Articles
- Page Path
- HOME > Acute Crit Care > Volume 34(3); 2019 > Article
- Case Report Acute aortic dissection developed after cardiopulmonary resuscitation: transesophageal echocardiographic observations and proposed mechanism of injury
-
Dong Keon Lee1
 , Kyung Sik Kang2, Yong Sung Cha2
, Kyung Sik Kang2, Yong Sung Cha2 , Kyoung-Chul Cha2
, Kyoung-Chul Cha2 , Hyun Kim2
, Hyun Kim2 , Kang Hyun Lee2
, Kang Hyun Lee2 , Sung Oh Hwang2
, Sung Oh Hwang2
-
Acute and Critical Care 2018;34(3):228-231.
DOI: https://doi.org/10.4266/acc.2015.00633
Published online: April 26, 2018
1Department of Emergency Medicine, Hallym University Sacred Heart Hospital, Hallym University College of Medicine, Anyang, Korea
2Department of Emergency Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea
- Correspondence to: Sung Oh Hwang, Department of Emergency Medicine, Yonsei University Wonju College of Medicine, 20 Ilsan-ro, Wonju 26426, Korea Tel: +82-33-741-1611, Fax: +82-33-742-3030, E-mail: shwang@yonsei.ac.kr
• Received: November 20, 2015 • Revised: May 17, 2016 • Accepted: October 13, 2016
Copyright © 2019 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- There has been no report about aortic dissection due to cardiopulmonary resuscitation (CPR). We present here a case of acute aortic dissection as a rare complication of CPR and propose the potential mechanism of injury on the basis of transesophageal echocardiographic observations. A 54-year-old man presented with cardiac arrest after choking and received 19 minutes of CPR in the emergency department. Transesophageal echocardiography (TEE) during CPR revealed a focal separation of the intimal layer at the descending thoracic aorta without evidence of aortic dissection. After restoration of spontaneous circulation, hemorrhagic cardiac tamponade developed. Follow-up TEE to investigate the cause of cardiac tamponade revealed aortic dissection of the descending thoracic aorta. Hemorrhagic cardiac tamponade was thought to be caused by myocardial hemorrhage from CPR.
Case Report
Discussion
Supplementary Materials
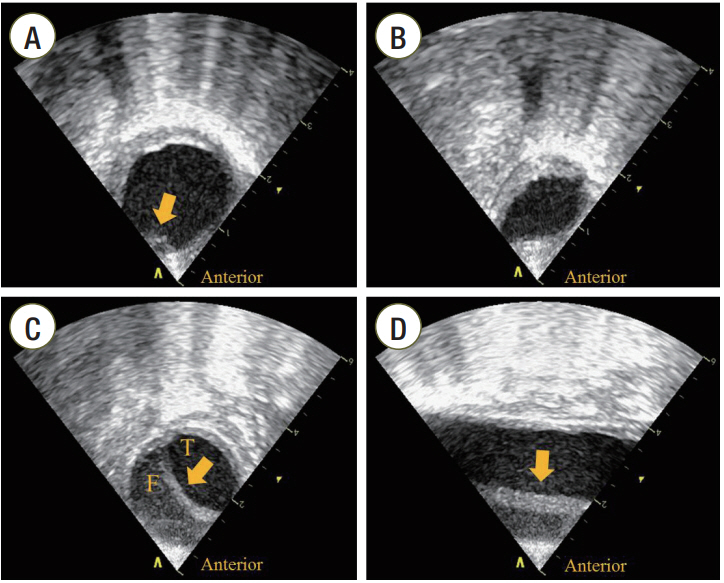
Figure 1.Transesophageal echocardiographic findings of the aorta. (A) Transesophageal echocardiography (TEE) findings in the aorta during cardiopulmonary resuscitation. A focal separation of the intimal layer at the anterior wall of descending thoracic aorta (DTA) with atherosclerotic changes (arrow) was seen on the short axis view of the aorta. (B) Compression of the DTA during compression systole. The DTA was compressed and deformed on the short axis view of the aorta when external chest compression was performed. (C) Short axis view of the aorta after restoration of spontaneous circulation (ROSC). Follow-up TEE revealed the intimal flap of the DTA (arrow) on the short axis view of the aorta. (D) Long axis view of the aorta after ROSC. The intimal flap (arrow) was observed between the distal portion of the aortic arch and 11 cm down from the aortic isthmus. T: true lumen; F: false lumen.


Figure 2.Thoraco-abdominal computed tomography (CT) angiographic findings. (A) Intimal tear in the proximal descending aorta (arrow). (B) Intimal flap in the dissected aorta (arrow). (C) Coronal view of chest CT angiography shows aortic dissection of the descending thoracic aorta (DTA) with no involvement of the ascending and abdominal aorta (arrow). (D) Three-dimensional CT angiography shows dissection of the DTA (arrow).


- 1. Hashimoto Y, Moriya F, Furumiya J. Forensic aspects of complications resulting from cardiopulmonary resuscitation. Leg Med (Tokyo) 2007;9:94-9.ArticlePubMed
- 2. Krischer JP, Fine EG, Davis JH, Nagel EL. Complications of cardiac resuscitation. Chest 1987;92:287-91.ArticlePubMed
- 3. Hoke RS, Chamberlain D. Skeletal chest injuries secondary to cardiopulmonary resuscitation. Resuscitation 2004;63:327-38.ArticlePubMed
- 4. Higano ST, Oh JK, Ewy GA, Seward JB. The mechanism of blood flow during closed chest cardiac massage in humans: transesophageal echocardiographic observations. Mayo Clin Proc 1990;65:1432-40.ArticlePubMedPMC
- 5. Ma MH, Hwang JJ, Lai LP, Wang SM, Huang GT, Shyu KG, et al. Transesophageal echocardiographic assessment of mitral valve position and pulmonary venous flow during cardiopulmonary resuscitation in humans. Circulation 1995;92:854-61.ArticlePubMed
- 6. Dyer DS, Moore EE, Ilke DN, McIntyre RC, Bernstein SM, Durham JD, et al. Thoracic aortic injury: how predictive is mechanism and is chest computed tomography a reliable screening tool? A prospective study of 1,561 patients. J Trauma 2000;48:673-82.ArticlePubMed
- 7. Mirvis SE, Shanmuganathan K, Buell J, Rodriguez A. Use of spiral computed tomography for the assessment of blunt trauma patients with potential aortic injury. J Trauma 1998;45:922-30.ArticlePubMed
- 8. Steenburg SD, Ravenel JG. Acute traumatic thoracic aortic injuries: experience with 64-MDCT. AJR Am J Roentgenol 2008;191:1564-9.ArticlePubMed
- 9. Ungar TC, Wolf SJ, Haukoos JS, Dyer DS, Moore EE. Derivation of a clinical decision rule to exclude thoracic aortic imaging in patients with blunt chest trauma after motor vehicle collisions. J Trauma 2006;61:1150-5.ArticlePubMed
- 10. Juan YH, Saboo SS, Desai NS, Khandelwal K, Khandelwal A. Aortic intramural hematoma and hepatic artery pseudoaneurysm: unusual complication following resuscitation. Am J Emerg Med 2014;32:107. e1-4.Article
- 11. Hwang SO, Lee KH, Cho JH, Yoon J, Choe KH. Changes of aortic dimensions as evidence of cardiac pump mechanism during cardiopulmonary resuscitation in humans. Resuscitation 2001;50:87-93.ArticlePubMed
References
Figure & Data
References
Citations
Citations to this article as recorded by 

- Thoracic Aortic Rupture Post Cardiopulmonary Resuscitation in a Patient With Previous Thoracic Aneurysm Repair
Aniekeme S Etuk, Olanrewaju F Adeniran , Bernard I Nkwocha, Nformbuh Asangmbeng, Mina Jacob
Cureus.2023;[Epub] CrossRef - Cardiac Arrest as an Uncommon Manifestation of Late Type A Aortic Dissection Associated with Transcatheter Aortic Valve Replacement
Jan Naar, Dagmar Vondrakova, Andreas Kruger, Marek Janotka, Iva Zemanova, Martin Syrucek, Petr Neuzil, Petr Ostadal
Journal of Clinical Medicine.2023; 12(16): 5318. CrossRef - Blunt Thoracic Aortic Injury and Contemporary Management Strategy
Ranjan Dahal, Yogesh Acharya, Alan H. Tyroch, Debabrata Mukherjee
Angiology.2022; 73(6): 497. CrossRef - Resuscitative endovascular occlusion of the aorta (REBOA) as a mechanical method for increasing the coronary perfusion pressure in non-traumatic out-of-hospital cardiac arrest patients
Dong-Hyun Jang, Dong Keon Lee, You Hwan Jo, Seung Min Park, Young Taeck Oh, Chang Woo Im
Resuscitation.2022; 179: 277. CrossRef - Blunt traumatic aortic dissection death by falling: an autopsy case report
Gentaro Yamasaki, Marie Sugimoto, Takeshi Kondo, Motonori Takahashi, Mai Morichika, Azumi Kuse, Kanako Nakagawa, Yasuhiro Ueno, Migiwa Asano
Forensic Science, Medicine and Pathology.2022; 19(3): 388. CrossRef - Intra-arrest transesophageal echocardiography during cardiopulmonary resuscitation
Sung Oh Hwang, Woo Jin Jung, Young-Il Roh, Kyoung-Chul Cha
Clinical and Experimental Emergency Medicine.2022; 9(4): 271. CrossRef - Intra-arrest transoesophageal echocardiographic findings and resuscitation outcomes
Woo Jin Jung, Kyoung-Chul Cha, Yong Won Kim, Yoon Seop Kim, Young-Il Roh, Sun Ju Kim, Hye Sim Kim, Sung Oh Hwang
Resuscitation.2020; 154: 31. CrossRef - Aortic Rupture as a Complication of Cardiopulmonary Resuscitation
Prashanth Venkatesh, Edward J. Schenck
JACC: Case Reports.2020; 2(8): 1150. CrossRef

 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite