Articles
- Page Path
- HOME > Acute Crit Care > Volume 37(2); 2022 > Article
-
Original Article
Pulmonary Cinematic virtual reality for anxiety management in mechanically ventilated patients: a feasibility and pilot study -
Alexander C. Haley1
 , David A. Wacker2
, David A. Wacker2
-
Acute and Critical Care 2022;37(2):230-236.
DOI: https://doi.org/10.4266/acc.2021.00843
Published online: February 4, 2022
1Integrative Health and Wellbeing Research Program at the Earl E. Bakken Center for Spirituality and Healing, University of Minnesota School of Nursing, Minneapolis, MN, USA
2Division of Pulmonary, Allergy, Critical Care and Sleep Medicine, Department of Internal Medicine, University of Minnesota Medical School, Minneapolis, MN, USA
- Corresponding author: David A. Wacker Division of Pulmonary, Allergy, Critical Care and Sleep Medicine, Department of Internal Medicine, University of Minnesota Medical School, 420 Delaware St SE, MMC 276, Minneapolis, MN 55455, USA Tel: +1-612-624-0999, Fax: +1-612-625-2174, E-mail: wack0012@umn.edu
Copyright © 2022 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Mechanically ventilated patients experience anxiety for many reasons. Pharmacological treatments such as benzodiazepines are commonly employed to manage anxiety; however, these therapies often cause undesired side effects. Additional therapies for anxiety management are needed. We sought to determine whether cell phone-based virtual reality therapy could feasibly be used for anxiety management in mechanically ventilated patients.
-
Methods
- Mechanically ventilated subjects underwent at least one session of virtual reality therapy in which they were shown a cinematic video of an outdoor green space or blue space with 360° visual range of motion. Goal session duration was 5 minutes. The primary outcome was incidence of predefined patient safety events, including self-extubation and accidental removal of tubes or lines.
-
Results
- Ten subjects underwent a total of 18 virtual reality sessions. Fifteen sessions lasted the planned 5 minutes, one session was extended at participant request, and two sessions were terminated early at participant request. There were no occurrences of the predefined safety events, and no occurrences of cybersickness. Use of a visual analog scale to measure anxiety level was feasible for this pilot study, demonstrating feasibility of this scale for future, larger scale studies.
-
Conclusions
- Virtual reality therapy shows potential as a means of managing anxiety in patients undergoing mechanical ventilation, and further rigorous exploration with this protocol is feasible.
INTRODUCTION
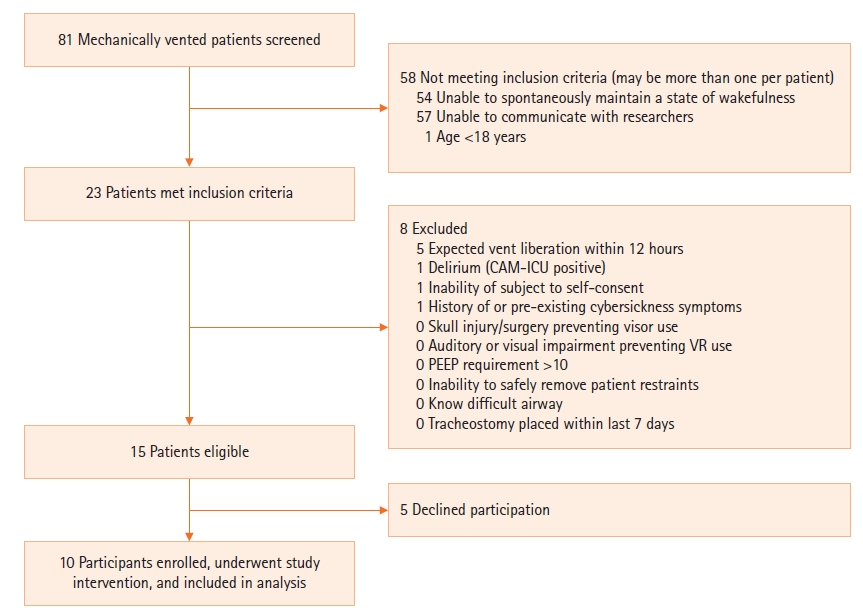
MATERIALS AND METHODS
RESULTS
DISCUSSION
KEY MESSAGE
-
CONFLICT OF INTEREST No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conceptualization: all authors. Data curation: DAW. Formal analysis: DAW. Funding acquisition: DAW. Methodology: all authors. Project administration: DAW. Visualization: ACH. Writing–original draft: all authors. Writing–review & editing: all authors.
NOTES
Acknowledgments

| Outcome | Value | P-valuea |
|---|---|---|
| Session during which a safety event occurred (n=18 sessions) | ||
| Cardiac arrhythmias (cardiac arrest or change to rhythm other than sinus or atrial fibrillation with rate <150) | 0 | |
| Hypotension (MAP <55) | 0 | |
| Hypertension (MAP >140) | 0 | |
| Oxygen desaturation (oxygen saturation < 85% for 3 minutes or greater) | 0 | |
| Fall from bed or chair | 0 | |
| Unintentional removal of medical device (lines, tubes, etc.) | 0 | |
| Session during which cybersickness occurred (n=18 sessions) | ||
| Subjective anxiety levels during session (100 point VAS-A scale) | ||
| Starting | 37 (19–74) | |
| Ending | 32 (7–64) | |
| Pairwise differenceb | –8.5 (–12.5 to –1.5) | 0.012 |
| Heart rate during session (beats/min) | ||
| Starting | 84 (71–88) | |
| Ending | 81 (72–79) | |
| Pairwise differenceb | 1 (–4 to 3) | 0.553 |
| Respiratory rate during session (beats/min) | ||
| Starting | 21 (18–26) | |
| Ending | 19 (17–24) | |
| Pairwise differenceb | –2 (–3 to 0) | 0.079 |
| Systolic blood pressure during session (mm Hg) | ||
| Starting | 130 (115–140) | |
| Ending | 118 (107–129) | |
| Pairwise differenceb | –9 (–17 to –1) | 0.003 |
| Diastolic blood pressure during session (mm Hg) | ||
| Starting | 70 (66–81) | |
| Ending | 67 (60–75) | |
| Pairwise differenceb | –4 (–12 to 1) | 0.040 |
| Reason for not performing planned sessions (n=5) | ||
| Subject preference or scheduling conflict with other therapies | 3 (60) | |
| Change in subject’s mental status | 1 (20) | |
| Symptom of cybersickness present prior to session start | 1 (20), dizziness |
Values are presented as median (interquartile range) or number (%).
MAP: mean arterial pressure; VAS-A: visual analog scale-anxiety.
a P-values obtained using the Wilcoxon signed-rank test to compare the pairwise differences in each variable to the null set;
b Pairwise differences were determined by subtracting the variable value at the beginning of a session from that at the end of the session for each session individually. Thus a positive value represents a net increase during the session, and a negative value a net decrease.
| Variable | Value (n=10) |
|---|---|
| Age (yr) | 58 (49–66) |
| Birth sexa | |
| Female | 5 (50) |
| Male | 5 (50) |
| Racea | |
| African American or Black | 2 (20) |
| Caucasian | 8 (80) |
| Ethnicity | |
| Hispanic or Latino | 0 |
| Not Hispanic or Latino | 10 (100) |
| Reason for ICU admission | |
| Respiratory failure, ARDS | 0 |
| Respiratory failure, COPD exacerbation | 1 (10) |
| Respiratory failure, pneumonia | 4 (40) |
| Respiratory failure, pulmonary edema | 0 |
| Respiratory failure, CLAD | 2 (20) |
| Respiratory failure, other | 2 (20) |
| Shock, septic | 1 (10) |
| Shock, non-septic | 0 |
| Total duration of ICU stay (day) | 10.5 (6–14) |
| SOFA score at enrollmentb | 3 (2–3) |
| Airway | |
| Endotracheal tube | 6 (60) |
| Tracheostomy | 4 (40) |
Values are presented as median (interquartile range) or number (%).
ICU: intensive care unit; ARDS: acute respiratory distress syndrome; COPD: chronic obstructive pulmonary disease; CLAD: chronic lung allograft dysfunction; SOFA: Sequential Organ Failure Assessment.
a As reported in the subject’s medical record;
b SOFA score lab parameters were missing for three subjects; these were assumed to be normal when calculating SOFA scores.
- 1. Pandharipande PP, Pun BT, Herr DL, Maze M, Girard TD, Miller RR, et al. Effect of sedation with dexmedetomidine vs lorazepam on acute brain dysfunction in mechanically ventilated patients: the MENDS randomized controlled trial. JAMA 2007;298:2644-53.ArticlePubMed
- 2. Ludmir J, Netzer G. Go big: measuring and tackling psychological morbidity after critical illness. Ann Am Thorac Soc 2016;13:1217-8.ArticlePubMed
- 3. Nikayin S, Rabiee A, Hashem MD, Huang M, Bienvenu OJ, Turnbull AE, et al. Anxiety symptoms in survivors of critical illness: a systematic review and meta-analysis. Gen Hosp Psychiatry 2016;43:23-29.ArticlePubMedPMC
- 4. Wunsch H, Christiansen CF, Johansen MB, Olsen M, Ali N, Angus DC, et al. Psychiatric diagnoses and psychoactive medication use among nonsurgical critically ill patients receiving mechanical ventilation. JAMA 2014;311:1133-42.ArticlePubMed
- 5. Parker AM, Sricharoenchai T, Raparla S, Schneck KW, Bienvenu OJ, Needham DM. Posttraumatic stress disorder in critical illness survivors: a metaanalysis. Crit Care Med 2015;May;43:1121-9.PubMed
- 6. Lee CH, Lee CY, Hsu MY, Lai CL, Sung YH, Lin CY, et al. Effects of music intervention on state anxiety and physiological indices in patients undergoing mechanical ventilation in the intensive care unit. Biol Res Nurs 2017;19:137-44.ArticlePubMed
- 7. Mofredj A, Alaya S, Tassaioust K, Bahloul H, Mrabet A. Music therapy, a review of the potential therapeutic benefits for the critically ill. J Crit Care 2016;35:195-9.ArticlePubMed
- 8. Chlan LL, Weinert CR, Heiderscheit A, Tracy MF, Skaar DJ, Guttormson JL, et al. Effects of patient-directed music intervention on anxiety and sedative exposure in critically ill patients receiving mechanical ventilatory support: a randomized clinical trial. JAMA 2013;309:2335-44.ArticlePubMedPMC
- 9. Faber AW, Patterson DR, Bremer M. Repeated use of immersive virtual reality therapy to control pain during wound dressing changes in pediatric and adult burn patients. J Burn Care Res 2013;34:563-8.ArticlePubMed
- 10. Markus LA, Willems KE, Maruna CC, Schmitz CL, Pellino TA, Wish JR, et al. Virtual reality: feasibility of implementation in a regional burn center. Burns 2009;35:967-9.ArticlePubMed
- 11. Small C, Stone R, Pilsbury J, Bowden M, Bion J. Virtual restorative environment therapy as an adjunct to pain control during burn dressing changes: study protocol for a randomised controlled trial. Trials 2015;16:329. ArticlePubMedPMC
- 12. Nilsson S, Finnström B, Kokinsky E, Enskär K. The use of virtual reality for needle-related procedural pain and distress in children and adolescents in a paediatric oncology unit. Eur J Oncol Nurs 2009;13:102-9.ArticlePubMed
- 13. Mosso-Vázquez JL, Gao K, Wiederhold BK, Wiederhold MD. Virtual reality for pain management in cardiac surgery. Cyberpsychol Behav Soc Netw 2014;17:371-8.ArticlePubMedPMC
- 14. Dascal J, Reid M, IsHak WW, Spiegel B, Recacho J, Rosen B, et al. Virtual reality and medical inpatients: a systematic review of randomized, controlled trials. Innov Clin Neurosci 2017;14:14-21.PubMedPMC
- 15. Turon M, Fernandez-Gonzalo S, Jodar M, Gomà G, Montanya J, Hernando D, et al. Feasibility and safety of virtual-reality-based early neurocognitive stimulation in critically ill patients. Ann Intensive Care 2017;7:81. ArticlePubMedPMC
- 16. Weech S, Kenny S, Barnett-Cowan M. Presence and cybersickness in virtual reality are negatively related: a review. Front Psychol 2019;10:158. ArticlePubMedPMC
- 17. Hoffmann MJ. FDA regulatory process. Silver Spring (MD), U.S. Food and Drug Administration. 2021;[cited 2021 July 7]. Available from: https://www.fda.gov/media/90419/download.
- 18. Rebenitsch L, Owen C. Review on cybersickness in applications and visual displays. Virtual Real 2016;20:101-25.Article
- 19. McMahan EA, Estes D. The effect of contact with natural environments on positive and negative affect: a meta-analysis. J Posit Psychol 2015;10:507-19.Article
- 20. Bruck S, Watters PA. Accessible virtual reality therapy using portable media devices. Stud Health Technol Inform 2010;154:87-91.PubMed
- 21. Sricharoenchai T, Parker AM, Zanni JM, Nelliot A, Dinglas VD, Needham DM. Safety of physical therapy interventions in critically ill patients: a single-center prospective evaluation of 1110 intensive care unit admissions. J Crit Care 2014;29:395-400.ArticlePubMed
- 22. Chlan LL. Relationship between two anxiety instruments in patients receiving mechanical ventilatory support. J Adv Nurs 2004;48:493-9.ArticlePubMed
- 23. Gerber SM, Jeitziner MM, Wyss P, Chesham A, Urwyler P, Müri RM, et al. Visuo-acoustic stimulation that helps you to relax: a virtual reality setup for patients in the intensive care unit. Sci Rep 2017;7:13228. ArticlePubMedPMC
- 24. Gerber SM, Jeitziner MM, Sänger SD, Knobel SEJ, Marchal-Crespo L, Müri RM, et al. Comparing the relaxing effects of different virtual reality environments in the intensive care unit: observational study. JMIR Perioper Med 2019;2:e15579.ArticlePubMedPMC
- 25. Ong TL, Ruppert MM, Akbar M, Rashidi P, Ozrazgat-Baslanti T, Bihorac A, et al. Improving the intensive care patient experience with virtual reality-a feasibility study. Crit Care Explor 2020;2:e0122.ArticlePubMedPMC
- 26. Lee JH, Lee HC, Jeon YT, Hwang JW, Lee H, Oh HW, et al. Clinical outcomes after unplanned extubation in a surgical intensive care population. World J Surg 2014;38:203-10.ArticlePubMed
- 27. Kiekkas P, Aretha D, Panteli E, Baltopoulos GI, Filos KS. Unplanned extubation in critically ill adults: clinical review. Nurs Crit Care 2013;18:123-34.ArticlePubMed
References
Figure & Data
References
Citations

- Opportunities for Managing Pain and Anxiety in the Intensive Care Unit Using Virtual Reality: Perspectives from Bedside Care Providers
Isabella P. Garito, Samantha Lewis-Fung, Brenna Lawson, Hannah Gabrielle Gray, Christopher Smith, Lora Appel
Journal of Medical Extended Reality.2024; 1(1): 53. CrossRef - Virtual Reality Therapy for People With Epilepsy and Related Anxiety: Protocol for a 3-Phase Pilot Clinical Trial
Hannah Gabrielle Gray, Danielle Tchao, Samantha Lewis-Fung, Susanna Pardini, Laurence R Harris, Lora Appel
JMIR Research Protocols.2023; 12: e41523. CrossRef - Virtual and augmented reality in intensive care medicine: a systematic review
Dominika Kanschik, Raphael Romano Bruno, Georg Wolff, Malte Kelm, Christian Jung
Annals of Intensive Care.2023;[Epub] CrossRef - A study to assess the communication and level of anxiety among mechanically ventilated conscious patients in intensive care units of sree mookambika medical college hospital at kanyakumari district
Derlin A. Ahisha, Retnam C. Ajitha
i-manager's Journal on Nursing.2023; 13(2): 20. CrossRef

 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite