Successful extracorporeal membrane oxygenation treatment of catecholamine-induced cardiomyopathy-associated pheochromocytoma: a case report
Article information
Abstract
The main mechanism of Takotsubo cardiomyopathy (TCM) is catecholamine-induced acute myocardial stunning. Pheochromocytoma, a catecholamine-secreting tumor, can cause several cardiovascular complications, including hypertensive crisis, myocardial infarction, toxic myocarditis, and TCM. A 29-year-old woman presented to our hospital with general weakness, vomiting, dyspnea, and chest pain. The patient was nullipara, 28 weeks’ gestation, and had a cachexic morphology. Her cardiac enzyme levels were elevated and bedside echocardiography showed apical akinesia, suggesting TCM. The next day, she could not feel the fetal movement, and an emergency cesarean section was performed. After delivery, the patient experienced cardiac arrest and was transferred to the intensive care unit for cardiopulmonary resuscitation (CPR). Spontaneous circulation returned after 28 minutes of CPR, but cardiogenic shock continued, and extracorporeal membrane oxygenation (ECMO) was initiated. On the third day of ECMO maintenance, left ventricular ejection fraction improved and blood pressure stabilized. On the eighth day after ECMO insertion, it was removed. However, complications of the left leg vessels occurred, and several surgeries and interventions were performed. A left adrenal gland mass was found on computed tomography and was removed while repairing the leg vessels. Pheochromocytoma was diagnosed and left adrenalectomy was performed.
Takotsubo cardiomyopathy (TCM), also known as stress cardiomyopathy, is believed to be caused by enhanced sympathetic tone [1]. Catecholamines and their oxidation have a direct toxic effect on the myocardium, causing intracellular calcium overload and myocardial cell damage [2]. Contraction band necrosis, increased fibrosis, and mononuclear inflammatory infiltrates have been found on endomyocardial biopsy in some patients with TCM. These findings are consistent with catecholamine-mediated cardiotoxicity [3]. Common causes of TCM include physical and emotional stress and, rarely, the overproduction of endogenous catecholamines (pheochromocytoma, neuroblastoma) or the administration of exogenous catecholamines [1].
Among these etiologies, pheochromocytoma can lead to several complications. Cardiovascular complications include hypertensive crisis, myocardial ischemia/infarction, toxic myocarditis, dilated cardiomyopathy, TCM, and arrhythmia. Nervous system complications include transient ischemic attack, stroke, and headache [4]. In particular, pheochromocytomas are accompanied by cardiomyopathy; cardiac function is restored in most patients once the pheochromocytoma is surgically removed [5]. However, abrupt changes in abdominal pressure in patients with a pheochromocytoma can cause a catecholamine surge, which may lead to cardiac arrest; therefore, it is important to minimize changes in abdominal pressure during pheochromocytoma surgery [6].
Here, we present the case of a pregnant woman with TCM caused by multiple underlying etiologies and her recovery through extracorporeal membrane oxygenation (ECMO) after she experienced cardiac arrest after cesarean section. This study was approved by the Institutional Review Board (No. 2021-06-027-001). The Board waived the need for informed consent for the publication of this report.
CASE REPORT
A 29-year-old woman had intermittent headache for several years, and general weakness for about 6 months that worsened 3 days prior to admission. Moreover, she presented with an 8-hour history of vomiting, dyspnea, and chest pain before admission to the emergency room in December 2020. The patient was also admitted to the Department of Psychiatry in 2006 for anorexia nervosa. The patient was a nullipara at 28 weeks’ gestation and had a cachexic morphology, with a height of 163 cm and weight of 38 kg (body mass index, 14.3 kg/m2). The patient was admitted to another hospital before visiting the emergency room and an increase in myocardial enzymes was detected at that time. However, considering her age and symptoms, myocarditis or pericarditis was suspected and she was transferred to our hospital. Upon admission to the emergency room, her vital signs were stable and no pulmonary or peripheral edema was present, but she had a marked increase in lactic acid levels (12.0 mmol/L; normal range, 0.5–1.6 mmol/L), urine output was reduced to about 0.5 ml/kg/hr, sinus tachycardia and poor R-wave progression were observed on electrocardiogram, and cardiac enzyme were as follows: creatinine kinase-MB, 50.31 ng/ml (normal range, 0–4.94 ng/ml); troponin T, 4.46 ng/ml (normal range, <0.014 ng/ml); N-terminal pro-B-type natriuretic peptide, 1,980 pg/ml (normal range, 0–263 pg/ml); and creatinine, 1.74 mg/dl. Her creatinine level was elevated compared to 1 month earlier (0.72 mg/dl); therefore, she was considered to have stage 2 acute kidney injury. There were no symptoms suggestive of infection, high-sensitivity C-reactive protein and white blood count levels were normal, and thyroid function tests were also normal. In addition, bedside echocardiography revealed apical akinesia, suggesting TCM. As the possibility of coronary artery disease was not high and the patient was pregnant, coronary angiography (CAG) was not performed.
On the morning of the second day of hospitalization, the patient complained to the obstetrician that she could no longer feel fetal movement. Fetal condition was not reassuring on prenatal ultrasound; therefore, an emergency cesarean section was performed. In the operating room, the patient’s blood pressure was 79/52 mm Hg, and her heart rate was 143 BPM. The patient was diagnosed with cardiogenic shock, and dopamine was started. During spinal anesthesia, 9 mg of bupivacaine was injected intrathecally. Since blood pressure was not maintained, dobutamine was administered. After delivery, the patient developed cardiac arrest and was transferred to the intensive care unit for cardiopulmonary resuscitation (CPR). Return of spontaneous circulation occurred after 28 minutes of CPR, but cardiogenic shock continued, and ECMO was inserted at bedside (drain catheter site, right femoral vein; perfusion catheter, left femoral artery). Echocardiography showed severe left ventricular systolic dysfunction and mid to apical akinesia, but left ventricular size was normal, and valvular dysfunction and pericardial effusion were not observed (Figure 1, Supplementary Material 1). No abnormal findings were observed on the CAG (Figure 2).

(A) Left ventricular end diastolic volume (75.01 ml [1]) on apical four-chamber view. (B) Left ventricular end systolic volume (59.25 ml [2]) on apical four-chamber view. (C) Left ventricular end diastolic volume (61.66 ml [1]) on apical two-chamber view. (D) Left ventricular end systolic volume (55.06 ml & ejection fraction 16% [2]) on apical two-chamber view.

(A) Left atrial venting via Brockenbrough needle (HeartSpan; Merit Medical). (B) Right coronary artery and (C) left coronary artery are normal.
On the third day of ECMO maintenance, left ventricular ejection fraction improved and blood pressure stabilized. On the eighth day after insertion, ECMO was removed. The patient stabilized and was moved to the general ward. Lower extremity computed tomography (CT) angiography was performed due to an ulcer-like color change on the patient’s right foot. The results showed no major abnormalities in the vessels of the right lower limb, but revealed complete obstruction from the left common iliac artery to the distal superficial femoral artery; thrombectomy was performed. The patient was discharged with no symptoms of claudication, but 3 months later, she complained of claudication in her left leg. Low-extremity CT angiography was repeated. A pseudoaneurysm and decreased size of the lumen of the arteries in the left lower extremity were observed. Plans were made to transfer the patient to another hospital for vascular surgery.
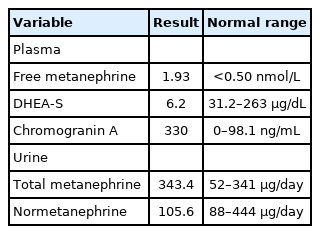
A left adrenal gland mass was found incidentally on CT while investigating a vascular abnormality in the leg. Plasma free metanephrine (1.93 nmol/L; normal range, <0.50 nmol/L), urine total metanephrine (343.4 µg/day; normal range, 52–341 µg/day), and chromogranin A (330 ng/ml; normal range, 0–98.1 ng/ml) were elevated, but dehydroepiandrosterone sulfate was not elevated. This suggested that it was not a malignant mass, but pheochromocytoma was suspected (Table 1). The diagnosis of pheochromocytoma was confirmed by positron emission tomography-CT showing high uptake in the left adrenal gland (Figure 3). A well-defined, encapsulated mass (3.4×2.5×2.1 cm) in the medulla of the adrenal gland was resected via left adrenalectomy. There was no vascular and lymphatic invasion.
DISCUSSION
A pheochromocytoma is a tumor derived from chromaffin cells in the adrenal medulla or extra-adrenal paraganglia [7]. Pheochromocytoma-induced cardiomyopathy has a variety of clinical manifestations, and the clinical manifestations usually worsen with catecholamine exposure [8]. Cardiac dysfunction of unknown cause in pregnant women include peripartum cardiomyopathy (PPCM) and TCM. Neither disease has clear diagnostic criteria; they can only be diagnosed after excluding other diseases. However, it is difficult to distinguish between the two diseases in the case of maternal cardiac dysfunction due to their similar clinical features [9].
This patient developed TCM due to catecholamine overdrive and developed cardiogenic shock. The patient underwent cesarean section and delivery. Excessive secretion of catecholamines in patients with pheochromocytoma often occurs during childbirth. This is because the process of childbirth causes a rapid change in intra-abdominal pressure. A tumor that was compressed during pregnancy becomes more active and secretes more catecholamines due to reduced pressure [10]. Cardiac dysfunction is uncommon in patients with pheochromocytoma in the absence of a catecholamine crisis. However, intraoperative release of catecholamines can occur during surgery. The increased risk of perioperative hemodynamic collapse is likely to be caused by acute stress and local release of catecholamines rather than chronic elevation of catecholamines [11].
The patient was in a cachexic state due to anorexia nervosa, which could have increased catecholamine secretion, and bupivacaine, which was used during spinal anesthesia, can also increase catecholamines [12,13]. This patient did not meet the criteria for diagnosis of PPCM in the last month of pregnancy or within 5 months of delivery. However, measurement of serum prolactin elevation, soluble fms-like tyrosine kinase-1 (sFlt-1), placenta growth factor (PIGF), and/or sFlt-1 to PIGF ratio, which can be used as indicators of PPCM, could have useful for diagnosis of PPCM and TCM [14,15]. This case shows that additional tests for catecholamine-producing tumors, such as pheochromocytomas, should be considered in cases of cardiomyopathy diagnosed during pregnancy.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS
Conceptualization: DHB, SYL. Data Curation: MK, DIL, JHL, JWB. Writing–original draft: SP. Writing–review & editing: DHB, SK, KKH, DWK, MCC.
FUNDING
None.
ACKNOWLEDGMENTS
None.
SUPPLEMENTARY MATERIALS
Supplementary materials can be found via https://doi.org/10.4266/acc.2021.01158.
In the apical four chamber view, severe left ventricular systolic dysfunction and mid to apex regional wall motion abnormality were observed.
acc-2021-01158-v1.mp4