Articles
- Page Path
- HOME > Acute Crit Care > Volume 32(3); 2017 > Article
- Letter to the Editor An Ofloxacin-Induced Anaphylaxis through an IgG4-Mediated but Not IgE-Mediated Basophil Activation Mechanism
-
Ji Hye Kim1
 , Dae-Hong Seo2, Ga-Young Ban3
, Dae-Hong Seo2, Ga-Young Ban3 , Eun-Mi Yang3, Yoo Seob Shin3
, Eun-Mi Yang3, Yoo Seob Shin3 , Young-Min Ye3
, Young-Min Ye3 , Hae-Sim Park3
, Hae-Sim Park3
-
Korean Journal of Critical Care Medicine 2017;32(3):302-305.
DOI: https://doi.org/10.4266/kjccm.2017.00108
Published online: August 31, 2017
1Division of Pulmonology and Allergy, Konyang University Hospital, Konyang University College of Medicine, Daejeon, Korea
2Division of Allergy and Pulmonology, Chungmu Hospital, Cheonan, Korea
3Department of Allergy and Clinical Immunology, Ajou University School of Medicine, Suwon, Korea
- Hae-Sim Park Department of Allergy and Clinical Immunology, Ajou University School of Medicine, 206 World cup-ro, Yeongtonggu, Suwon 16499, Korea Tel: +82-31-219-5196 Fax: +82-31-219-5154 E-mail: hspark@ajou.ac.kr
• Received: February 14, 2017 • Revised: April 20, 2017 • Accepted: April 21, 2017
Copyright © 2017 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 6,158 Views
- 81 Download
- 1 Crossref
Dear Editor:
Figure 1.Detection of serum specific IgG4 antibodies to ofloxacin-human serum albumin (HSA) conjugate by ELISA from the patient (●) and 20 non-atopic normal controls (○). OD: optical density; ELISA: enzyme-linked immunosorbent assay.


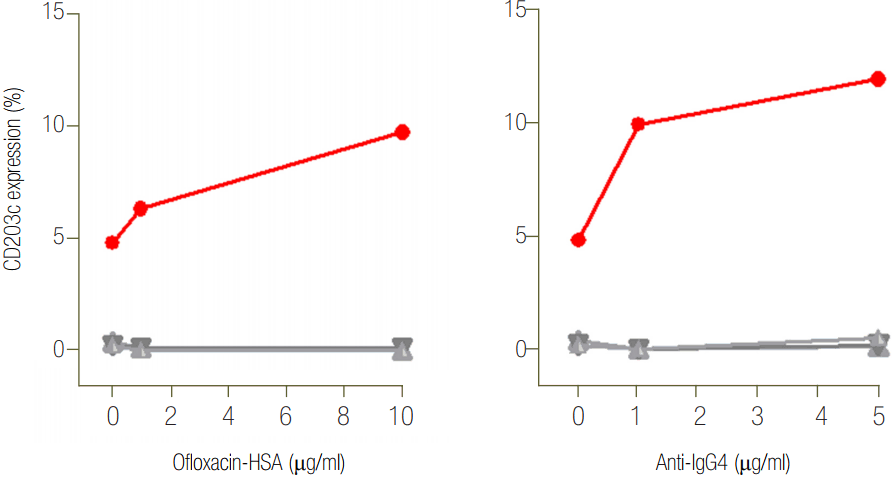
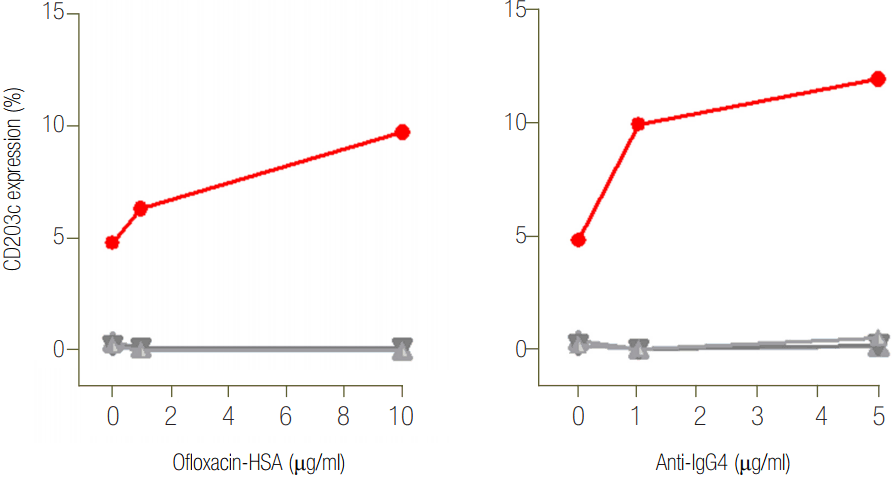
Figure 2.Results of basophil activation test with addition of ofloxacin-human serum albumin (HSA) conjugate (A) and anti-IgG4 antibodies (B) in the patient (●) and three healthy controls (◇, △, ∇).

- 1. Blanca-López N, Andreu I, Torres Jaén MJ. Hypersensitivity reactions to quinolones. Curr Opin Allergy Clin Immunol 2011;11:285-91.ArticlePubMed
- 2. Nam YH, Kim JE, Kim SH, Jin HJ, Hwang EK, Shin YS, et al. Immunologic evaluation of ofloxacin hypersensitivity. Allergy Asthma Immunol Res 2012;4:367-9.ArticlePubMedPMC
- 3. Hicks LA, Bartoces MG, Roberts RM, Suda KJ, Hunkler RJ, Taylor TH Jr, et al. US outpatient antibiotic prescribing variation according to geography, patient population, and provider specialty in 2011. Clin Infect Dis 2015;60:1308-16.ArticlePubMedPDF
- 4. Jones SC, Budnitz DS, Sorbello A, Mehta H. USbased emergency department visits for fluoroquinolone-associated hypersensitivity reactions. Pharmacoepidemiol Drug Saf 2013;22:1099-106.ArticlePubMedPMC
- 5. Aranda A, Mayorga C, Ariza A, Doña I, Rosado A, Blanca-Lopez N, et al. In vitro evaluation of IgE-mediated hypersensitivity reactions to quinolones. Allergy 2011;66:247-54.ArticlePubMed
- 6. Manfredi M, Severino M, Testi S, Macchia D, Ermini G, Pichler WJ, et al. Detection of specific IgE to quinolones. J Allergy Clin Immunol 2004;113:155-60.ArticlePubMed
- 7. Lobera T, Audícana MT, Alarcón E, Longo N, Navarro B, Muñoz D. Allergy to quinolones: low crossreactivity to levofloxacin. J Investig Allergol Clin Immunol 2010;20:607-11.PubMed
- 8. Nam YH, Hwang EK, Ban GY, Jin HJ, Yoo HS, Shin YS, et al. Immunologic evaluation of patients with cefotetan-induced anaphylaxis. Allergy Asthma Immunol Res 2015;7:301-3.ArticlePubMedPMC
- 9. Finkelman FD. Anaphylaxis: lessons from mouse models. J Allergy Clin Immunol 2007;120:506-15.ArticlePubMed
- 10. Tsujimura Y, Obata K, Mukai K, Shindou H, Yoshida M, Nishikado H, et al. Basophils play a pivotal role in immunoglobulin-G-mediated but not immunoglobulin-E-mediated systemic anaphylaxis. Immunity 2008;28:581-9.ArticlePubMed
- 11. Gómez E, Torres MJ, Mayorga C, Blanca M. Immunologic evaluation of drug allergy. Allergy Asthma Immunol Res 2012;4:251-63.ArticlePubMedPMC
- 12. Kim SY, Kim JH, Jang YS, Choi JH, Park S, Hwang YI, et al. The basophil activation test is safe and useful for confirming drug-induced anaphylaxis. Allergy Asthma Immunol Res 2016;8:541-4.ArticlePubMedPMC
- 13. Song JW, Jang YS, Jung MC, Kim JH, Choi JH, Park S, et al. Levodropropizine-induced anaphylaxis: case series and literature review. Allergy Asthma Immunol Res 2017;9:278-80.ArticlePubMedPMC
- 14. Simons FE, Ebisawa M, Sanchez-Borges M, Thong BY, Worm M, Tanno LK, et al. 2015 Update of the evidence base: World Allergy Organization anaphylaxis guidelines. World Allergy Organ J 2015;8:32. ArticlePubMedPMCPDF
- 15. Simons FE. Anaphylaxis pathogenesis and treatment. Allergy 2011;66 Suppl 95:31-4.ArticlePubMed
References
Figure & Data
References
Citations
Citations to this article as recorded by 

- A CASE PRESENTATION ON OFLOXACIN INDUCED DERMAL HYPERSENSITIVITY REACTION
BHAVANAM DIVYA, LEDO THANKACHAN
International Journal of Pharmacy and Pharmaceutical Sciences.2022; : 53. CrossRef

 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite