with a greater severity of symptoms in patients with heat stroke or COVID-19 [6,8-10]. Our patient likely suffered a double burden of cytokine release from his underlying SARS-CoV-2 infection and his high fever.
This case demonstrates that severe hyperthermia can cause cortical injury significant enough to trigger MSE. Hyperthermia to the extent seen in our patient should be treated emergently with the most aggressive measures available due to the possibility of devastating neurological injury. Our patient ultimately responded to surface cooling, which was initiated only after failure of more conservative procedures. Finally, providers should have a low threshold for electroencephalography in intubated patients with a recent history of severe hyperthermia.
NOTES
-
CONFLICT OF INTEREST No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conceptualization: SMS. Investigation: all authors. Data Curation: KAH, JJP. Writing–original draft: KAH. Writing–review & editing: all authors.
-
FUNDING
None.
-
ACKNOWLEDGMENTS
Consent to publish the case report was obtained from the patient's next of kin.
Figure 1.Electroencephalogram taken after fever resolution shows midline spikes.
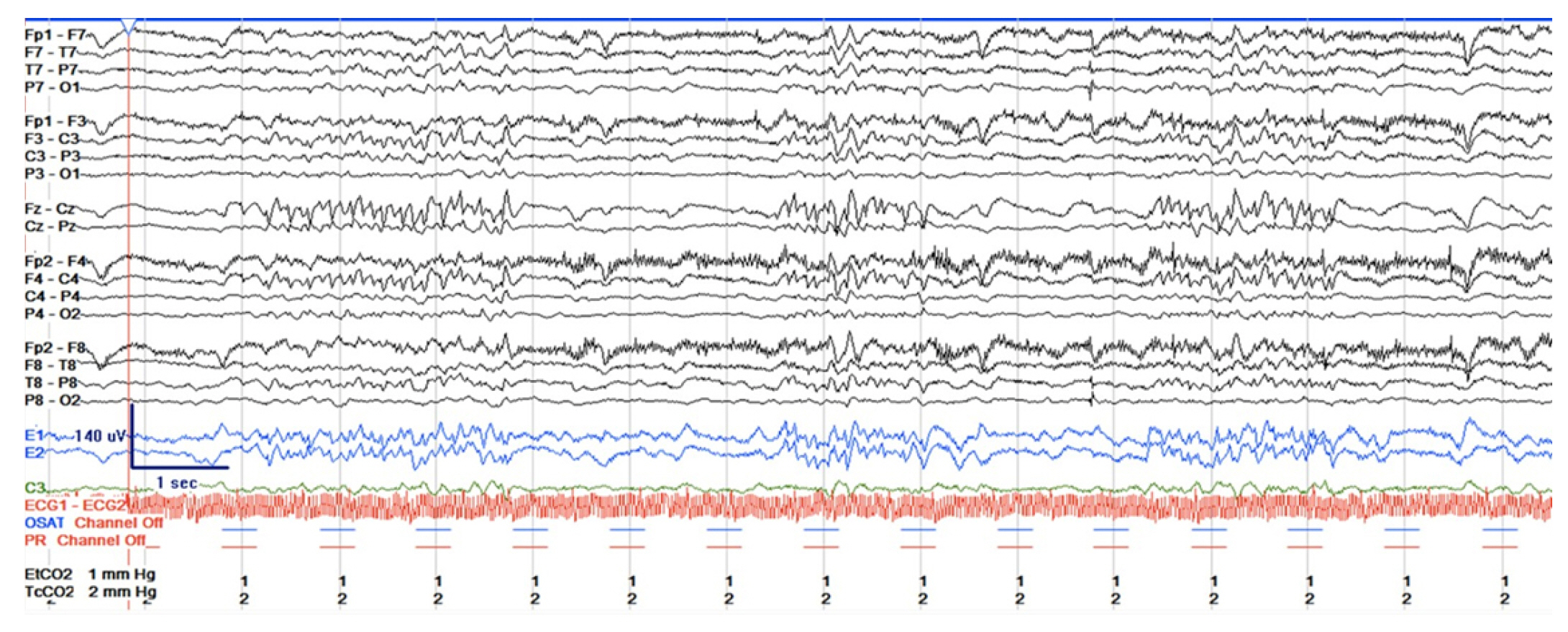
Figure 2.(A, B) Brain magnetic resonance imaging taken 12 days after fever resolution did not demonstrate cortical or metabolic injury.

References
- 1. Wijdicks EF, Parisi JE, Sharbrough FW. Prognostic value of myoclonus status in comatose survivors of cardiac arrest. Ann Neurol 1994;35:239-43.ArticlePubMed
- 2. Hui AC, Cheng C, Lam A, Mok V, Joynt GM. Prognosis following postanoxic myoclonus status epilepticus. Eur Neurol 2005;54:10-3.ArticlePubMed
- 3. Celesia GG, Grigg MM, Ross E. Generalized status myoclonicus in acute anoxic and toxic-metabolic encephalopathies. Arch Neurol 1988;45:781-4.ArticlePubMed
- 4. Baysal Kirac L, Aydogdu I, Acarer A, Alpaydin S, Bayam FE, Onbasi H, et al. Myoclonic status epilepticus in six patients without epilepsy. Epilepsy Behav Case Rep 2012;1:10-3.ArticlePubMedPMC
- 5. Chen W, Toprani S, Werbaneth K, Falco-Walter J. Status epilepticus and other EEG findings in patients with COVID-19: a case series. Seizure 2020;81:198-200.ArticlePubMedPMC
- 6. Walter EJ, Carraretto M. The neurological and cognitive consequences of hyperthermia. Crit Care 2016;20:199. ArticlePubMedPMC
- 7. White MG, Luca LE, Nonner D, Saleh O, Hu B, Barrett EF, et al. Cellular mechanisms of neuronal damage from hyperthermia. Prog Brain Res 2007;162:347-71.ArticlePubMed
- 8. Hashim IA, Al-Zeer A, Al-Shohaib S, Al-Ahwal M, Shenkin A. Cytokine changes in patients with heatstroke during pilgrimage to Makkah. Mediators Inflamm 1997;6:135-9.ArticlePubMedPMC
- 9. Hammami MM, Bouchama A, Al-Sedairy S, Shail E, AlOhaly Y, Mohamed GE. Concentrations of soluble tumor necrosis factor and interleukin-6 receptors in heatstroke and heatstress. Crit Care Med 1997;25:1314-9.ArticlePubMed
- 10. Narula N, Joseph R, Katyal N, Daouk A, Acharya S, Avula A, et al. Seizure and COVID-19: association and review of potential mechanism. Neurol Psychiatry Brain Res 2020;38:49-53.ArticlePubMedPMC
Citations
Citations to this article as recorded by

 , John J Peters2
, John J Peters2 , Sara M Schaefer2
, Sara M Schaefer2





 KSCCM
KSCCM

 PubReader
PubReader ePub Link
ePub Link Cite
Cite



