The ABCDEF Implementation Bundle
Article information
Abstract
Long-term morbidity, long-term cognitive impairment and hospitalization-associated disability are common occurrence in the survivors of critical illness, with significant consequences for patients and for the caregivers. The ABCDEF bundle represents an evidence-based guide for clinicians to approach the organizational changes needed for optimizing ICU patient recovery and outcomes. The ABCDEF bundle includes: Assess, Prevent, and Manage Pain, Both Spontaneous Awakening Trials (SAT) and Spontaneous Breathing Trials (SBT), Choice of analgesia and sedation, Delirium: Assess, Prevent, and Manage, Early mobility and Exercise, and Family engagement. The purpose of this review is to describe the core features of the ABCDEF bundle.
Introduction
The Society of Critical Care Medicine (SCCM) has endorsed and published the 2013Clinical Practice Guidelines for the Management of Pain, Agitation, and Delirium (PAD) in Adult Patients in the Intensive Care Unit (ICU), which outline the best evidence available for addressing the inextricably linked elements of patient comfort and safety—pain, agitation, and delirium.[1] There is more than one way of changing practice to implement the PAD guidelines, and perhaps the most important thing for a team or individual clinician to acknowledge is that change is indeed needed.
The SCCM is embarking on a PAD implementation program called the ICU Liberation Collaborative, which is framed around a 6-step approach called the ABCDEF bundle (Assess, prevent, and manage pain; Both spontaneous awakening trials and spontaneous breathing trials; Choice of sedation and analgesia; Delirium assessment, prevention, and management; Early mobility and exercise; and Family communication and involvement).[2,3]
Balas et al.[4] evaluated, in a prospective, before-after study, the effectiveness and safety of implementing the ABCDE bundle into everyday practice. The 150 patients in the post-implementation period spent three more days breathing without mechanical assistance (median [interquartile range, IQR], 24 [7 to 26] vs. 21 [0 to 25]; p = 0.04), experienced a near halving of the odds of delirium (odds ratio [OR], 0.55; 95% confidence interval [CI], 0.33-0.93; p = 0.03) and increased odds of mobilizing out of bed at least once during an ICU stay (OR, 2.11; 95% CI, 1.29-3.45; p = 0.003) than did those in the pre-implementation period (146 pts).[4]
The purpose of this review is to describe the core evidence and features behind the ABCDEF bundle approach to optimize our patient recovery and outcomes.
A: Assessment, Prevention, and Management of Pain
ICU patients commonly experience pain, with an incidence of up to 50% in surgical and medical patients. It is a major clinical symptom that requires systematic diagnosis and treatment.[5,6] In the Europain study, a prospective, cross-sectional, multicenter, multinational study of pain intensity, Puntillo et al. showed that common ICU procedures induced a significant increase in pain, although no procedure caused severe pain. For the three most painful procedures (i.e., chest tube removal, wound drain removal, and arterial line insertion) pain intensity more than doubled during the procedure compared with the pre-procedural levels.[7]
Inadequately controlled pain is a risk factor for nosocomial infections and longer durations of mechanical ventilation and may precipitate delirium.[8,9]
The PAD guidelines recommend that all adult ICU patients be routinely assessed for pain with an objective, valid, and reliable instrument.[1]
Patient’s self-reporting of pain using a 1-10 numerical rating scale (NRS) is considered the gold standard and is highly recommended by many critical care societies. [1,5,6] In the absence of a patient’s self-report, observable behavioral and physiological indicators become important indices for the assessment of pain.[10] The Behavioral Pain Scale (BPS) and the Critical‐Care Pain Observation
Tool (CPOT) are the most valid and reliable behavioral pain scales for ICU patients unable to communicate.[8,11] The BPS is used to monitor 3 behavioral domains (facial expression, upper limbs, and compliance with mechanical ventilation).[12] The CPOT, the most commonly used of the two instruments, is feasible, easy to complete, and simple to understand and includes evaluation of 4 behaviors (facial expressions, body movements, muscle tension, and compliance with the ventilator for mechanically ventilated patients or vocalization for nonintubated patients).[10]
According to ICU PAD Guidelines, pain medications should be routinely administered in the presence of significant pain (i.e., NRS > 4, BPS > 5, or CPOT > 3) and prior to performing painful invasive procedures.[1]
B: Both Spontaneous Awakening Trials and Spontaneous Breathing Trials
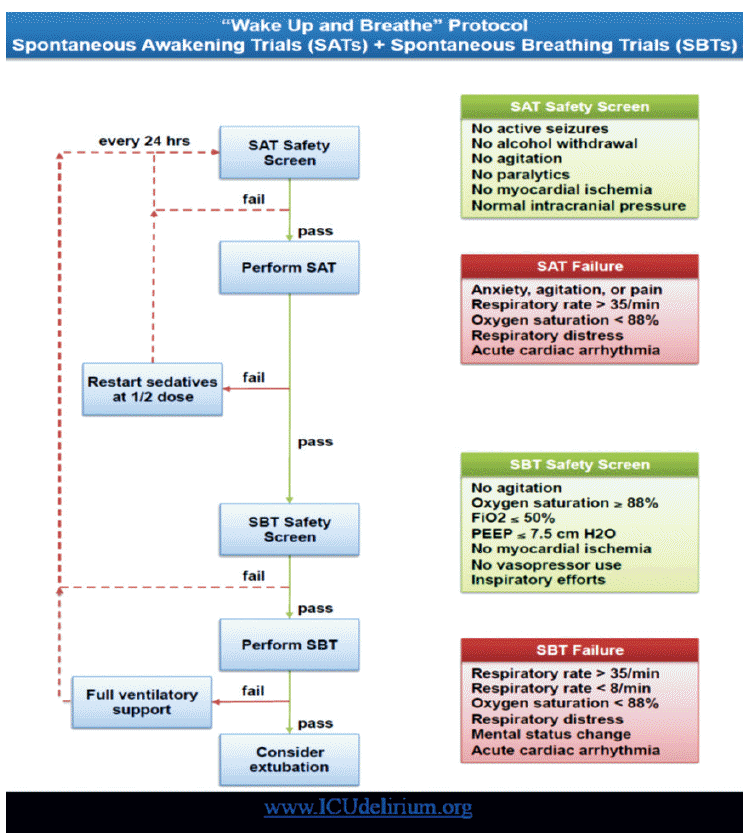
The next step in ICU liberation is to conduct both a spontaneous awakening trial (SAT) and a spontaneous breathing trial (SBT). This component of the modern-day processes of care, for all patients receiving mechanical ventilation, involves testing each patient who passes the safety screens for his or her ability to tolerate removal of sedatives and narcotics (as long as pain control is achieved) and removal of mechanical ventilation (ie, allowing the patient to experience spontaneous awakening and breathing) (Fig. 1).[13]
The 2013 ICU PAD Guidelines stressed the importance of minimizing sedative use and maintaining a light level of sedation in patients, using either a daily sedative interruption strategy (i.e., SAT), or by continuously titrating sedatives to maintain a light level of sedation (i.e., targeted sedation strategy).
Kress et al.[14] conducted a randomized, controlled trial involving 128 adult patients who were receiving mechanical ventilation and continuous infusions of sedative drugs in a medical ICU. In the intervention group, the sedative infusions were interrupted daily until the patients were awake; in the control group, the infusions were interrupted only at the discretion of the clinicians. The daily interruption of the infusion of sedative drugs shortened the duration of mechanical ventilation by more than 2 days and the length of stay in the ICU by 3.5 days.[14]
There is a consistent relationship between deeper sedation and worse ICU outcomes. Deep sedation in the first 48 hours of an ICU stay has been associated with delayed time to extubation, higher need for tracheostomy, increased risk of hospital and long term death.[15-17] Shehabi et al.[15] examined the relationships between early sedation and outcomes. Every additional Richmond Agitation-Sedation Score (RASS) assessment in the deep sedation range in the first 48 hours was associated with delayed time to extubation of 12.3 hours, a 10% increased risk of hospital death, and an 8% increased risk of death at 6 months.[15] In the study of Balzer et al.[17] 1,884 patients receiving mechanical ventilation were grouped as either lightly or deeply sedated (light sedation: RASS −2 to 0; deep: RASS −3 or below). Deep sedation (27.2%, n = 513) was associated with an in-hospital mortality hazard ratio of 1.661 (95% CI: 1.074 to 2.567; p = 0.022) and a two-year hazard ratio of 1.866 (95% CI: 1.351 to 2.576; p < 0.001). In summary, deeply sedated patients had longer ventilation times, increased length of stay and higher rates of mortality.[17] According to the results of these studies, early deep sedation is a modifiable risk factor and that the implementation of sedation protocols to achieve light sedation is feasible and reproducible in the early phase of ICU treatment.
Numerous randomized trials support the use of ventilator weaning protocols that include daily SBTs as their centerpiece.[18,19] Girard et al.[13] undertook the Awakening and Breathing Controlled (ABC) trial, a multicenter, randomized controlled trial to assess the efficacy and safety of a protocol of daily SATs paired with SBTs (intervention group, n = 168) versus a standard SBT protocol in patients receiving patient-targeted sedation as part of usual care (control group, n = 168). Patients in the intervention group (both SAT and SBT) spent more days breathing without assistance during the 28-day study period (14.7 days versus 11.6 days; mean difference 3.1 days, 95% CI: 0.7-5.6, p = 0.02) and were discharged earlier from the ICU (median time in ICU of 9.1 days versus 12.9 days, p = 0.01) and earlier from the hospital (median hospital time 14.9 days versus 19.2 days, p = 0.04).[13] During the year after enrollment, patients receiving SATs with SBTs (intervention) were less likely to die than were patients receiving only SBTs (control) (hazard ratio = 0.68, 95% CI: 0.50-0.92, p = 0.01).[13] Conversely in the Daily Sedative Interruption in Critically Ill Patients Being Managed With a Sedation Protocol (SLEAP) trial (protocolized light sedation in combination with daily SAT versus protocolized light sedation alone), found no difference between the groups with regard to time to extubation, duration of ICU and hospital stays.[20] One possible reason the SLEAP study might not have showed an effect is because both the treatment and control groups received higher average sedative doses that would result in moderate to deep levels rather than light levels of sedation.[21] Ultimately, the core features of the ABCDEF bundle involve coordination of SATs and SBTs emphasizing narcotic and sedation titration resulting in earlier liberation from mechanical ventilation, ICU, and hospitalization
C: Choice of Sedation and Analgesia
Choice of sedation is crucial to patients’ clinical outcomes. Psychoactive medication administration should be goal-directed to ensure adequate pain control, anxiolysis, and prevention and treatment of delirium. In addition to selection of drug, the dose, titration, and prompt discontinuation of these medications are of paramount importance.
The 2013 PAD guidelines emphasize the need for goal-directed delivery of psychoactive medications to avoid over-sedation, to promote earlier extubation, and to help the medical team agree on a target sedation level by using sedation scales. Of the available reliable and valid sedation scales, the PAD guidelines recommend the use of the RASS and the Riker Sedation-Agitation Scale (SAS) based on published literature and the psychometric properties of the scale.[1]
To maximize patient outcomes, it is essential to carefully choose sedatives and analgesic medications, as well as consider medication doses, titration, and discontinuation.[22] There is a clear association between decreased exposure to sedative medications, particularly benzodiazepines, and improved patient outcome.[15,17,23] Pandharipande et al.[24] evaluated 198 mechanically ventilated patients to determine the probability of daily transition to delirium, as a function of sedative and analgesic dose administration during the previous 24 h. They found that every unit dose of lorazepam was associated with a higher risk for daily transition to delirium (OR, 1.2 [95% CI, 1.1–1.4]; p = 0.003).[24] Similarly Seymour et al.[25] confirmed that benzodiazepines are an independent risk factor for development of delirium during critical illness even when given more than 8 hours before a delirium assessment. The SEDCOM trial (Safety and Efficacy of Dexmedetomidine Compared with Midazolam) showed a reduction in the prevalence of delirium and in the duration of mechanical ventilation in patients sedated with dexmedetomidine compared with midazolam.[26] The MENDS study (Maximizing Efficacy of Targeted Sedation and Reducing Neurological Dysfunction) evaluated the role of changing sedation paradigms on acute brain dysfunction, comparing dexmedetomidine with lorazepam.[27] The dexmedetomidine sedative strategy resulted in more days alive without delirium or coma, but without differences in mortality or ventilator-free days. Notably, the subgroup of septic patients sedated with dexmedetomidine in the MENDS study had shorter durations of delirium and coma, lower daily probability of delirium, shorter time on the ventilator, and improved 28-day survival.[28] There is an ongoing trial (MENDS II study) to determine the best sedative medication to reduce delirium and improve survival and long-term brain function in the ventilated septic patient (ClinicalTrials.gov Identifier: NCT01739933).
D: Delirium Assessment, Prevention, and Management
Delirium is defined in the Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition) as an acute disturbance of consciousness with inattention accompanied by a change in cognition or perceptual disturbance that fluctuates over time.[29] Three key points to clarifying the syntax surrounding delirium and coma are as follows: First, coma represents the clinical state of a patient who is unarousable to voice (whether this is due to disease or to iatrogenic causes such as deliberate or unintentional overuse of sedation). Second, hallucinations and delusions are not key major components of the diagnosis of delirium. That is, many patients are delirious who do not have either hallucinations or delusions. Third, clinicians need to realize that inattention is the most important diagnostic criterion for delirium.[30]
Delirium is prevalent, under-recognized, and an independent predictor of poor outcomes such as mortality, prolonged mechanical ventilation, increased length of hospital stay, and long-term cognitive impairment.[31,32]
Between 50% and 80% of mechanically ventilated ICU patients develop delirium, and this organ dysfunction is missed 75% of the time if not monitored, which prompted the PAD guidelines ‘recommendation that all ICU patients be screened regularly for delirium with a valid and reliable tool.[1,33,34]
Several methods have been developed and validated to diagnose delirium in ICU patients but the Intensive Care Delirium Screening Checklist (ICDSC) and the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU) are the most frequently employed tools for this purpose.[35] The CAM-ICU was translated and validated in numerous languages.[36-42] In 2011 Heo et al.[42] translated and validated the CAM-ICU for use in the Korean ICU setting. Two nurses and one psychiatrist independently evaluated 22 patients. The Korean CAM-ICU showed an high interrater reliability(κ = 0.81, p < 0.001). The sensitivities and the specificities of the two nurses’ evaluations were 89.80% and 72.40% for nurse 1 and 77.40% and 75.80% for nurse 2 respectively. The CAM-ICU can be completed in less than 1 minute and can be used in both verbal and nonverbal patients. The CAM-ICU measures 4 features of delirium: (1) acute change or fluctuation in mental status from baseline; (2) inattention, the cardinal and universal feature; (3) altered level of consciousness; and (4) disorganized thinking. A patient screens positive for delirium if features 1 and 2 are present along with either feature 3 or 4. Overall accuracy of the CAM-ICU is excellent, with pooled values for sensitivity and specificity of 80% and 95.9%, respectively.[35] The ICDSC is an 8-item checklist completed over an 8- to -24 hour period. One point is given for each checklist item present. The 8 items are level of consciousness, inattention, disorientation, hallucinations/delusions/psychosis, psychomotor agitation or retardation, inappropriate speech or mood, sleep-wake cycle disturbances, and symptom fluctuation. A score of 4 points or more constitutes a positive ICDSC and the presence of delirium. The pooled values for the sensitivity and specificity of the ICDSC are 74% and 81.9%, respectively.[35]
Regardless of what approach is taken with delirium, it can be very advantageous for the team to have a standardized, agreed-upon method of considering the differential diagnostic causes of a patient’s delirium (Fig. 2). For example, if delirium is present, the clinical team can briefly consider the most common risk factors using a simple mnemonic called the “Dr. DRE” (Fig. 3).
After reversible causes and modifiable risk factors have been addressed and nonpharmacologic strategies have been implemented, then and only then should pharmacologic interventions be considered.
There are no published data determining a positive effect of haloperidol or atypical antipsychotics on decreasing the duration of delirium in adult ICU patients. The Modifying the Incidence of Delirium (MIND) study showed no difference in the duration of delirium between haloperidol, ziprasidone, or placebo when used for prophylaxis and treatment.[43] A small, prospective, randomized, double-blind, multicenter, placebo-controlled study randomized 18 patients to scheduled quetiapine and 18 patients to placebo.[44] The quetiapine group had a faster resolution of delirium compared with the placebo group (1 day vs. 4.5 days, p = 0.001) and a shorter duration of delirium (36 days vs. 120 days, p = 0.006). The quetiapine group required fewer days of as-needed haloperidol (3 vs. 4 days). Kim et al.[45] performed an open trial of olanzapine in a medico-surgical population of 22 Korean patients with delirium, evaluated by Delirium Rating Scale (DRS). In this study olanzapine significantly improved i the scores of DRS from 20.0 ± 3.6, at the time of pretreatment, to 9.3 ± 4.6 at the post-treatment with doses of 5.9 ± 1.5 mg/day without serious side-effects. Ely et al.[46] are conducting the MIND-USA (Modifying the Impact of ICU-Induced Neurological Dysfunction-USA) Study (ClinicalTrials.gov Identifier NCT01211522) to define the role of antipsychotics in the management of delirium in vulnerable critically ill patients.
Delirium prophylaxis with medications is discouraged in the PAD guidelines. Recently Wang et al.[47] in a prospective, randomized, multicenter trial compared a low dose haloperidol infusion administered for 12 hours (0.5 mg intravenous bolus injection followed by continuous infusion at a rate of 0.1 mg/h) for 12 hrs, n = 229 pts) vs. placebo (n = 228 pts) in the immediate postoperative period, showing that haloperidol could reduce the incidence of delirium within the first 7 days postoperatively in patients undergone noncardiac surgery (15.3% in the haloperidol group vs. 23.2% in the control group (p = 0.031). By contrast, the HOPE ICU study showed no benefit of early administration of intravenous haloperidol in a mixed population of medical and surgical adult ICU patients.[48] In this double-blind, placebo-controlled randomised trial 142 patients were randomized to receive haloperidol or placebo intravenously every 8 h irrespective of coma or delirium status. Patients in the haloperidol group spent about the same number of days alive, without delirium, and without coma as did patients in the placebo group (median 5 days [IQR 0-10] vs. 6 days [0-11] days; p = 0.53).[48]
Perhaps the most important element of delirium management in the ICU setting involves communication among the team. To that end, many ICU teams now present on rounds using the “brain road map,” which is a set of data containing 3 elements (Fig. 4). At each bedside, the nurse (or another team member such as an intern or a pharmacist) will present the patient’s (1) target RASS or SAS; (2) actual RASS or SAS, CAM-ICU, or ICDSC result; and (3) sedatives and narcotics received. This helps the team discuss the patient’s cognitive status, compare it with the patient’s desired cognitive status for that day, determine adjustments needed, and then explore the causes of delirium if the patient is CAM-ICU or ICDSC positive that day (using, for example, the Dr. DRE tool shown in Fig. 3).
E: Early Mobility and Exercise
Early mobility is an integral part of the ABCDEF bundle and has been the only intervention resulting in a decrease in days of delirium.[49]
The consequence of physical dysfunction in critically ill patients can be profound and long-term with significant reduction in functional status being observed even 1 year and 5 years after ICU discharge.[50-52]
Physical therapy has shown to be feasible, safe, even in the most complicated patients receiving the most advanced medical therapies (e.g., continuous renal replacement therapy, extracorporeal cardiopulmonary support).[53,54] Early activity can be done without increases in usual ICU staffing and with a low risk (< 1%) of complications.[55] Schweickert et al.[56] showed that a daily SAT combined with physical and occupational therapy, versus SAT alone, resulted in an improved return to independent functional status at hospital discharge, shorter duration of ICU-delirium, higher survival, and more days breathing without assistance. However, in a study where ICU patients were enrolled 4 days after the initiation of mechanical ventilation (average 8 days), an intensive physical therapy program did not improve long-term physical functioning when compared to a standard of care program.[57]
The focus on rehabilitation of critically ill patients should begin as early as possible in the ICU and continue all the way to recovery at home.
F: Family Communication and Involvement
The ABCDE bundle has evolved to include Family Engagement, as no ICU treatment plan is complete without incorporation of the family’s wishes, concerns, questions, and participation.
Family members should be educated about delirium and its potential deleterious effects on the patient (post-traumatic stress disorder, depression, cognitive impairment) upon discharge from the hospital. Family members themselves may also suffer long-term psychological complications such as anxiety, depression, and posttraumatic stress disorder, commonly persisting for years. Frequent and effective communication and family presence on rounds have been recommended to maximize support to family members.[58] Family presence has been encouraged in traumatizing medical events and procedures, such as Cardiopulmonary Resuscitation (CPR). In some studies, the family presence during CPR is associated with positive results on psychological variables, and did not interfere with medical efforts, increase stress in the health care team, or result in medicolegal conflicts.[59]
Increased focus on communication with family members, through routine ICU family conferences, palliative care consultation, or ethics consultation can reduce ICU length of stay for those patients whose trajectory is ultimately mortal.[60-63] One study of communication occurring during ICU family conferences sought to understand how ICU clinicians conduct communication concerning withdrawing life-sustaining treatments or the delivery of bad news, and how this communication might be improved.[64] Most clinicians failed to listen and respond appropriately, failed to acknowledge the expression of family members’ emotions, and failed to explain key tenets of palliative care. An important missed opportunity when communicating with families is exploring patient treatment preferences that are fundamental to clinical decision making in the ICU setting.[64]
Critical illness usually impacts not only an individual, but their entire support system, family, friends or other caregivers who are actively engaged in supportive roles. In light of this, it is crucial to recognize the needs of the patient and of their family as well.
Conclusions
For the sake of our patients’ well-being, we must use the ABCDEF bundle to incorporate delirium awareness, monitoring, prevention, and treatment into ICU health systems. The ABCDEF bundle represents one method to approach the organizational changes needed to shift our culture for optimizing our patient recovery and outcomes. Studies show that there are negligible adverse consequences of implementing these recommended strategies and minimal costs associated with changing commonly prescribed medications, and no evidence of adverse short or long-term psychiatric or neuropsychological effects of minimizing sedation exposure, so we need to change our way to treat patient focusing our resources on having more interactive patients with well-controlled pain who can participate in physical and cognitive activities at the earliest possible safe point in their critical illness.
Notes
No potential conflict of interest relevant to this article was reported.
Acknowledgements
EWE is supported by National Institutes of Health HL111111 (Bethesda, MD), by the Veterans Affairs Tennessee Valley Geriatric Research, Education and Clinical Center (Nashville, TN). EWE is supported by the VA Clinical Science Research and Development Service (Washington, DC) and the National Institutes of Health AG027472 and AG035117 (Bethesda, MD). EWE has received honoraria from Abbott Laboratories, Hospira, Inc., and Orion Corporation, and research grants from Abbott EWE have received research grants from Hospira, Inc.