Abstract
-
Background
- Critically ill patients experience muscle weakness, which leads to functional disability. Both functional electrical stimulation (FES) and in-bed cycling can be an alternative measure for intensive care unit (ICU) patients who are not feasible for active exercise. The aim of this study was to examine whether FES and in-bed cycling have a positive effect on muscle mass in ICU patients.
-
Methods
- Critically ill patients who received mechanical ventilation for at least 24 hours were included. After passive range of motion exercise, in-bed cycling was applied for 20 minutes, and FES was applied for 20 minutes on the left leg. The right leg received in-bed cycling and the left leg received both FES and in-bed cycling. Thigh circumferences and rectus femoris cross-sectional area (CSA) were assessed with ultrasonography before and after the intervention. Muscle strength was assessed by Medical Research Council scale.
-
Results
- A total of 10 patients were enrolled in this study as a pilot study. Before and after the intervention, the CSA of right rectus femoris increased from 5.08 ± 1.51 cm2 to 6.01 ± 2.21 cm2 , which was statistically significant (P = 0.003). The thigh circumference was also increased and statistically significant (P = 0.006). There was no difference between left and right in regard to FES application. There is no significant change in muscle strength before and after the intervention (right and left, P = 0.317 and P = 0.368, respectively).
-
Conclusions
- In-bed cycling increased thigh circumferences rectus femoris CSA. Adding FES did not show differences.
-
Keywords: electric stimulation therapy; muscle weakness; rehabilitation; ultrasonography
INTRODUCTION
Intensive care unit-acquired weakness (ICUAW) is a common finding in critically ill patients [1]. The reported incidence of ICUAW varies from one fourth to three fourths in different ICU populations [2]. The major risk factors of ICUAW include inflammation, hyperglycemia, long duration of immobility, poor nutritional support, mechanical ventilation, neuromuscular blockade, and sepsis [3].
Neuropathy and loss of muscle mass are considered to be the causes of ICUAW. ICUAW is known to be associated with poor short- and long-term outcomes in critically ill patients. It is related with delayed mechanical ventilation weaning, which leads to prolonged ICU and hospital stay. Also, functional disability from muscle weakness leads to worse long-term outcomes such as quality of life, post-intensive care syndrome and survival [4,5].
Recently, early physical rehabilitation in ICU has been advocated as an effective measure to facilitate early recovery and to prevent functional disability after critical illness [6,7]. There are many technologies suggested for rehabilitation of the critically ill patients [8]. Among the technologies used for the critically ill population, there is a rising interest in functional electrical stimulation (FES) and in-bed cycling because both techniques do not require voluntary movements of the patients [9-12]. FES and early physical rehabilitation are known to prevent ICUAW and to preserve muscle mass, but there is no study that investigates their effects on muscle strength and mass. Therefore, the aim of this study was to investigate if FES and in-bed cycling have positive effects on muscle mass in mechanically ventilated patients. We also compared the effects of cycling alone and cycling with FES.
MATERIALS AND METHODS
Data collection
This study was conducted at Severance Hospital, Seoul, Republic of Korea. This study protocol was approved by the Institutional Review Board (IRB No. 2016-2447-004). Patients aged 20 years or older who were admitted to the ICU and those who required mechanical ventilation for at least 24 hours were enrolled in the study after getting informed consent. The experiment day was the first day of rehabilitation treatment after weaning from mechanical ventilation. We screened patients who had no previous history of rehabilitation prior to registration. Patients were excluded if they were hemodynamically unstable, unable to cooperate, unable to obey to verbal command, and had wound or injury on the legs.
Study protocol
After the patients’ consent, both leg thigh circumferences were measured with a ruler at 50% and 25% of the femur length before and after the intervention (Figure 1). Rectus femoris muscle cross-sectional area (CSA) was measured with an ultrasound machine (M-Turbo; SonoSite, Bothell, WA, USA). A 6–13 MHz linear transducer was placed perpendicular to the long axis of the thigh, at 50% and 25% of the thigh length between the anterior superior iliac spine and the superior margin of the patella (Figure 2). Muscle strength of both legs was measured with Medical Research Council (MRC) scale before and after the intervention. MRC scale ranges from 0 (no visible contraction) to 5 (active movement against full resistance). During the whole rehabilitation session including in-bed cycling exercise and FES, the patients’ vital signs were monitored.
In-bed cycling
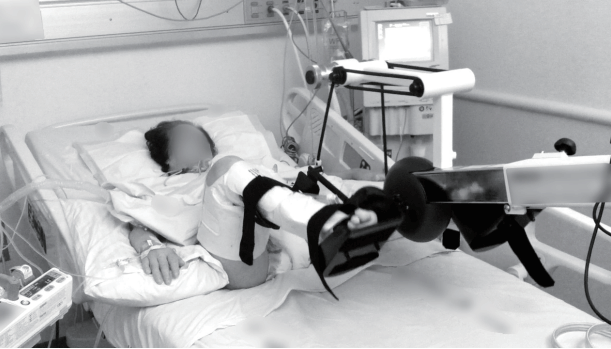
A 20-minute in-bed cycling of the lower limbs using a bedside cycle ergometer (Letto 2; MOTOmed, Betzenweiler, Germany) was implemented (Figure 3). All inbed cycling sessions were conducted by an experienced physiotherapist and the ICU nurse team. The in-bed cycling started with a passive mode at the default speed of the cycle ergometer (20 cycle/min), and then the resistance applied by the cycle ergometer was adjusted until the patient indicated fatigue. Each patient will receive a preprogrammed standardized treatment template. Each session will start with a 1-minute motor-driven passive cycling warm-up at a rate of 20 revolutions per minute (RPM). Patients will continue with passive, active-assisted or active cycling for the next 19 minutes, according to their level of participation. The device offers the possibility to conduct passive or active cycling at six levels of increasing resistance. The session will finish with a 30-second motor-driven cool-down (30:30 total). Since ICU patients’ level of physical strength may vary throughout their exercise, we will allow patients to cycle at a self-selected RPM and will not change the resistance. Resistance was kept stable or downgraded if there was over 10% change in heart rate or blood pressure and desaturation less than 95%.
Functional electrical stimulation
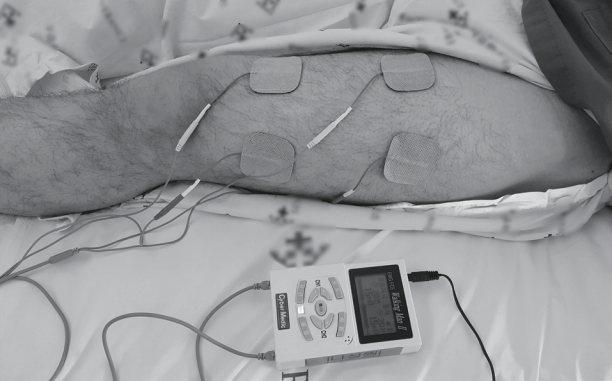
After in-bed cycling, there was a 10-minute rest period before FES was applied for 20 minutes on the left thigh (Figure 4). Four electrode pads were placed along with the lateral border of quadriceps muscle. Biphasic symmetrical square wave pulse was delivered by a stimulator (Walking Man II; Cyber Medic, Iksan, Korea). During a total 20-minute treatment session, electrical stimulation was given at 35 Hz, a duty cycle of 10 seconds on and 12 seconds off, and a pulse time of 250 seconds. The threshold current was defined as the lowest stimulation current with visual muscle contraction.
Data analysis
Statistical analysis was performed using IBM SPSS version 23.0 (IBM Corp., Armonk, NY, USA). The differences of thigh circumference and rectus femoris CSA were analyzed with paired t-test. Muscle strength was compared with McNemar-Bowden test. Null hypotheses of no difference were rejected if P-values were less than 0.05.
RESULTS
This study was discontinued after 10 patients were enrolled; the primary analysis showed statistically significant differences. General characteristics of the study patients are summarized in Table 1.
Rectus femoris CSA were significantly increased after in-bed cycling and FES (CSA right side 50% cycling only, 5.08 ± 1.51 cm2 vs. 6.01 ± 2.21 cm2, P = 0.003; left side 50% cycling and FES, 5.28 ± 1.89 cm2 vs. 6.59 ± 2.23 cm2, P = 0.008) (Table 2).
The thigh circumference was also increased and statistically significant (preintervention vs. postintervention, 47.43 ± 5.79 cm vs. 48.24 ± 5.56 cm; P = 0.006) (Table 2). There is no significant change in muscle strength before and after in-bed cycling and FES (McNemar test; right and left, P = 0.317 and P = 0.368, respectively) (Table 2). The increment of the left thigh circumference and rectus femoris CSA were not different from that of the right leg, which meant that FES may not show additive effect on muscle mass comparing to cycling only (Table 3).
DISCUSSION
This study showed that rehabilitation course composed of in-bed cycling in ICU patients leads to acute increase of lower limb muscle mass measured by ultrasound. We expected synergies when additional FES was applied; there was no significant difference according to FES application. Only in-bed cycling was applied on the right leg and cycling followed by FES on the left leg, which revealed that there was no additive effect of FES to cycling compared to cycling alone.
The onset of ICUAW is known to be unexpectedly fast. It is reported that muscle fiber atrophy and weakness starts just after 4 hours of bed rest, and 10% of muscle atrophy is expected with 1 week of immobilization [13].
ICUAW is also associated with short- and long-term outcomes in critically ill patients. From the short-term outcome standpoint, a few studies have demonstrated that weakness is an independent predictor of prolonged mechanical ventilation. There was an observational study that evaluated ICUAW in septic patients receiving ventilation for more than a week [14]. Patients without electro-physiologic evidence of ICUAW had a shorter duration of mechanical ventilation than patients with ICUAW. Also, the patients with ICUAW were associated with an increased length of ICU and hospital stay. ICUAW is related to acute mortality in critically ill patients. Leijten et al. [15] performed a cohort study of 50 patients receiving ventilation for at least 7 days, and demonstrated that patients diagnosed with ICUAW electrophysiologically showed higher ICU mortality (48% vs. 19%, P = 0.03).
To minimize the adverse effects of ICUAW due to immobility, early physical activity and rehabilitation have been advocated among many intensivists. Griffiths et al. [16] performed an interesting study. They enrolled five critically ill patients who required muscle relaxants for at least 7 days due to ventilation. They applied passive exercise to one leg for 3 hours daily and the other leg received only routine care. Passive exercise resulted in muscle mass increase of 11%, while routine care without passive exercise resulted in a muscle mass decrease of 35%. Schweickert et al. [6] investigated the effects of early physical and occupational therapy in mechanically ventilated patients. They randomized over 100 ventilated patients into control versus early physical and occupational therapy groups. The authors found that patients who received physical and occupational therapy had shorter duration of delirium and more ventilator-free days during the 28-day follow-up period than the controls. In addition, functional independency was achieved more in the intervention group (59%) compared with patients in the control group (35%).
There are many technologies and protocols utilized to facilitate early mobilization in ICU. Needham et al. [8] reviewed technologies to enhance ICU rehabilitation, and the authors suggested neuromuscular electrical stimulation, cycle ergometry, and a few devices to facilitate ambulation of mechanically ventilated patients. They adopted the MOVER Aid to assist with mobilizing a mechanically ventilated patient: a wheeled walker with a safety seat and an equipment tower mounting the monitor, intravenous pump, portable ventilator, and oxygen tanks. Despite the benefits of the MOVER Aid, this walker may not be suitable for understaffed ICUs. With the MOVER Aid, a minimum of two staff members, including a respiratory therapist and physical therapist, are required to ambulate a mechanical ventilated patient.
Compared to ICU in developed countries, Korean intensive care unit has not enough human resource for rehabilitation such as nursing, respiratory, physical therapist, occupational therapist. Compared with ambulation, in-bed cycling is very easy to apply to the critically ill patients, and the expected adverse events are few and rare. For ICU patients, neuromuscular electrical stimulation has been recently reported to preserve muscle mass [10] and prevent ICUAW [17]. However, data about the efficacy and synergy effect when applying FES and ergometry together are limited. Therefore, we designed a study of rehabilitation using cycling and FES feasible for low staffed ICU.
Recently, Patsaki et al. [18] studied the effects of neuromuscular stimulation (NMES) and individualized rehabilitation on muscle strength in ICU survivors. He applied NMES combined with personalized physiotherapy 7 days a week for 55 minutes on rectus femoris and peroneus longus of both lower extremities. In an analysis of muscle strength and functional status at hospital discharge, NMES and personalized physiotherapy in ICU survivors did not show significant differences compared with controlled group. It is in agreement with our conclusion that it is difficult to induce muscle strength improvement with NMES.
There are several limitations in this study. The major limitation of this study is that we investigated temporary change of muscle mass in this study. There was a cohort study of septic patients who received daily 1-hour course of FES for 7 days, and the authors reported no changes in muscle mass was noticed with electrical stimulations [9]. When considering why FES does not affect muscle mass, it is important to keep in mind that the number of times FES is applied or the degree of electrical stimulation may not be sufficient to change the amount of muscle. This study measured muscle CSA directly after the exercise and FES, therefore, temporary increase of perfusion and muscle mass was reflected in ultrasound measurement. Further long-term follow-up considering the effect of inbed cycling and FES are needed.
Another weak point of this study is that we enrolled very heterogeneous and a relatively small number of the patients. Although the initial study design intended to recruit up to 30 patients, interim analysis revealed statistical significance with enrollment of 10 patients. In addition, many studies we referred to in this study included a relatively small number of patients up to 20 patients, identifying statistical differences [13,16]. In addition, although we chose MRC score because manual muscle testing is reliable for longitudinal and comprehensive assessment of strength for trained research staff [19], subjective aspect of the MRC score could be an uncontrolled limited factor. Further research is needed using objective muscle strength measurement tools such as the handgrip dynamometer to determine the muscle strength of the critically ill patients.
In conclusion, this study confirmed that short-term inbed cycling has an effect of increasing muscle mass, but it did not reveal synergistic effects of FES use with inbed cycling. Long-term effect of cycling and FES needs further investigation.
NOTES
-
No potential conflict of interest relevant to this article was reported.
Acknowledgments
This study was supported by a faculty research grant of Yonsei University College of Medicine (6-2011-0203).
Figure 1.Thigh circumference and rectus femoris measurement level.
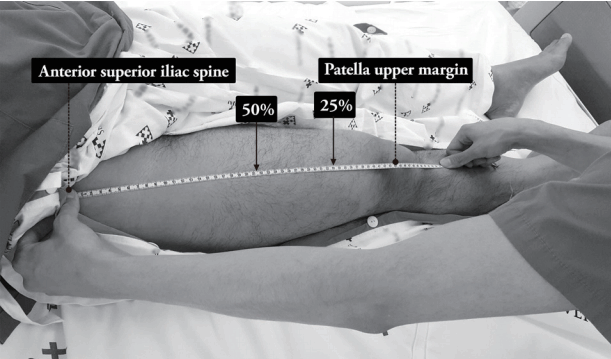
Figure 2.Rectus femoris muscle cross-sectional area. A 6–13 MHz linear transducer was placed perpendicular to the long axis of the thigh, at 50% and 25% of the thigh length between the anterior superior iliac spine and the superior margin of the patella. A: cross-sectional area and circumference, B: muscle depth.
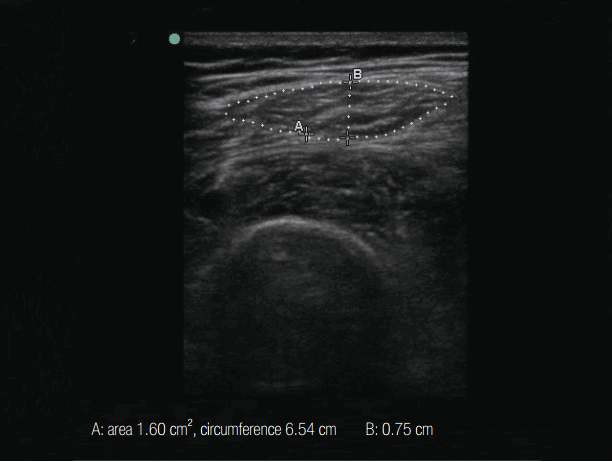
Figure 3.In-bed cycling. A bedside cycle ergometer (Letto 2; MOTOmed, Betzenweiler, Germany) was applied for 20 minutes.
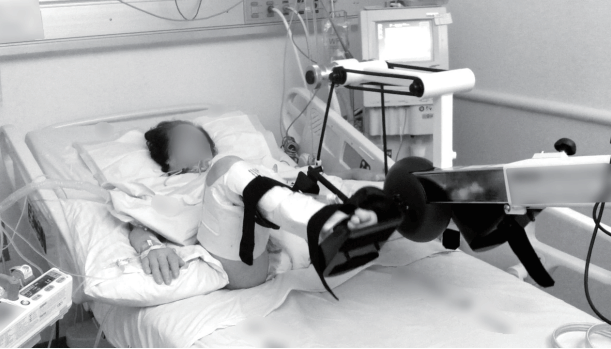
Figure 4.Functional electrical stimulation. For 20 minutes, biphasic symmetrical square wave pulse was delivered by a stimulator (Walking Man II; Cyber Medic, Iksan, Korea).
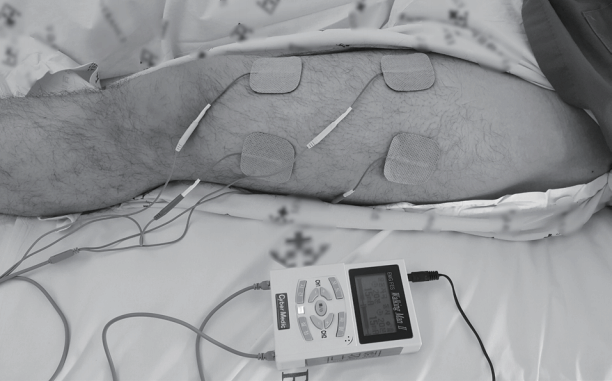
Table 1.Demographics
|
Case |
Age (yr)/sex |
BMI (kg/m2) |
Diagnosis |
Cause of MV |
Duration of MV (hr) |
ICUD (day) |
APACHE II score |
SOFA score |
|
#1 |
68/Male |
21.3 |
Idiopathic pulmonary fibrosis |
Postoperative respiratory care |
166 |
6 |
26 |
5 |
|
#2 |
79/Male |
18.3 |
Spinal stenosis |
Aspiration pneumonia |
214 |
12 |
21 |
2 |
|
#3 |
64/Male |
23.9 |
Renal stone |
Fluid overload |
131 |
6 |
23 |
2 |
|
#4 |
48/Male |
21.8 |
Idiopathic pulmonary fibrosis |
ARDS |
544 |
26 |
9 |
4 |
|
#5 |
72/Male |
23.8 |
Upper gastrointestinal bleeding |
Hypovolemic shock |
117 |
4 |
30 |
7 |
|
#6 |
53/Male |
23.8 |
Tonsillar cancer |
Postoperative respiratory care |
32 |
1 |
19 |
0 |
|
#7 |
56/Female |
21.2 |
Oral cavity cancer |
Postoperative respiratory care |
45 |
2 |
22 |
2 |
|
#8 |
63/Male |
25.8 |
Hepatocellular carcinoma |
Aspiration pneumonia |
392 |
64 |
46 |
4 |
|
#9 |
31/Female |
28.6 |
Postpartum bleeding |
Fluid overload |
28 |
3 |
24 |
2 |
|
#10 |
82/Female |
27.3 |
Renal cell cancer rupture |
Pulmonary edema |
383 |
18 |
32 |
2 |
Table 2.Pre- and postintervention results for patient completing in-bed cycling and FES
|
Variable |
|
|
Preintervention |
Postintervention |
P-value |
|
Circumference of rectus femoris (cm) |
Right |
25% |
40.19 ± 3.78 |
40.88 ± 3.86 |
<0.001**
|
|
|
50% |
47.43 ± 5.79 |
48.24 ± 5.56 |
0.006**
|
|
Left |
25% |
40.21 ± 3.29 |
40.76 ± 3.12 |
<0.001**
|
|
|
50% |
47.83 ± 5.11 |
48.75 ± 4.73 |
0.027*
|
|
Cross-sectional area of rectus femoris (cm2) |
Right |
25% |
2.10 ± 0.95 |
2.72 ± 1.24 |
0.002**
|
|
|
50% |
5.28 ± 1.89 |
6.59 ± 2.23 |
0.003**
|
|
Left |
25% |
2.19 ± 1.14 |
2.99 ± 1.22 |
0.002**
|
|
|
50% |
5.28 ± 1.89 |
6.59 ± 2.23 |
0.008**
|
|
MRC scale |
Right |
|
4 (3.75–4.25) |
4 (4–4.25) |
0.317 |
|
Left |
|
4 (3.75–4.25) |
4 (4–4) |
0.368 |
Table 3.Comparison of effects of FES addition
|
Variable |
Measurement level (%) |
Right in-bed cycling only (% increase) |
Left in-bed cycling + FES (% increase) |
P-value |
|
Circumference (cm) |
25 |
1.72 |
1.41 |
0.451 |
|
50 |
1.78 |
2.04 |
0.417 |
|
Cross-sectional area (cm2) |
25 |
30.34 |
46.32 |
0.462 |
|
50 |
15.96 |
27.44 |
0.626 |
References
- 1. Schweickert WD, Hall J. ICU-acquired weakness. Chest 2007;131:1541-9.ArticlePubMed
- 2. De Jonghe B, Sharshar T, Lefaucheur JP, Authier FJ, Durand-Zaleski I, Boussarsar M, et al. Paresis acquired in the intensive care unit: a prospective multicenter study. JAMA 2002;288:2859-67.ArticlePubMed
- 3. Stevens RD, Dowdy DW, Michaels RK, Mendez-Tellez PA, Pronovost PJ, Needham DM. Neuromuscular dysfunction acquired in critical illness: a systematic review. Intensive Care Med 2007;33:1876-91.ArticlePubMed
- 4. Ali NA, O’Brien JM Jr, Hoffmann SP, Phillips G, Garland A, Finley JC, et al. Acquired weakness, handgrip strength, and mortality in critically ill patients. Am J Respir Crit Care Med 2008;178:261-8.ArticlePubMed
- 5. Elliott D, Davidson JE, Harvey MA, Bemis-Dougherty A, Hopkins RO, Iwashyna TJ, et al. Exploring the scope of post-intensive care syndrome therapy and care: engagement of non-critical care providers and survivors in a second stakeholders meeting. Crit Care Med 2014;42:2518-26.ArticlePubMed
- 6. Schweickert WD, Pohlman MC, Pohlman AS, Nigos C, Pawlik AJ, Esbrook CL, et al. Early physical and occupational therapy in mechanically ventilated, critically ill patients: a randomised controlled trial. Lancet 2009;373:1874-82.ArticlePubMedPMC
- 7. Burtin C, Clerckx B, Robbeets C, Ferdinande P, Langer D, Troosters T, et al. Early exercise in critically ill patients enhances short-term functional recovery. Crit Care Med 2009;37:2499-505.ArticlePubMed
- 8. Needham DM, Truong AD, Fan E. Technology to enhance physical rehabilitation of critically ill patients. Crit Care Med 2009;37(10 Suppl):S436-41.ArticlePubMed
- 9. Poulsen JB, Møller K, Jensen CV, Weisdorf S, Kehlet H, Perner A. Effect of transcutaneous electrical muscle stimulation on muscle volume in patients with septic shock. Crit Care Med 2011;39:456-61.ArticlePubMed
- 10. Gerovasili V, Stefanidis K, Vitzilaios K, Karatzanos E, Politis P, Koroneos A, et al. Electrical muscle stimulation preserves the muscle mass of critically ill patients: a randomized study. Crit Care 2009;13:R161. ArticlePubMedPMC
- 11. Kho ME, Molloy AJ, Clarke FJ, Ajami D, Mc-Caughan M, Obrovac K, et al. TryCYCLE: a prospective study of the safety and feasibility of early in-bed cycling in mechanically ventilated patients. PLoS One 2016;11:e0167561.ArticlePubMedPMC
- 12. Kho ME, Martin RA, Toonstra AL, Zanni JM, Mantheiy EC, Nelliot A, et al. Feasibility and safety of in-bed cycling for physical rehabilitation in the intensive care unit. J Crit Care 2015;30:1419.e1-5.ArticlePubMed
- 13. Topp R, Ditmyer M, King K, Doherty K, Hornyak J 3rd. The effect of bed rest and potential of prehabilitation on patients in the intensive care unit. AACN Clin Issues 2002;13:263-76.ArticlePubMed
- 14. Garnacho-Montero J, Amaya-Villar R, García-Garmendía JL, Madrazo-Osuna J, Ortiz-Leyba C. Effect of critical illness polyneuropathy on the withdrawal from mechanical ventilation and the length of stay in septic patients. Crit Care Med 2005;33:349-54.ArticlePubMed
- 15. Leijten FS, Harinck-de Weerd JE, Poortvliet DC, de Weerd AW. The role of polyneuropathy in motor convalescence after prolonged mechanical ventilation. JAMA 1995;274:1221-5.ArticlePubMed
- 16. Griffiths RD, Palmer TE, Helliwell T, MacLennan P, MacMillan RR. Effect of passive stretching on the wasting of muscle in the critically ill. Nutrition 1995;11:428-32.ArticlePubMed
- 17. Routsi C, Gerovasili V, Vasileiadis I, Karatzanos E, Pitsolis T, Tripodaki E, et al. Electrical muscle stimulation prevents critical illness polyneuromyopathy: a randomized parallel intervention trial. Crit Care 2010;14:R74. ArticlePubMedPMC
- 18. Patsaki I, Gerovasili V, Sidiras G, Karatzanos E, Mitsiou G, Papadopoulos E, et al. Effect of neuromuscular stimulation and individualized rehabilitation on muscle strength in intensive care unit survivors: a randomized trial. J Crit Care 2017;40:76-82.ArticlePubMed
- 19. Fan E, Ciesla ND, Truong AD, Bhoopathi V, Zeger SL, Needham DM. Inter-rater reliability of manual muscle strength testing in ICU survivors and simulated patients. Intensive Care Med 2010;36:1038-43.ArticlePubMedPMC
Citations
Citations to this article as recorded by

- Assisted mobilisation in critical patients with COVID-19
M. Polastri, F. Daniele, F. Tagariello
Pulmonology.2024; 30(2): 152. CrossRef - Ultrasound for measurement of skeletal muscle mass quantity and muscle composition/architecture in critically ill patients: A scoping review on studies' aims, methods, and findings
Júlia Lima, Estéfani Foletto, Rafaella C.B. Cardoso, Charlles Garbelotto, Aline P. Frenzel, Juliana U. Carneiro, Larissa S. Carpes, Thiago G. Barbosa-Silva, Maria Cristina Gonzalez, Flávia M. Silva
Clinical Nutrition.2024; 43(1): 95. CrossRef - Current Concepts in Early Mobilization of Critically Ill Patients Within the Context of Neurologic Pathology
Thaís Ferreira Lopes Diniz Maia, Paulo André Freire Magalhães, Dasdores Tatiana Silva Santos, Jorge Luiz de Brito Gomes, Paulo Adriano Schwingel, Aline de Freitas Brito
Neurocritical Care.2024;[Epub] CrossRef - Human skeletal muscle size with ultrasound imaging: a comprehensive review
Masatoshi Naruse, Scott Trappe, Todd A. Trappe
Journal of Applied Physiology.2022; 132(5): 1267. CrossRef - Rehabilitation Therapy after the COVID-19 Era: Focused on
Cardiopulmonary Rehabilitation
Hyung Ik Shin
Annals of CardioPulmonary Rehabilitation.2021; 1(1): 17. CrossRef - Rehabilitation Programs for Bedridden Patients with Prolonged Immobility: A Scoping Review Protocol
Vitor Parola, Hugo Neves, Filipa Margarida Duque, Rafael A. Bernardes, Remy Cardoso, Carla A. Mendes, Liliana B. Sousa, Paulo Santos-Costa, Cândida Malça, Rúben Durães, Pedro Parreira, João Apóstolo, Arménio Cruz
International Journal of Environmental Research and Public Health.2021; 18(22): 12033. CrossRef - Non-paretic lower limb muscle wasting during acute phase is associated with dependent ambulation in patients with stroke
Masafumi Nozoe, Masashi Kanai, Hiroki Kubo, Miho Yamamoto, Shinichi Shimada, Kyoshi Mase
Journal of Clinical Neuroscience.2020; 74: 141. CrossRef - Intensive Care Unit-Acquired Weakness: Not Just Another Muscle Atrophying Condition
Heta Lad, Tyler M. Saumur, Margaret S. Herridge, Claudia C. dos Santos, Sunita Mathur, Jane Batt, Penney M. Gilbert
International Journal of Molecular Sciences.2020; 21(21): 7840. CrossRef - Problems with Rehabilitation for Critically ill Patients
Masaji Nishimura
The Japanese Journal of Rehabilitation Medicine.2019; 56(1): 48. CrossRef - Exploring the Potential Effectiveness of Combining Optimal Nutrition With Electrical Stimulation to Maintain Muscle Health in Critical Illness: A Narrative Review
Selina M. Parry, Lee‐anne S. Chapple, Marina Mourtzakis
Nutrition in Clinical Practice.2018; 33(6): 772. CrossRef
 , Hye Bin Kim1
, Hye Bin Kim1 , Hyunwoo Choi1, Kibum Kim2, Donghyung Lee3, Sungwon Na1
, Hyunwoo Choi1, Kibum Kim2, Donghyung Lee3, Sungwon Na1












 KSCCM
KSCCM

 PubReader
PubReader ePub Link
ePub Link Cite
Cite