2021 KSCCM clinical practice guidelines for pain, agitation, delirium, immobility, and sleep disturbance in the intensive care unit
Article information
Abstract
We revised and expanded the “2010 Guideline for the Use of Sedatives and Analgesics in the Adult Intensive Care Unit (ICU).” We revised the 2010 Guideline based mainly on the 2018 “Clinical Practice Guidelines for the Prevention and Management of Pain, Agitation/Sedation, Delirium, Immobility, and Sleep Disruption (PADIS) in Adult Patients in the ICU,” which was an updated 2013 pain, agitation, and delirium guideline with the inclusion of two additional topics (rehabilitation/mobility and sleep). Since it was not possible to hold face-to-face meetings of panels due to the coronavirus disease 2019 (COVID-19) pandemic, all discussions took place via virtual conference platforms and e-mail with the participation of all panelists. All authors drafted the recommendations, and all panelists discussed and revised the recommendations several times. The quality of evidence for each recommendation was classified as high (level A), moderate (level B), or low/very low (level C), and all panelists voted on the quality level of each recommendation. The participating panelists had no conflicts of interest on related topics. The development of this guideline was independent of any industry funding. The Pain, Agitation/Sedation, Delirium, Immobility (rehabilitation/mobilization), and Sleep Disturbance panels issued 42 recommendations (level A, 6; level B, 18; and level C, 18). The 2021 clinical practice guideline provides up-to-date information on how to prevent and manage pain, agitation/sedation, delirium, immobility, and sleep disturbance in adult ICU patients. We believe that these guidelines can provide an integrated method for clinicians to manage PADIS in adult ICU patients.
INTRODUCTION
Critically ill adult patients treated in an intensive care unit (ICU) are exposed to an unpleasant environment, with uncontrolled light and noise. They experience moderate to severe pain at rest, as well as during general care procedures (such as mechanical ventilation and invasive procedures, nursing care, and trauma or comorbidity-induced pain). Severe pain can induce various stress responses in critically ill patients and lead to agitation, sleep disturbance, and delirium. A deep level of sedation and delirium is related to detrimental clinical outcomes such as longer ICU length of stay (LOS), a longer duration of mechanical ventilation, and increased mortality. Therefore, a multidisciplinary strategy, including optimizing pain management, maintaining light sedation, performing routine monitoring and treatment for delirium, providing active rehabilitation in the ICU, and treating sleep disturbance using a sleep-promoting protocol, needs to be implemented to improve clinical outcomes of critically ill ICU patients. Furthermore, the appropriate administration of medications for pain, sedation, and delirium has varied among clinicians. Improper pharmacologic interventions might be associated with negative outcomes in critically ill adult patients.
The Standardization Committee in the Korean Society of Critical Care Medicine (KSCCM) developed the “2010 Guideline for the Use of Sedatives and Analgesics in the Adult Intensive Care Unit” to assist clinicians with the provision of adequate treatment for pain and sedation in the ICU. Since the 2010 guideline was published, there have been significant advances in the therapeutic approach and management for pain, agitation, and delirium (PAD) in critically ill ICU patients. In addition, as our knowledge has expanded, we have come to understand that rehabilitation/mobilization may be beneficial for delirium treatment, and that sleep is a modifiable risk factor influencing the recovery of critically ill patients.
Herein, we report recommendations regarding how to prevent and manage pain, agitation/sedation, delirium, rehabilitation/mobilization, and sleep disturbance in critically ill ICU patients by updating the “2010 Guideline for the Use of Sedatives and Analgesics in the Adult Intensive Care Unit.”
MATERIALS AND METHODS
Selection of Panel Members
Board members of the KSCCM appointed an editor for the revision and update of the “2010 Guideline for the Use of Sedatives and Analgesics in the Adult Intensive Care Unit.” The panel members of the Guideline Committee were recruited from the members of the KSCCM. The KSCCM approved all panelists. The panelists included two surgeons, a neurosurgeon, and an anesthesiologist, and all panelists were intensivists. None of the panelists had any conflicts of interest with the related topic.
Guideline Development
The 2010 guidelines were not published in a specific journal, but were released as a booklet during the 30th KSCCM Annual Congress and posted on the KSCCM homepage. We revised the “2010 Guideline for the Use of Sedatives and Analgesics in the Adult Intensive Care Unit” based mainly on the 2018 “Clinical Practice Guidelines for the Prevention and Management of Pain, Agitation/Sedation, Delirium, Immobility, and Sleep Disruption (PADIS) in Adult Patients in the ICU,” which was an updated 2013 PAD guideline with the inclusion of two additional topics (rehabilitation/mobility and sleep). Because it was not possible to hold face-to-face meetings of the panels due to the coronavirus disease 2019 (COVID-19) pandemic, all discussions took place via virtual conference platforms and e-mail with the participation of all panelists. All authors drafted the recommendations, and all panelists discussed and revised the recommendations several times. All panelists voted on the quality level of each recommendation, and each recommendation was required to have at least 75% agreement with a 100% response rate of the entire panel to be included in the final guideline. The development of this guideline was independent of any industry funding.
Assessing Quality of Evidence
The quality of evidence for each recommendation was ranked as high (level A), moderate (level B), or low/very low (level C) based on both study design and specific study characteristics that could prompt a reviewer to either downgrade or upgrade the quality of the evidence. The levels were defined as follows. Level A: Scientific evidence provided by well-designed, well conducted, controlled trials (randomized or nonrandomized) with statistically significant results that consistently support the recommendation of the guideline; Level B: Scientific evidence provided by observational studies or by controlled trials with less consistent results to support the recommendation of the guideline; Level C: Although expert opinion supports the recommendation of the guideline, scientific evidence provides inconsistent results, or such evidence is lacking.
Peer Review
External reviewers who were not involved in the development of the guideline reviewed it before it was published. The final manuscript was reviewed and approved by the Board of the KSCCM.
RESULTS
Pain
According to the definition of the International Association for the Study of Pain, pain is an unpleasant sensory and/or emotional experience related to real or potential tissue injury [1]. Pain management is complicated because pain is highly individual. It can arise from diverse origins, including neuropathic, visceral, and somatic causes. Each patient has subjective perceptions of pain with a different degree of endurance. Critically ill patients in the ICU experience moderate to severe pain not only from standard care procedures, but also at rest. Severe pain can induce stress responses such as tachycardia, increased oxygen consumption in the myocardium, hypercoagulation, respiratory compromise, immunosuppression, and increased catabolism, leading to tissue perfusion disorders and reduced tissue oxygen partial pressure. Thus, appropriate pain control in ICU patients is indispensable. The implementation of assessment-driven and standardized pain management is vital due to the characteristics of critically ill patients, such as communication disorders, altered mental status, mechanical ventilation, invasive procedure and devices, and immobility. Compared to conventional treatment, protocol-based pain control and sedation may reduce nosocomial infections, hypotension, bradycardia, the use of sedatives without increased narcotic analgesics, the duration of mechanical ventilation, ICU LOS, and severity of pain [2,3]. Thus, it is recommended to regularly conduct a protocol-driven pain assessment using validated tools and provide a definite guideline for medication selection and dosage. In addition, step-by-step pain management that prioritizes pain control prior to the administration of sedatives is recommended.
Risk Factors
KSCCM Recommendation
▪ All adult ICU patients have the right to receive adequate analgesia and pain management. (grade A)
ICU patients commonly complain of moderate to severe pain and physical discomfort even at rest due to various factors, such as one or more comorbidities, regular nursing care, invasive procedures, and various injuries [4]. Psychological factors, such as anxiety and depression, and demographic and clinical factors such as young age, various comorbidities, and operation history can influence pain at rest [5]. In particular, procedures frequently performed in critically ill adults, including insertion of an arterial catheter, removal of the chest tube and wound drain, position change and turning, and endotracheal suction, are associated with the greatest increases in pain intensity. Pain severity before treatment, the procedure type, diagnosed traumatic and surgical injuries, and demographic characteristics, including young age, female sex, and non-Caucasian race, influence the intensity of pain during a procedure. Inadequate pain treatment causes sleep deprivation, exhaustion, and disorientation, as well as agitation, which is often observed in ICU patients [6].
Assessment
KSCCM Recommendation
▪ We recommend that clinicians should perform routine and reproducible pain assessments and evaluate responses to pain treatment using the patient’s self-report or Behavioral Pain Scales (BPS). (Grade B) [Update]
▪ A patient’s self-report of pain intensity is a reference standard for evaluation and treatment of pain. The 0–10 Numeric Rating Scale (NRS) is recommended for pain assessment. (Grade B)
▪ The most reliable tools for pain assessment in adult ICU patients who cannot self-report are the BPS and the Critical-Care Pain Observation Tool (CPOT). (Grade B) [Update]
For patients who can communicate, the most reliable and appropriate method of pain assessment is self-reporting. The assessment of pain should consider the characteristics of pain such as site, feature, aggravating or alleviating factors, and intensity. The 0–10 NRS is a scale in which the patient selects the intensity of pain from 0 to 10 corresponding to their pain, with a score of 3 or less indicating adequate pain control and a score of 10 indicating the worst pain. The 0–10 NRS (in verbal or visual format) for adult ICU patients who can self-report is an effective and feasible method [7].
The Verbal Descriptor Scale (VDS) is a descriptive pain assessment scale using a series of descriptive phrases referring to different levels of pain intensity, with a horizontal line of 10 cm indicating “no pain,” “very severe pain,” or “extremely severe pain” at each end. It may be applied in ICU patients who cannot use a numeric assessment scale such as the 0–10 NRS. In one study, ICU patients undergoing cardiac surgery stated that the 0–10 NRS or VDS was superior to the visual analog scale in identifying pain intensity and preferred to use the VDS for pain assessment [8]. The 0–10 Faces Pain Scale, which was validated in 105 postoperative cardiac surgery ICU patients, is easy to use and useful for identifying pain intensity and shows a good correlation with the VDS (Figure 1) [9].
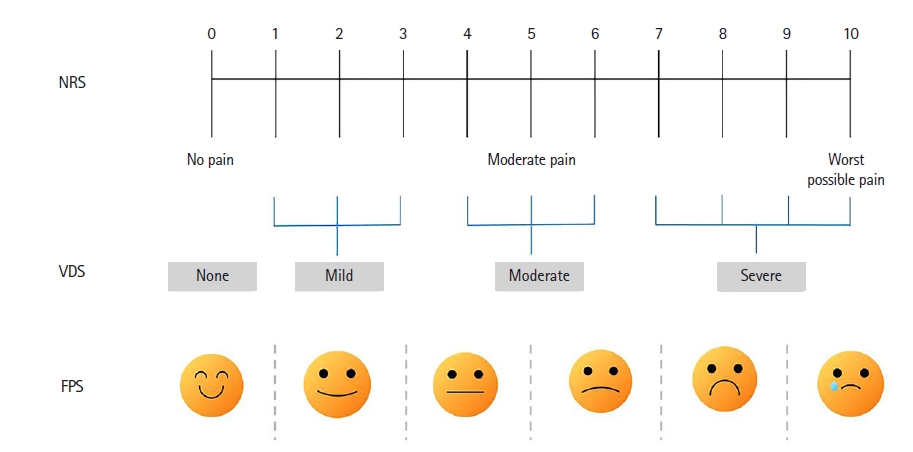
Various pain assessment scales: Numeric Rating Scale (NRS), Verbal Descriptor Scale (VDS), and Faces Pain Scale (FPS).
Critically ill ICU patients often become unable to express pain intensity while receiving sedatives, anesthetics, neuromuscular blockers, and mechanical ventilation. In critically ill adults who cannot self-report pain with observable behaviors, the BPS and CPOT demonstrate the greatest validity and reliability for monitoring pain (Tables 1 and 2) [10,11]. When a patient cannot self-report, the family can participate in the patient’s pain assessment, and family-reported pain has been shown to be closer to the patient’s self-report than the reports of attending nurses and physicians [12]. However, the degree of agreement between the family and the patient for pain intensity is moderate. Compared with seriously ill patients’ self-reports, surrogates tended to overestimate pain intensity. Thus, family participation in pain assessment cannot replace the role of attending nurses and physicians [13]. Vital signs including heart rate, blood pressure, respiratory rate, oxygen saturation, and end-tidal CO2 are not reliable parameters for pain monitoring and assessment in adult ICU patients. They can be considered as a signal to begin a further evaluation using reliable and adequate tools, including a patient’s self-report or behavioral scale (BPS and CPOT) [14].
Choice of analgesia
The selection of appropriate analgesia should be preceded by nonpharmacologic therapy that can maintain patients’ comfort through a proper body posture, fixation of fractures, and removal of other physical stimuli. Nonpharmacologic therapy can reduce the unnecessary use of analgesia and maximize the effect of analgesia. Other nonpharmacologic interventions to reduce pain include massage, cold therapy, and relaxation therapy. However, their level of evidence supporting their recommendation is low.
Various pain-modulating medications such as opioids, non-steroidal anti-inflammatory drugs (NSAIDs), acetaminophen, and anticonvulsants can be used to manage pain in adult ICU patients (Figure 2). To select an optimal analgesic and its dosage suitable for individual patients, the drug’s pharmacologic properties and adverse effects should be considered. Local analgesia, volatile anesthetics, NSAID gels, and non-opioid drugs or NSAIDs, whether intravenous (IV) or orally administered, are not recommended for use before an ICU procedure to avoid pain originating from the procedure.

Flowchart of pain management for critically ill patients in the intensive care unit. aOpioids: morphine, hydromorphone, fentanyl, and remifentanil; bSide effects: respiratory depression, coma, lower gastrointestinal tract paralysis/ileus, hyperalgesia, and immunosuppression; cKetorolac: recommended for use within 5 days; dNeuropathic agents: gabapentin, pregabalin, and carbamazepine.
Opioids
KSCCM Recommendation
▪ We recommend that the treatment plan and the goal of optimal pain management should be determined individually or personalized and that all participating clinicians should share this information for consistent analgesic therapy. (Grade C)
▪ If continuous IV administration of opioids is required, fentanyl, hydromorphone, and remifentanil are recommended. (Grade C) [Update]
IV opioids are the most important treatment for non-neuropathic pain in adult ICU patients. Opioids act on several central and peripheral opium receptors to mediate the analgesic effect. Among those receptors, μ and κ receptors are the most important. The activities of these two receptors and other receptors are related to side effects. The prerequisite conditions for ideal opioid therapy include rapid onset of action, easy dose titration, no accumulation of the drug or its metabolites in the body, and low cost. Commonly used opioids in adult ICU patients include fentanyl, morphine, hydromorphone, and remifentanil [15]. In particular, remifentanil has the advantages of rapid onset/offset action and safety regardless of liver and kidney dysfunction. Thus, remifentanil can be applied through continuous IV administration in patients requiring frequent awakening (Table 3) [16].
Non-opioid analgesics
KSCCM Recommendation
▪ A “multi-modal analgesia” strategy is likely to reduce opioid consumption with improved pain-modulating effects, pain control, and patient-centered outcomes. (Grade C) [Update]
▪ NSAIDs, acetaminophen, and nefopam can be used as adjunctive opioid analgesics in critically ill adults. (Grade C) [Update]
▪ Gabapentin, carbamazepine, and pregabalin can be used with an opioid for neuropathic pain management. (Grade A) [Update]
▪ Low-dose ketamine can be used as an adjunct to an opioid for pain management. (Grade C) [Update]
IV opioids are the mainstay of pain treatment in most ICUs. However, their adverse effects, including sedation, delirium, respiratory depression, and ileus, might prolong the ICU LOS and aggravate post-ICU patient results. A “multi-modal analgesia” strategy is needed to reduce opioid use and increase the pain-modulating effects, improving pain control and patient-centered outcomes [17]. Non-opioid analgesics are able used as adjunctive painkillers to decrease the dosing of opioids. Their use should be adjusted according to each patient’s conditions and symptoms to reduce the risk (Table 4).
Few NSAIDs have analgesic effects similar to those of opioids. Ketorolac (a non-COX-1-specific NSAID) is currently the only NSAID approved for IV use as an alternative to opioids for pain management. The analgesic effect of ketorolac after intramuscular (IM) injection begins to appear at 1 hour, reaching its maximum effect at 2 hours. It lasts 5 to 6 hours. A single 30-mg dose of IM ketorolac shows an equivalent effect to a single 4-mg dose of IV morphine. Although ketorolac can be used as a single agent, its administration together with opioids can reduce the opioid dose by 25%–50%. Acetaminophen and nefopam can be used as adjunctive analgesics in critically ill patients to reduce opioid use and improve analgesic effectiveness [18]. Nefopam exerts an analgesic effect by inhibiting dopamine, noradrenaline, and serotonin reuptake. A 20-mg dose shows an equivalent effect to a 6-mg dose of IV morphine [19]. Ketamine can improve pain relief and reduce opioid requirements by reducing hyperalgesia at doses lower than an anesthetic dose. An adjunctive dose of ketamine is common at a 0.1–0.5 mg/kg IV bolus followed by a 1–2 μg/kg/min continuous IV infusion. For neuropathic pain management in critically ill patients, neuropathic analgesics such as gabapentin, carbamazepine, and pregabalin should be used with an opioid [20]. Lidocaine and COX-1-selective NSAIDs should not be used routinely as adjuncts to opioids for pain management in adult ICU patients.
Methods of dosing analgesics
KSCCM Recommendation
▪ Continuous IV infusion or an intermittent scheduled-administration strategy of analgesics is more useful for retaining consistent analgesic action than administration upon patient demand. (Grade B)
▪ A patient-controlled analgesia (PCA) device helps administer opioids to patients who can understand and handle the device. (Grade B)
▪ Fentanyl or remifentanil for rapid pain management is useful for patients with acute conditions. (Grade C) [Update]
▪ Fentanyl, hydromorphone, or remifentanil is useful for patients with hemodynamic instability or renal failure. (Grade C) [Update]
▪ It is recommended to use morphine and hydromorphone with longer durations of action if a patient inevitably needs intermittent boluses. (Grade C)
In principle, analgesics should be administered as a continuous IV infusion unless there is a particular reason not to do so. A continuous IV infusion can maintain a constant blood concentration and avoid drug toxicity since a lower dose is administered at one time than in a single bolus administration. In addition, IV administration has the advantage of making it possible to adjust the blood concentration according to the infusion rate. As an alternative option, PCA, which refers to a system when an analgesic is administered upon the patient’s need, has the disadvantage of administering a smaller dose than the prescribed dose, which can cause a severe delay in pain management [21]. Daily awakening of patients from analgesics and sedation can effectively manage pain intensity, reduce total opioid consumption, and shorten the ICU LOS and the duration of mechanical ventilation [22]. PCA used in non-critical patients shows a stable drug concentration, high-quality analgesia, less sedation, reduced opioid consumption, and low incidence of respiratory complications compared to other opioid administration strategies. When considering PCA, clinicians should pay attention to the patient’s consciousness, hemodynamic reservoir, and history of opioid abuse [23]. A transdermal fentanyl patch is useful for long-term analgesia in hemodynamically stable patients, as it can release the drug uniformly, although the degree of absorption varies according to permeability, body temperature, tissue perfusion, and skin thickness. The maximal blood concentration differs significantly between patients. Since a fentanyl patch takes 12 to 24 hours to reach maximal effectiveness, it is not recommended for rapid pain control. Moreover, it takes a similar amount of time for the drug effect to disappear after removing the patch.
Adverse effects of analgesics
KSCCM Recommendation
▪ Opioids should be used cautiously due to respiratory depression, opioid-induced hypotension, and reduced bowel motility. (Grade C)
▪ Ketorolac administration should be used for up to 5 days. Clinicians should carefully monitor for possible renal failure and gastrointestinal bleeding. (Grade C)
▪ IV acetaminophen should be used cautiously due to IV acetaminophen-associated hypotension, which can occur in up to 50% of patients. (Grade C) [Update]
Opioids
The side effects of opioid analgesics are respiratory depression, effects on the cardiovascular system, and changes in bowel motility. Opioids can cause a dose-dependent decrease in the respiratory rate and tidal volume. They can also reduce blood pressure and the heart rate by inhibiting the sympathetic nervous system and enhancing the parasympathetic nervous system. However, opioid-induced hypotension responds well to fluid therapy or vasopressors in most cases. Naloxone at 4 to 8 mg can antagonize the decreased bowel motility caused by opioids without antagonizing the analgesic effect. A small dose of naloxone (0.25 to 1 μg/kg/hr) can control opioid-induced pruritis that does not respond to an antihistamine without antagonizing the analgesic effect. Opioids can stimulate chemical receptors in the vomiting center of the brainstem, thus inducing vomiting. All opioids have the same degree of emetogenic effect. However, if one drug causes vomiting, switching to another drug can sometimes improve symptoms. Serotonin antagonists and low-dose opioid antagonists can also help relieve symptoms. Opioids can increase muscle tone and evoke muscle rigidity in severe cases. Regarding the mechanism, opioids act on the spinal cord or supraspinal level of the central nervous system, not directly on the muscle [24]. Severe muscle rigidity can induce a decrease in lung elasticity and a decline of functional residual capacity of the lung, leading to ventilation impairment [25]. Non-depolarizing muscle relaxants can reduce the severity of muscle rigidity. Thiopental and low-dose diazepam or midazolam can decrease and prevent muscle rigidity [26].
Non-opioid analgesics
Non-opioid analgesics occasionally cause gastrointestinal bleeding or surgical wound bleeding and suppress renal function by interfering with prostaglandin synthesis in the kidneys. NSAID-associated renal injuries frequently occur in patients with hypovolemic status, renal hypoperfusion due to old age, and renal dysfunction. In addition, ketorolac use for over 5 days more than doubles the risk of renal toxicity and bleeding. When acetaminophen is administered through the IV route, hypotension (a reduction in mean arterial pressure >15 mm Hg) may occur in up to 50% of patients [27]. Nefopam should be used selectively due to its association with tachycardia, glaucoma, seizure, and delirium. The side effects of ketamine include nausea, delirium, hallucination, hypoventilation, pruritus, and sedation, and have an incidence similar to that of opioid side effects.
Agitation/Sedation
In critically ill patients, sedatives are frequently administered to relieve anxiety, reduce the stress of dependence on mechanical ventilation, and control agitation [28]. Agitation can cause difficulties with mechanical ventilation, hypoxia due to increased oxygen consumption, barotrauma, hypotension, accidental removal of instruments or catheters, and in-hospital infections. However, sedatives can increase patients’ morbidity. Thus, specific reasons are required to use them. Patients’ sedation status must be frequently reassessed using valid and reliable scales while using sedatives [29,30].
Goals for the sedation level
KSCCM Recommendation
▪ Clinicians should establish a sedation level and time to discontinue sedatives in each patient and regularly adjust those targets. (Grade C)
▪ The sedative dose can be titrated to maintain a light rather than a deep level of sedation in critically ill patients on mechanical ventilation unless clinically necessary. (Grade B) [Update]
▪ A light level of sedation can be achieved and maintained using daily sedation interruption (DSI) and a nurse-protocolized targeted sedation protocol. (Grade C) [Update]
An appropriate sedation level depends on the patient's disease course and treatment regimen. Since the prognosis is poor when deep sedation occurs in the ICU, other than in exceptional cases, a light sedation state wherein the patient can be easily awakened while maintaining a normal sleep-wake cycle is desired rather than deep sedation. Although there is no consensus on the definition of the depth of sedation (light, moderate, or deep), light sedation usually ranges from –1 to –2 on the Richmond Agitation-Sedation Scale (RASS) [22]. However, a RASS score in the –2 to +1 range has also been considered light sedation in a few studies [30]. Maintaining light sedation shortens the weaning time, reduces the frequency of tracheostomy, and shortens the ICU LOS. Contrary to the 2013 guidelines, however, the maintenance of light sedation does not reduce 90-day mortality, delirium, post-traumatic stress disorder, or depression or increase unplanned self-extubation [31,32]. In critically ill intubated adult patients, a light level of sedation can be achieved and maintained using DSI and nurse-protocolized targeted sedation protocol (NP-targeted sedation). DSI attempts to help patients arouse and be weaned from mechanical ventilation. NP-targeted sedation is a sedation protocol applied by nurses who adjust drug concentration at the bedside to achieve target sedation scores [33,34].
Sedatives can relieve patients’ stress and help facilitate general procedures in the ICU. They are essential for the treatment process of critically ill patients, as they can dramatically help maintain patients’ safety and comfort. However, to prevent side effects caused by excessive use, patients’ sedation levels should be assessed. After an assessment, continuous dosing is recommended whenever possible to achieve an appropriate level of sedation. If necessary, intermittent dosing may be used. The target sedation level should be determined at the start of treatment depending on the patient's condition. It should be evaluated and adjusted again from time to time. Consideration should be given to writing sedative prescriptions in such a way that the daily sedative requirement may be adjusted up and down.
Assessment of the sedation level
KSCCM Recommendation
▪ We recommend using the RASS and Sedation-Agitation Scale (SAS) to assess the degree and level of sedation. (Grade B) [Update]
▪ Sedation monitoring based on the bispectral index (BIS) rather than a subjective scale may facilitate titration of sedation when a sedation assessment scale cannot be used due to deep sleep or muscle relaxants. (Grade C) [Update]
There are various sedation assessment tools, including the RASS, Ramsay scale, Riker SAS, Motor Activity Assessment Scale (MAAS), and Observer Assessment of Alertness/Sedation (OAA/S) scale (Table 5) [35]. Among them, the RASS and SAS are widely used to assess the degree and depth of sedation in ICU patients. The SAS was the first sedation level assessment method with confirmed reliability. It consists of seven items describing patient behavior [36]. The RASS is a tool that evaluates arousal state, cognitive function, and response sustainability based on a score of –5 to +4. It is the most effective and stable sedation assessment tool, along with the SAS [37]. The MAAS is a modified version of the SAS. It consists of seven categories of patient behavior. Its reliability has been verified as a method of evaluating the sedation level of critically ill patients [38]. The OAA/S scale indicates the degree of response to pinching or calling a name on a scale of 0 to 5. It is known to have significant sensitivity to the degree of sedation [22].
Objective sedation monitoring is useful when a patient’s behavior cannot be observed due to deep sedation or therapeutic muscle relaxants. Methods for objectively measuring brain function include auditory evoked potentials, the BIS, the patient state index, and state entropy. These methods are not recommended for critically ill patients who are not unconscious or paralyzed. However, recent studies have shown that using BIS may have a potential effect on patients with light sedation, such as reducing the need for tracheostomy, treatment-related side effects, total sedative and fentanyl doses, and shortening the ICU stay [39,40]. Vital signs are not specific or sensitive for assessing the sedation level.
Sedatives
The medications used for sedation include benzodiazepines, propofol, central α2-agonists, and other agents (Table 6). Indications, goals, clinical pharmacology, and cost of sedatives are important determinants when selecting sedatives.
Benzodiazepines
Benzodiazepines can cause anterograde amnesia, which interferes with the memory of unpleasant experiences that occur after drug use. They do not cause retrograde amnesia. They have an anticonvulsant effect without an analgesic action. Nonetheless, they can lower the expected pain response. Thus, the analgesic dose can be reduced when benzodiazepines are used together with analgesics [41]. Their main indications are sedative treatment and short-term use for anxiety, panic disorder, alcohol withdrawal, preoperative anxiety, initial treatment of convulsions, muscle spasms, and insomnia. Benzodiazepine drugs have many differences in efficacy, onset, duration of action, metabolism, and the presence of active intermediates. The intensity and duration of effects may vary depending on age, underlying diseases, past alcohol abuse history, medication history, and other factors. Thus, the appropriate dose may vary. In elderly patients, the clearance of benzodiazepines (and their intermediate metabolites) is slow, or benzodiazepines have a larger volume of distribution. Thus, the drug clearance time may be significantly longer in elderly patients than in younger patients [42]. Liver failure or renal insufficiency can also slow drug clearance [43]. In case of a benzodiazepine overdose, flumazenil can be used as an antagonist to reverse its excessive sedative and hypnotic effects. A dose of 0.3−2.0 mg should be administered in divided IV injections [44]. However, its use is not recommended because it may increase withdrawal symptoms and myocardial oxygen consumption. When testing for continuous sedation, it is recommended to use a single shot at a low dose.
Benzodiazepines include midazolam, lorazepam, and diazepam. Midazolam is effective for acute excitatory patients because of its rapid onset time. However, it has been reported that its sedative effect continues to appear in critically ill patients with obesity, low serum albumin, or renal failure [45,46]. Its long-term sedative effects can also occur due to the accumulation of active metabolites such as α-hydroxymidazolam, especially in patients with renal failure [46]. The metabolism of midazolam is significantly inhibited by propofol, diltiazem, macrolide antibiotics, and other cytochromes P450 isoenzyme 3A4 antagonists, prolonging the duration of its drug action [43]. Midazolam has high level of fat solubility. It can act very quickly and produce a sedative effect within 2–5 minutes of an IV bolus injection. However, it is not possible to induce a normal sleep pattern. Continuous IV administration of midazolam can cause excessive sedation due to its accumulation in tissues. Thus, it is recommended to limit the administration of midazolam to within 48 hours. With daily awakening, drug interruption, and dose re-optimization according to the RASS, it is possible to reduce the drug demand and shorten the length of ventilation and ICU stay [47]. Lorazepam has a slow onset of action, making it difficult to use in patients with acute anxiety. It has a long half-life of 12–15 hours, making it difficult to quickly optimize the dose during continuous infusion. Nonetheless, it has few interactions with other drugs and sedation can be maintained by intermittent administration or continuous infusion. The initial loading dose should be administered through an IV push of a set dose. It should be diluted to a concentration of 1 mg/ml or less. Precipitation may occur despite these precautions. When lorazepam is administered at a high dose, polyethylene glycol and propylene glycol (PG) as solvents are known to cause acute reversible renal tubular necrosis, lactic acidosis, and a hyperosmolar state. PG toxicity refers to significant accumulation of PG when the difference in osmotic pressure before and after lorazepam administration is 10–12 mOsm/L or more. Diazepam provides a rapid onset of sedation and rapid awakening when it is administered as a single dose. Since its metabolite has a long-term effect, the sedation state can be sustained during repeated administration. Thus, diazepam can be used for long-term sedative treatment.
Propofol
Propofol is administered through IV injections for general anesthesia and sedation. It has sedative, sleep-inducing, anti-anxiety, memory loss, antiemetic, and anticonvulsant effects. However, it has no analgesic effect. It can reduce cerebral blood flow and brain metabolism. It can also lower intracranial pressure more effectively than fentanyl in cases of severe brain injury. It is used as a sedative in patients with elevated intracranial pressure. No pharmacokinetic changes have been reported in patients with renal or hepatic failure. Due to its high fat solubility, propofol can quickly cross the blood-brain barrier, leading to rapid sedation. Its sedative effect is lost quickly after short-term use due to its rapid redistribution and high liver and extrahepatic clearance. Because its sedative effect dissipates quickly, temporary discontinuation during infusion allows a neurological evaluation, which is advantageous when frequent waking for neurological examinations is required or when performing a daily waking protocol. However, there are reports showing that awakening is delayed after 12 hours of infusion [48]. Thus, caution is needed. Propofol can induce injection site pain, bradycardia, respiratory depression, and dose-dependent hypotension due to systemic vasodilation, which occurs more frequently when it is administered with other sedatives and analgesics or when it is used in patients with preexisting respiratory failure and cardiovascular instability. Since long-chain triglycerides have a caloric value of 1.1 kcal/ml, long-chain triglycerides used as a drug carrier should be included in the total calories provided to the patient at an amount corresponding to the administered drug. Long-term or high-dose use may cause hypertriglyceridemia [49]. It should be noted that when propofol is administered, propofol infusion syndrome (PRIS) can occur with an incidence of 1%, resulting in the exacerbation of metabolic acidosis, hypertriglyceridemia, hypotension, arrhythmias, acute renal failure, hyperkalemia, rhabdomyolysis, and hepatic dysfunction. In adults, PRIS is related to high-dose administration of more than 70 μg/kg/min. It can also occur with a low-dose continuous IV infusion. The mortality rate of PRIS is as high as 33%. It may not improve even if continuous dosing is stopped [50]. To prevent PRIS, it is recommended to substitute propofol with other sedatives for 24 hours every 5 days. Propofol does not mix well with other drugs, and it can be a source of infection. Thus, a dedicated catheter should be used for continuous infusion. Among the existing propofol formulations, Diprivan (AstraZeneca) contains edetic acid. If an IV infusion is continued for more than a week, a drug holiday is recommended to prevent abnormalities in trace elements. For other long-chain triglyceride propofol formulations, it is recommended that one injection period does not exceed 12 hours and that the mixed solution should be used within 6 hours after preparation.
Dexmedetomidine
A central α2-agonist, clonidine has been used to enhance the efficacy of opioid analgesics and general anesthetics, as well as to treat drug withdrawal syndrome. Dexmedetomidine is a more selective α2-agonist and has sedative, analgesic, and sympathetic suppression effects. However, it has no anticonvulsant effect. Dexmedetomidine causes less respiratory depression than other sedatives. On electroencephalography, the sleep pattern induced by dexmedetomidine is similar to that of normal physiological sleep. Dexmedetomidine can induce cooperative sedation from which it is easy to awaken patients even during drug injection. It enables communication and command obedience. The onset of action is within 15 minutes of continuous infusion. The strongest sedative effect is induced within 1 hour. Administration of a loading dose may shorten the onset of action, but may lead to hemodynamic instability in severely ill patients. Dexmedetomidine does not have a significant effect on respiratory depression. Thus, it can be used in patients who have not been intubated. It can be given through the IV route after extubation. However, it is necessary to continuously monitor the respiratory system for hypoventilation and hypoxic partial pressure because dexmedetomidine can cause airway obstruction in patients who are not intubated through loss of muscle tone in the oropharynx.
Choice of sedatives
KSCCM Recommendation
▪ Propofol is useful in patients requiring rapid and frequent awakening for neurologic evaluations or extubation. (Grade B)
▪ Midazolam use is recommended for shorter time periods because when it is used for more than 48 to 72 hours, it becomes difficult to predict the recovery time or time for extubation. (Grade A)
▪ It is recommended to use propofol rather than benzodiazepines as a sedative in post-cardiac surgery adults on mechanical ventilation. (Grade C) [Update]
▪ Propofol and dexmedetomidine are recommended as sedatives rather than benzodiazepines in mechanically ventilated medical and surgical patients not undergoing cardiac surgery. (Grade C) However, benzodiazepines are recommended for alcohol withdrawal syndrome. [Update]
▪ Systematically applied reduction or adjustment of the sedative dose to reach the daily goal of sedation level is recommended to avoid unnecessary long-term sedation. (Grade A)
▪ Serum triglyceride concentrations should be monitored starting two days after propofol administration, and the fat calories in the emulsion should be included in the total calories. (Grade B)
▪ It is recommended to make and use an appropriate sedative administration guideline and treatment flow chart or protocol for the ICU. (Grade B)
Because elective cardiac surgery patients are often hospitalized urgently, the duration of stay in the ICU and mechanical ventilation are different from those of internal medicine or other surgical intensive care patients. Therefore, this guideline divides patients requiring mechanical ventilation into cardiac surgery and medical/surgical critical patients. The 2013 PAD guidelines stated that propofol or dexmedetomidine were favored over benzodiazepine sedatives such as midazolam and lorazepam because of more favorable short-term prognostic outcomes, such as the length of ICU stay and ventilation, as well the improvement of delirium symptoms [28]. In this guideline, the short-term prognosis (time to weaning from mechanical ventilation, time to light sedation), delirium, and the long-term prognosis (90-day mortality, cognitive and physical function, and psychological dysfunction) were evaluated.
It is recommended to use propofol rather than benzodiazepines as a sedative in adults who are mechanically ventilated after cardiac surgery. In comparison to benzodiazepines, propofol can shorten the average light sedation induction time by 52 minutes and the time to extubation of the endotracheal tube by 100 minutes on average [51-53].
We compared (1) propofol versus benzodiazepines, (2) dexmedetomidine versus benzodiazepines, and (3) propofol versus dexmedetomidine in medical and surgical patients who did not undergo cardiac surgery. In comparison to benzodiazepines, propofol shortened the induction time of shallow sedation by an average of 7.2 hours and the time to extubation of the endotracheal tube by an average of 11.6 hours [54,55]. However, the delirium incidence showed no difference between the two sedatives [56]. In randomized controlled trials (RCTs) comparing benzodiazepines and dexmedetomidine, dexmedetomidine reduced the time to extubation of the endotracheal tube by an average of 1.9 days, shortened the duration of mechanical ventilation, and significantly reduced the incidence of delirium [57,58]. However, dexmedetomidine and benzodiazepines did not show any significant differences in the duration of endotracheal extubation, LOS in the ICU, or the risk of delirium in an integrated analysis. Although the dexmedetomidine group showed frequent bradycardia, no intervention was needed in most cases [58].
There were no significant differences of the time to endotracheal extubation, bradycardia, or hypotension between propofol and dexmedetomidine. However, when dexmedetomidine was used, the occurrence of delirium was reduced and patients were able to communicate more easily [58]. Since neither propofol nor dexmedetomidine is superior, one of these two drugs is recommended as a sedative for critically ill adults. However, dexmedetomidine should not be used if a deep level of sedation with or without a muscle relaxant is needed. Figure 3 shows a pharmacologic treatment flowchart of agitation in mechanically ventilated patients. Benzodiazepines are recommended for alcohol withdrawal syndrome [59]. However, since the role of nonpharmacologic treatment for the reduction of anxiety, agitation, and psychologic distress is uncertain, sedatives are not recommended for alcohol withdrawal syndrome.
Discontinuation of sedatives
KSCCM Recommendation
▪ Benzodiazepine and propofol withdrawal symptoms are more likely to occur during high-dose treatment and if the patient has been infused for more than a week. These drugs should be systematically reduced to prevent withdrawal symptoms. (Grade B)
Patients taking analgesics or sedatives for more than a week may develop neurological changes or physiological dependence. Withdrawal symptoms may occur if analgesics or sedatives are abruptly discontinued. Benzodiazepine withdrawal symptoms and signs are myalgia, tremor, headache, nausea, diaphoresis, fatigue, anxiety, excitation, perceptual dysfunction, elevated sensitivity to light and sound, muscle spasms, myoclonus, sleep disturbances, delirium, and convulsions. Propofol withdrawal symptoms are similar to benzodiazepine withdrawal symptoms. High-risk patients for the development of withdrawal symptoms are those who have been admitted to the ICU for more than 7 days or have received lorazepam at doses higher than 35 mg/day [60,61]. In case of intermittent administration, switching to a long-acting drug is helpful in preventing withdrawal symptoms. After lowering the sustained infusion rate by 20%–40% for the first time, an additional 10% reduction every 12–24 hours depending on the patient's response is also allowed to help reduce symptoms [61].
Physical restraints
KSCCM Recommendation
▪ Although physical restraints have various advantages, such as protecting staff from combative patients and preventing self-extubation, self-removal of medical devices, and falling accidents, a careful consideration of the advantages and disadvantages of physical restraints is recommended before initiation/maintenance of physical restraints. (Grade C) [Update]
Medical staff can consider using physical restraints to improve patient safety, protect staff from combative patients, prevent self-extubation, prevent self-removal of medical devices, prevent falling accidents, regulate patient behavior, and maintain patient posture/position [62,63]. Restraints usually fix the ankles, wrists, and upper body in place. However, in a few studies, the use of physical restraints was associated with a prolonged ICU stay, increased agitation, increased demand for opioids and sedatives, and risk of delirium or disorientation [64,65]. Risk factors that increase the use of physical restraints in critically ill adults include old age; non-comatose consciousness level; neurologic and psychologic status including delirium; sedative type, administration method, and dosing; mechanical ventilation; use of invasive devices; the nurse-to-patient ratio and perceived workload; and specific times of the day. Some patients who received physical restraints during their ICU stay have shown strong affective reactions even after transfer from the ICU to general ward [32]. Given the frequency of use of physical restraints, unintended consequences, and patient perceptions, health care providers should carefully consider the risks and effects before initiating/maintaining the use of restraints in an adult ICU.
Delirium
Delirium is defined as acute cerebral dysfunction accompanied by a change of level of consciousness, disorientation, and cognitive dysfunction during a short duration (hours to days). Delirium is classified into three subtypes: hyperactive (agitated), hypoactive (calm or lethargic), and mixed (fluctuation between the two subtypes). Among these three types, the mixed subtype occurs the most, and the hypoactive subtype has a worse prognosis. Approximately 20% to 80% of critically ill adult patients have experienced delirium during their ICU stay. Delirium is associated with increased ICU mortality, longer LOS in hospital, higher healthcare costs, and more long-term cognitive dysfunction, such as dementia. Thus, early recognition and treatment for delirium are essential [66,67]. Patients in whom delirium is detected early, followed by prompt treatment, show the same prognosis as patients who do not develop delirium.
Risk factors
Delirium occurrence, duration, and severity in adult ICU patients are strongly related to various risk factors (Table 7) [68]. Modifiable factors risk include benzodiazepine use and blood transfusion. Sex, opioid sedatives, mechanical ventilation, past history of pulmonary disease, hospital admission, nicotine use, dialysis, or continuous venovenous hemofiltration, and a lower Glasgow Coma Scale have been identified as having no relationship with an increased incidence of delirium.
The PREdiction of DELIRium in ICu patients (PRE-DELIRIC) model and early (E)-PRE-DELIRIC model can predict delirium in critically ill adult patients. The PRE-DELIRIC model consists of 10 predictors (age, Acute Physiology and Chronic Health Evaluation [APACHE] II score, admission group, emergent admission, infection, coma, sedation, morphine use, urea level, and metabolic acidosis). It can predict ICU delirium within 24 hours after ICU admission [69,70]. The E-PRE-DELIRIC model includes nine predictors (age, history of cognitive dysfunction, history of alcohol abuse, blood urea nitrogen, admission class, emergent admission, mean arterial blood pressure, use of steroids, and respiratory failure). It can predict ICU delirium at ICU admission [71].
Assessment and outcomes
KSCCM Recommendation
▪ Delirium in critically ill ICU patients should be monitored using the Confusion Assessment Method for the ICU (CAM-ICU) and intensive care delirium screening checklist (ICDSC) as valid tools. (Grade A)
The early diagnosis of delirium can lead to rapid identification and correction of the cause or causes, relief of symptoms, pharmacologic or nonpharmacologic therapeutic interventions, and evaluation of treatment effectiveness. Thus, adult ICU patients should be routinely evaluated for delirium using a valid tool. The CAM-ICU and ICDSC are recommended as the most verified and reliable tools for delirium assessment in critically ill adult patients (Table 8) [72-74]. The level of consciousness of patients is checked using the RASS or SAS. Patients with a RASS score >–4 or SAS score >2 are eligible for delirium evaluation using the CAM-ICU (Figure 4). Ideally, clinicians should perform the CAM-ICU at least twice a day (day and night) to assess delirium. The CAM-ICU can even be used for delirium assessment in patients with speech impairment, endotracheal intubation, dementia, and severe depressive disorder. When evaluating delirium using the ICDSD, eight items derived from the Diagnostic and Statistical Manual of Mental Disorders criteria are identified over a 24-hour period. Four or more points on the ICDSC corresponds to a diagnosis of delirium.

Delirium assessment tool: the Confusion Assessment Method for the intensive care unit (CAM-ICU) flowchart. RASS: Richmond Agitation-Sedation Scale.
A patient’s level of arousal may affect the assessment of delirium using verified screening tools. The diagnosis rate of delirium is significantly higher when patients have a RASS score of –2. However, the effect of the level of arousal from sedatives on the evaluation of delirium needs further studies. Delirium is related to poor outcomes, even when accompanied by decreased arousal levels. Thus, clinicians should not underestimate the possibility and importance of delirium even in patients with decreased arousal levels. The occurrence of delirium in critically ill patients is closely related to cognitive dysfunction at 3 and 12 months after discharge from the ICU, and may be related to a prolonged hospital stay. It has been consistently shown that delirium in critically ill adult patients is not related to post-traumatic stress disorder or post-ICU distress. However, the outcomes of patients with rapidly reversible delirium are similar to those of patients who never experience delirium [75].
Pharmacologic prevention and treatment
KSCCM Recommendation
▪ Pharmacologic treatment for preventing delirium in critically ill patients is not recommended. (Grade B) [Update]
▪ Routine use of haloperidol, an atypical antipsychotic, a statin, or dexmedetomidine is not recommended for delirium treatment. However, treatment of delirium using these drugs can be considered if the symptoms of delirium may be harmful to patients. (Grade B) [Update]
Currently, the pharmacologic agents for delirium treatment include a typical antipsychotic (haloperidol), atypical antipsychotics (risperidone, olanzapine, quetiapine), and dexmedetomidine (Table 9).
Pharmacologic treatment
Routine use of haloperidol, atypical antipsychotics, 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors (statins), or dexmedetomidine is not recommended for delirium treatment. In six RCTs, these drugs were not found to reduce delirium duration, shorten the ICU LOS, or diminish mortality [76-81]. However, short-duration use of haloperidol or an atypical antipsychotic may be helpful for patients who experience meaningful distress secondary to delirium symptoms, including agitation, panic, hallucination, and delusion, or who may be physically harmful to themselves or others due to anxiety. Atypical antipsychotics are preferred as they have a lower risk of extrapyramidal symptoms than haloperidol. However, atypical antipsychotics are not recommended for use in patients at risk of torsades de pointes (e.g., those with a prolonged QT interval on electrocardiography, administration of drugs that prolong the QT interval, or a history of arrhythmias). Dexmedetomidine use may be considered in patients with delirium on mechanical ventilation in whom agitation delays withdrawal/extubation of mechanical ventilation . Although benzodiazepines can induce delirium, they can be used in patients with secondary delirium related to alcohol or benzodiazepine withdrawal. The use of haloperidol or an atypical antipsychotic in critically ill patients with subsyndromal delirium is not associated with a reduced incidence of delirium or an improved prognosis [82].
Pharmacologic prevention
The use of haloperidol, atypical antipsychotics, statins, dexmedetomidine, and ketamine for delirium treatment is not recommended to prevent delirium in any adult ICU patients. Although a few studies reported that these drugs significantly reduced delirium incidence, their use is not associated with a reduced duration of mechanical ventilation, ICU LOS, or mortality. However, they can increase the risk of side effects. Thus, a nonpharmacologic prevention strategy is needed to decrease the incidence of delirium [83,84].
Nonpharmacologic prevention and treatment
KSCCM Recommendation
▪ We recommend a multicomponent (ABCDEF bundle) nonpharmacologic intervention strategy for the prevention and treatment of delirium. (Grade B) [Update]
A multicomponent, nonpharmacologic intervention that is focused on reducing modifiable risk factors for delirium, enhancing cognitive capability, and optimizing sleep, rehabilitation/mobilization, hearing, and vision in critically ill patients is significantly associated with a lower incidence of delirium, a reduced duration of delirium, a decreased duration of mechanical ventilation, a shortened ICU LOS, and a lower risk of mortality [85-89]. Thus, clinicians should perform multicomponent interventions to decrease or shortened the incidence and duration of delirium by performing reorientation, stimulating cognitive capability, using clocks, improving sleep quality by minimizing light and noise, minimizing sedation, reducing immobility through early rehabilitation and exercise, and decreasing hearing and/or visual impairment by using available devices including hearing aids or eyeglasses.
The bundle of awakening and breathing coordination, delirium monitoring/management, and early exercise/mobilization (ABCDE) meaningfully reduces the incidence of delirium. The increase in compliance with the ABCDEF bundle, which includes family participation (“F”), is significantly related to decreased mortality and prolongation of the period without coma or delirium in the ICU [90]. No studies have reported adverse effects from the application of multicomponent, nonpharmacologic intervention strategies.
Immobility (Rehabilitation/Mobilization)
KSCCM Recommendation
▪ As a vital component of critical care, the rehabilitation of critically ill patients is related to long-term outcomes. (Grade C) [Update]
In the past, the treatment of critically ill patients was focused on treating underlying internal/surgical problems by sedating patients and having them rest in bed. However, survivors of intensive care treatment suffer from complications such as ICU-acquired muscle weakness, which leads to problems such as a decreased long-term survival rate, reduced physical function, and diminished quality of life [91-93]. Since the 2000s, active rehabilitation treatment and walking have been attempted in the ICU. Evidence for their safety and effectiveness has been reported. The 2013 PAD guidelines recommended that “rehabilitation/mobilization be useful as part of a delirium treatment strategy [28].” In the 2018 PADIS guidelines, the rehabilitation/mobilization section was separately selected as a single topic and active rehabilitation treatment was recommended [94].
Efficacy and benefits of rehabilitation
KSCCM Recommendation
▪ Rehabilitation in critically ill patients is related to a shortened duration of delirium, mechanical ventilation, and ICU LOS. (Grade C) [Update]
▪ Although rehabilitation interventions have no effect on mortality, continuous rehabilitation for critically ill patients can improve long-term survival. (Grade B)
Several RCTs have reported that intensive care rehabilitation treatment can strengthen limb and respiratory muscle strength, improve physical function and quality of life, shorten the duration of delirium and mechanical ventilation, and shorten the ICU LOS.
In a controlled study in which 60 patients were randomly assigned to a rehabilitation group and a control group, the duration of ventilation and the LOS in the ICU were shortened by 1.7 days and 2.5 days, respectively, in the rehabilitation group [95]. In a randomized controlled study of 90 patients, the rehabilitation group showed better results in the 6-minute walking distance test, quadriceps muscle strength test, and quality of life analysis (Physical Functioning Scale of the Short Form 36-item questionnaire) upon discharge from the hospital [96]. A meta-analysis of 43 RCTs also reported that rehabilitation decreased the duration of ventilation by 1.7 days and the ICU LOS by 1.2 days. However, it did not reduce the mortality rate. Many studies have similarly reported that intensive care rehabilitation does not reduce mortality [97].
The reason why rehabilitation treatment alone does not significantly affect mortality might be that many factors affect mortality. However, it has been reported that the proportion of 10-year survival was improved in patients who received rehabilitation treatment from the ICU even after discharge [98]. Based on this, it can be said that continuous rehabilitation treatment for critically ill patients can help improve their long-term survival rate.
Safety and risks of rehabilitation
KSCCM Recommendation
▪ Rehabilitation is an intervention that can be safely applied in the ICU. (Grade B) [Update]
▪ A rehabilitation program should consist of a rehabilitation protocol, safety and effectiveness index, and indications for stopping via a multidisciplinary approach according to the environment of each ICU. (Grade B) [Update]
Although the usefulness of rehabilitation for critically ill patients is recognized, the biggest stumbling block to its practical application is medical staff's anxiety about patient safety. Critically ill patients have a variety of instruments such as a ventilator, central venous line, and arterial line, making it challenging to apply rehabilitation easily due to the risk of disconnection or accidental instrument removal and anxiety that a patient's condition may worsen during rehabilitation associated with hemodynamic instability in critically ill patients.
However, many studies have reported that serious harm does not occur in intensive care rehabilitation. It has been reported that rehabilitation can be safely applied to those who are on ventilators and patients receiving continuous renal replacement therapy or extracorporeal membrane oxygenation [99,100].
In a prospective observational study with 1,100 patients and a total of 5,267 rehabilitation sessions, 34 (0.6%) adverse reactions occurred. The most common adverse reactions were arrhythmias (10 cases, 0.2%) and an increase in mean arterial pressure to 140 mm Hg or more (8 cases, 0.2%), which resulted in an increased LOS in the ICU. However, no serious adverse reactions were reported [101]. In a meta-analysis of 48 studies, only 78 cases (0.6%) out of a total of 14,398 rehabilitation sessions had adverse reactions. Very few adverse reactions may cause harm, such as falls and endotracheal tube dislocation [102].
It is important to set criteria for the implementation and discontinuation of rehabilitation treatment appropriate for the characteristics of each ICU and then implement the rehabilitation treatment accordingly (Table 10) [103]. Collaboration of various medical staff is essential for achieving this goal. It is important that the ICU doctor, nurse, rehabilitation medicine doctor, and physical therapist form a team to develop a rehabilitation program tailored to the characteristics of each ICU. It is also necessary to develop a rehabilitation treatment program suitable for each hospital's situation by discussing the patients who will receive intensive care rehabilitation, evaluation indicators for safety and effectiveness, the rehabilitation protocol, and criteria for stopping treatment.
Sleep Disturbance
Causes
Sleep disturbance is one of the most common complaints of critically ill patients. Sleep disturbance in the ICU includes sleep segmentation, increased light sleep (N1+N2 stage), and decreased rapid eye movement (REM) sleep [104]. Sleep disturbance can cause delirium, prolong mechanical ventilation, and decrease immune function. Since sleep disturbance is a controllable risk factor for the development of delirium, the incidence of delirium can be lowered by correcting factors that may cause sleep disturbance.
Potential causes of sleep disturbance include the environment of the ICU, such as continuously bright lights and alarm sounds, the patient treatment process, pain, and ventilation. Sleep disturbance can also be caused by systemic inflammatory conditions and drugs used [105]. The mode of the ventilator also plays an important role. According to the 2018 PADIS guidelines, using the assist-control mode at night may help improve sleep quality in comparison to using the pressure-support mode [94,106].
Treatment
KSCCM Recommendation
▪ Sleep disturbance is a common risk factor for delirium in the ICU. Appropriate control of sleep disturbance can prevent delirium. (Grade B) [Update]
▪ A sleep-promoting protocol (offering earplugs and eyeshades, avoiding unnecessary examinations) should be used to improve the sleep of critically ill patients. (Grade A) [Update]
Nonpharmacologic therapy
The main factors that interfere with sleep in the ICU are often aspects of the environment, such as bright lighting, machine alarms, and ventilator alarms. Therefore, minimizing unnecessary nursing treatment or examinations at night, using earplugs and eyeshades, and similar steps can produce a significant effect on sleep disturbance at a low cost. One RCT comparing patients who used earplugs at night (n=69) with a control group that did not (n=67) reported that the use of earplugs improved the sleep quality and lowered the risk of delirium [107]. In addition, in a study that performed quality improvement activities by bundling various environmental interventions such as the use of earplugs and sleeping masks, reduction of nursing activities, and avoidance of unnecessary blood draws and examinations, the incidence of delirium was reduced (33% vs. 14%). The duration of delirium was also reduced (3.4 days vs. 1.2 days) [108]. As such, nonpharmaceutical treatment alone can improve sleep disorders, and appropriate nonpharmacologic strategies play a significant role in the treatment of critically ill patients (Table 11).
Pharmacologic treatment
Various studies have been conducted on sleep-inducing drugs (melatonin, dexmedetomidine, and propofol) in treating critically ill patients. However, no drugs have shown effects. As a sedative, dexmedetomidine, which has recently been used extensively in ICUs, has been used in small studies to induce sleep only at night. It has been reported that dexmedetomidine can increase stage 2 sleep and preserve the day-night sleep cycle [109,110]. However, an RCT of 100 subjects reported that low-dose dexmedetomidine had no significant effects on sleep [84]. Considering the high cost of this drug and its hemodynamic side effects, it is not recommended for inducing sleep. Since there are few large-scale studies on drugs administered at night to promote sleep in adult ICU patients, further studies should be conducted.
DISCUSSION
Compared to the 2010 guidelines, we aimed to provide recent updates on the information that clinicians need to advance treatment for critically ill ICU patients. The 2021 KSCCM clinical guideline was developed based on the 2018 PADIS guideline, which was an update of the 2013 PAD guideline with the addition of rehabilitation/mobility and sleep. Routine monitoring and assessment for pain, sedation, and delirium using the most valid and reliable tools can improve outcomes associated with nonpharmacologic and pharmacologic therapeutic interventions. After initial nonpharmacologic therapy, the optimal medication choice and dose can be considered for pharmacologic treatment to treat PAD. A multi-modal analgesia strategy is needed to reduce opioid use and increase pain-modulating effects, thereby improving pain control and patient-centered outcomes. The strategy of enhancing patient comfort while maintaining a light level of sedation improves clinical outcomes. The 2021 KSCCM guideline particularly emphasizes active rehabilitation and sleep as factors influencing the recovery of critically ill ICU patients. In summary, a multidisciplinary strategy, including administering adequate pain treatment, maintaining a light level of sedation, performing routine monitoring and treatment for delirium, providing active rehabilitation in the ICU, and offering treatment for sleep disturbance, is needed to improve the clinical outcomes of critically ill ICU patients. Although these guidelines cannot offer definitive answers on all topics that are important to critical care clinicians, we believe that they can serve as a cornerstone for a comprehensive discussion on clinical issues relevant to patient management.
HIGHLIGHTS
• A multidisciplinary strategy, including optimizing pain management, maintaining light sedation, performing routine monitoring and treatment for delirium, providing active rehabilitation in the intensive care unit (ICU), and treating sleep disturbance using a sleep-promoting protocol, needs to be implemented to improve the clinical outcomes of critically ill ICU patients.
• The 2021 clinical practice guideline provides up-to-date information on how to prevent and manage pain, agitation/sedation, delirium, immobility, and sleep disturbance (PADIS) in critically ill ICU patients.
• It also provides information about pharmacologic and nonpharmacologic therapeutic interventions to prevent and manage PADIS.
Notes
CONFLICT OF INTEREST No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS
Conceptualization: TSH. Data curation: HJL, EJH. Methodology: HJL, EJH. Writing–original draft: YS. Writing–review & editing: TSH.












